Abstract
Purpose
Even though Malassezia yeast may play an important role in the exacerbation of atopic dermatitis (AD), only a few studies of Malassezia infection have been conducted in children with AD. Thus, we compared each of clinical findings, including the severity of head and neck dermatitis and laboratory results depending on specific IgE against Malassezia furfur.
Methods
This cross-sectional study was carried out on 121 children aged 3 months to 18 years between April and July of 2014. Retrospective data was collected using the medical records, and patients were divided into 2 groups according to the presence of Malassezia sensitization.
Results
Specific IgE against Malassezia (group 1) was observed in 28 of all patients (23.1%). Group 1 children were at an older age (9.1±6.9 vs. 2.1±3.7, P<0.001). Group 1 children had a higher SCORing Atopic Dermatitis (SCORAD) index (46.4±21.0 ng/mL vs. 37.2±13.4 ng/mL, P=0.001), and total IgE (1,324.2±1,166.0 IU/mL vs. 209.5±532.5 IU/mL, P<0.001) compared to group 2 children (Malassezia-). In the group 1, the correlation between the Malassezia-specific IgE and 25-hydroxyvitamin D3 was negatively weak (r=–0.106) and not statistically significant (P=0.246). Furthermore, Malassezia-specific IgE and the SCORAD index (r=0.281, P=0.002) or total IgE (r=0.380, P<0.001) were positively correlated.
Figures and Tables
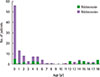
Fig. 2
Correlations of the level of 25-(OH)D (A), SCORAD index (B), and total IgE (C) to Malassezia furfur-specific IgE antibodies. P-value was applied by Pearson correlation. 25-(OH)D, 25-hydroxy vitamin D; sIgE, specific IgE; SCORAD, SCORing Atopic Dermatitis; ImmunoCAP (Phadia AB, Uppsala, Sweden).

Table 1
The clinical characteristics of the two groups classified according to the presence of specific Malassezia IgE
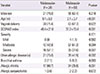
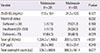
Table 2
The laboratory findings of the two groups classified according to the presence of specific Malassezia IgE
References
1. Spergel JM. Epidemiology of atopic dermatitis and atopic march in children. Immunol Allergy Clin North Am. 2010; 30:269–280.

3. Leung DY, Boguniewicz M, Howell MD, Nomura I, Hamid QA. New insights into atopic dermatitis. J Clin Invest. 2004; 113:651–657.

4. Suh SH. Prevalence of allergic diseases in Korean children, 2010. Public Health Wkly Rep. 2011; 4:425–431.
5. Ring J, Abeck D, Neuber K. Atopic eczema: role of microorganisms on the skin surface. Allergy. 1992; 47(4 Pt 1):265–269.

6. Nikkels AF, Pierard GE. Framing the future of antifungals in atopic dermatitis. Dermatology. 2003; 206:398–400.

7. Bayrou O, Pecquet C, Flahault A, Artigou C, Abuaf N, Leynadier F. Head and neck atopic dermatitis and malassezia-furfur-specific IgE antibodies. Dermatology. 2005; 211:107–113.

8. Darabi K, Hostetler SG, Bechtel MA, Zirwas M. The role of Malassezia in atopic dermatitis affecting the head and neck of adults. J Am Acad Dermatol. 2009; 60:125–136.

9. Zhang E, Tanaka T, Tajima M, Tsuboi R, Kato H, Nishikawa A, et al. Anti-Malassezia-specific IgE antibodies production in Japanese patients with head and neck atopic dermatitis: relationship between the level of specific IgE antibody and the colonization frequency of cutaneous Malassezia species and clinical severity. J Allergy (Cairo). 2011; 2011:645670.

10. Hanifin JM, Rajka G. Diagnostic features of atopic dermatitis. Acta Derm Venereol (Stockh). 1980; 92:Suppl. 44–47.
11. Oranje AP, Glazenburg EJ, Wolkerstorfer A, de Waard-van der Spek FB. Practical issues on interpretation of scoring atopic dermatitis: the SCORAD index, objective SCORAD and the three-item severity score. Br J Dermatol. 2007; 157:645–648.

12. Bischoff-Ferrari HA, Giovannucci E, Willett WC, Dietrich T, Dawson-Hughes B. Estimation of optimal serum concentrations of 25-hydroxyvitamin D for multiple health outcomes. Am J Clin Nutr. 2006; 84:18–28.

13. Ljubojevic S, Skerlev M, Lipozencic J, Basta-Juzbasic A. The role of Malassezia furfur in dermatology. Clin Dermatol. 2002; 20:179–182.

14. Gueho E, Midgley G, Guillot J. The genus Malassezia with description of four new species. Antonie Van Leeuwenhoek. 1996; 69:337–355.

15. Pedrosa AF, Lisboa C, Goncalves Rodrigues A. Malassezia infections: a medical conundrum. J Am Acad Dermatol. 2014; 71:170–176.

16. Johansson C, Sandstrom MH, Bartosik J, Sarnhult T, Christiansen J, Zargari A, et al. Atopy patch test reactions to Malassezia allergens differentiate subgroups of atopic dermatitis patients. Br J Dermatol. 2003; 148:479–488.

17. Kato H, Sugita T, Ishibashi Y, Nishikawa A. Detection and quantification of specific IgE antibodies against eight Malassezia species in sera of patients with atopic dermatitis by using an enzyme-linked immunosorbent assay. Microbiol Immunol. 2006; 50:851–856.

18. Casagrande BF, Fluckiger S, Linder MT, Johansson C, Scheynius A, Crameri R, et al. Sensitization to the yeast Malassezia sympodialis is specific for extrinsic and intrinsic atopic eczema. J Invest Dermatol. 2006; 126:2414–2421.

19. Tengvall Linder M, Johansson C, Zargari A, Bengtsson A, van der Ploeg I, Jones I, et al. Detection of Pityrosporum orbiculare reactive T cells from skin and blood in atopic dermatitis and characterization of their cytokine profiles. Clin Exp Allergy. 1996; 26:1286–1297.

20. Lange L, Alter N, Keller T, Rietschel E. Sensitization to Malassezia in infants and children with atopic dermatitis: prevalence and clinical characteristics. Allergy. 2008; 63:486–487.

21. Selander C, Zargari A, Mollby R, Rasool O, Scheynius A. Higher pH level, corresponding to that on the skin of patients with atopic eczema, stimulates the release of Malassezia sympodialis allergens. Allergy. 2006; 61:1002–1008.

22. Koyama T, Kanbe T, Ishiguro A, Kikuchi A, Tomita Y. Antigenic components of Malassezia species for immunoglobulin E antibodies in sera of patients with atopic dermatitis. J Dermatol Sci. 2001; 26:201–208.

23. Aspres N, Anderson C. Malassezia yeasts in the pathogenesis of atopic dermatitis. Australas J Dermatol. 2004; 45:199–205.

24. Ramasastry P, Downing DT, Pochi PE, Strauss JS. Chemical composition of human skin surface lipids from birth to puberty. J Invest Dermatol. 1970; 54:139–144.

25. Devos SA, van der Valk PG. The relevance of skin prick tests for Pityrosporum ovale in patients with head and neck dermatitis. Allergy. 2000; 55:1056–1058.

26. Brodska P, Panzner P, Pizinger K, Schmid-Grendelmeier P. IgE-mediated sensitization to malassezia in atopic dermatitis: more common in male patients and in head and neck type. Dermatitis. 2014; 25:120–126.
27. Peroni DG, Piacentini GL, Cametti E, Chinellato I, Boner AL. Correlation between serum 25-hydroxyvitamin D levels and severity of atopic dermatitis in children. Br J Dermatol. 2011; 164:1078–1082.

28. Chiu YE, Havens PL, Siegel DH, Ali O, Wang T, Holland KE, et al. Serum 25-hydroxyvitamin D concentration does not correlate with atopic dermatitis severity. J Am Acad Dermatol. 2013; 69:40–46.

29. Zargari A, Midgley G, Back O, Johansson SG, Scheynius A. IgE-reactivity to seven Malassezia species. Allergy. 2003; 58:306–311.

30. Lindgren L, Wahlgren CF, Johansson SG, Wiklund I, Nordvall SL. Occurrence and clinical features of sensitization to Pityrosporum orbiculare and other allergens in children with atopic dermatitis. Acta Derm Venereol. 1995; 75:300–304.
31. Nissen D, Petersen LJ, Esch R, Svejgaard E, Skov PS, Poulsen LK, et al. IgE-sensitization to cellular and culture filtrates of fungal extracts in patients with atopic dermatitis. Ann Allergy Asthma Immunol. 1998; 81:247–255.

32. Mayser P, Gross A. IgE antibodies to Malassezia furfur, M. sympodialis and Pityrosporum orbiculare in patients with atopic dermatitis, seborrheic eczema or pityriasis versicolor, and identification of respective allergens. Acta Derm Venereol. 2000; 80:357–361.

33. Back O, Scheynius A, Johansson SG. Ketoconazole in atopic dermatitis: therapeutic response is correlated with decrease in serum IgE. Arch Dermatol Res. 1995; 287:448–451.

34. Kaffenberger BH, Mathis J, Zirwas MJ. A retrospective descriptive study of oral azole antifungal agents in patients with patch test-negative head and neck predominant atopic dermatitis. J Am Acad Dermatol. 2014; 71:480–483.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download