Abstract
Purpose
To investigate the causes, clinical features and characteristics of food-dependent exercise-induced anaphylaxis (FDEIA) in Korea.
Methods
A retrospective medical chart review was performed on the patients diagnosed with anaphylaxis between 2007 and 2011 in 14 hospitals in Korea. Cases with FDEIA were subsequently identified among anaphylaxis patients, and subgroup analyses were done to assess clinical characteristics of FDEIA.
Results
A total of 62 subjects with FDEIA (male, 72.6%; aged 16 to 70 years) were enrolled in 10 hospitals. Wheat (69.3%) was the most common cause of FDEIA, followed by meat (8.1%), seafood (6.5%), and vegetables (6.5%). The clinical manifestations were cutaneous (100%), respiratory (64.5%), cardiovascular (61.3%), and gastrointestinal (9.7%), respectively. In severity assessment, approx-imately 40% of FDEIA were classified as severe anaphylaxis. Portable epinephrine autoinjector was prescribed to 17.2% of patients, and about one fifth of the patients experienced redevelopment of anaphylactic symptoms during follow-up period. There was no significant difference of age, gender, latent period, total immunoglobulin E, and past history of allergic disease between patients with severe anaphylaxis group and patients with mild-to-moderate group.
Go to : 
REFERENCES
2. Morita E, Kunie K, Matsuo H. Food-dependent exercise-induced anaphylaxis. J Dermatol Sci. 2007; 47:109–17.

3. Barg W, Medrala W, Wolanczyk-Medrala A. Exercise-induced anaphylaxis: an update on diagnosis and treatment. Curr Allergy Asthma Rep. 2011; 11:45–51.

4. Maulitz RM, Pratt DS, Schocket AL. Exercise-induced anaphylactic reaction to shellfish. J Allergy Clin Immunol. 1979; 63:433–4.

5. Lee BJ, Ban JW, Kim YK, Cho SH, Min KU, Kim YY. Three cases of food-dependent exercise-induced anaphylaxis. Korean J Med. 1998; 54:718–22.
6. Kim MK, Lee KS, Kim MS. Food (wheat flour) dependent exerclse - induced anaphylaxis in asthmatics: related with the amont of wheat flour and exercise. J Asthma Allergy Clin Immunol. 1999; 19:724–9.
7. Shin YS, Youn SH, Kim MC, Choi JH, Suh YJ, Suh CH, et al. A case of wheat-induced anaphylaxis in an adult. J Asthma Allergy Clin Immunol. 2003; 23:539–43.
8. Kim JH, Lee JI, Yang BY, Ahn SJ, Park SJ, Kim S, et al. A case of food-de-pendent exercise-induced anaphylaxis developed only in winter. Korean J Med. 2005; 69:331–5.
9. Choi JH, Lee HB, Ahn IS, Park CW, Lee CH. Wheat-dependent, exercise-induced Anaphylaxis: a successful case of prevention with ketotifen. Ann Dermatol. 2009; 21:203–5.

10. Lee HB, Ahn IS, Choi JH, Park CW. A case of wheat-dependent exercise-induced anaphylaxis. Ann Dermatol. 2009; 21:447–9.

11. Jeon KW, Kim C, Kim YK, Kang MS, Bong JD, Ki SY, et al. A case of parsely dependent sxercise-induced anaphylaxsis. J Asthma Allergy Clin Immunol. 1998; 18:728–32.
12. Yang MS, Lee SH, Kim KM, Kwon HS, Kim DI, Park CH, et al. A Case report of food-dependent exercise-induced anaphylaxis to apples. Korean J Asthma Allergy Clin Immunol. 2006; 26:242–5.
13. Yoon TY, Choi KH, Lee KM, Ahn JY, Kim MK. Crown daisy-dependent exercise-induced anaphylaxis in a patient with mugwort-sensitized polli-nosis. Korean J Asthma Allergy Clin Immunol. 2011; 31:63–6.
14. Kim SM, Yoo SH, Kim MK. A case of squash-dependent exercise-induced anaphylaxis. Korean J Asthma Allergy Clin Immunol. 2011; 31:140–3.
15. Pang SJ, No SJ, Kim DW, Lee SM, Lee EJ, Kim CH, et al. A case report of food-dependent exercise-induced anaphylaxis in a patient who was sen-sitive to pork. Pediatr Allergy Respir Dis. 2012; 22:116–21.

16. Gelincik A, Demirturk M, Yılmaz E, Ertek B, Erdogdu D, Colakoglu B, et al. Anaphylaxis in a tertiary adult allergy clinic: a retrospective review of 516 patients. Ann Allergy Asthma Immunol. 2013; 110:96–100.
17. Ye YM, Kim MK, Kang HR, Kim TB, Sohn SW, Koh YI, et al. Anaphylaxis in Korean adults: a multicenter retrospective case study. Korean J Asthma Allergy Clin Immunol. 2012; 32(S2):S226.
18. Sampson HA, Munoz-Furlong A, Campbell RL, Adkinson NF Jr, Bock SA, Branum A, et al. Second symposium on the definition and management of anaphylaxis: summary report–Second National Institute of Allergy and Infectious Disease/Food Allergy and Anaphylaxis Network symposium. J Allergy Clin Immunol. 2006; 117:391–7.

19. Brown SG. Clinical features and severity grading of anaphylaxis. J Allergy Clin Immunol. 2004; 114:371–6.

20. Romano A, Di Fonso M, Giuffreda F, Papa G, Artesani MC, Viola M, et al. Food-dependent exercise-induced anaphylaxis: clinical and laboratory findings in 54 subjects. Int Arch Allergy Immunol. 2001; 125:264–72.

21. Aihara Y, Takahashi Y, Kotoyori T, Mitsuda T, Ito R, Aihara M, et al. Fre-quency of food-dependent, exercise-induced anaphylaxis in Japanese ju-nior-high-school students. J Allergy Clin Immunol. 2001; 108:1035–9.

22. Morita E, Matsuo H, Chinuki Y, Takahashi H, Dahlstrom J, Tanaka A. Food-dependent exercise-induced anaphylaxis -importance of omega-5 gliadin and HMW-glutenin as causative antigens for wheat-dependent exercise-induced anaphylaxis-. Allergol Int. 2009; 58:493–8.
23. Koh YI, Choi IS, Chung SU, Cho S. Clinical features of adult patients with anaphylaxis associated with food in Gwangju and Chonnam area. Korean J Asthma Allergy Clin Immunol. 2004; 24:217–23.
24. Kim MJ, Choi GS, Um SJ, Sung JM, Shin YS, Park HJ, et al. Anaphylaxis; 10 years' experience at a university hospital in Suwon. Korean J Asthma Allergy Clin Immunol. 2008; 28:298–304.
25. Yang MS, Lee SH, Kim TW, Kwon JW, Lee SM, Kim SH, et al. Epidemio-logic and clinical features of anaphylaxis in Korea. Ann Allergy Asthma Immunol. 2008; 100:31–6.

26. Palosuo K, Varjonen E, Kekki OM, Klemola T, Kalkkinen N, Alenius H, et al. Wheat omega-5 gliadin is a major allergen in children with immediate allergy to ingested wheat. J Allergy Clin Immunol. 2001; 108:634–8.
27. Park HJ, Kim JH, Kim JE, Jin HJ, Choi GS, Ye YM, et al. Diagnostic value of the serum-specific IgE ratio of ω-5 gliadin to wheat in adult patients with wheat-induced anaphylaxis. Int Arch Allergy Immunol. 2012; 157:147–50.

28. Shadick NA, Liang MH, Partridge AJ, Bingham C, Wright E, Fossel AH, et al. The natural history of exercise-induced anaphylaxis: survey results from a 10-year follow-up study. J Allergy Clin Immunol. 1999; 104:123–7.

29. Tanaka S. An epidemiological survey on food-dependent exercise-induced anaphylaxis in kindergartners, schoolchildren and junior high school students. Asia Pac J Public Health. 1994; 7:26–30.

Go to : 
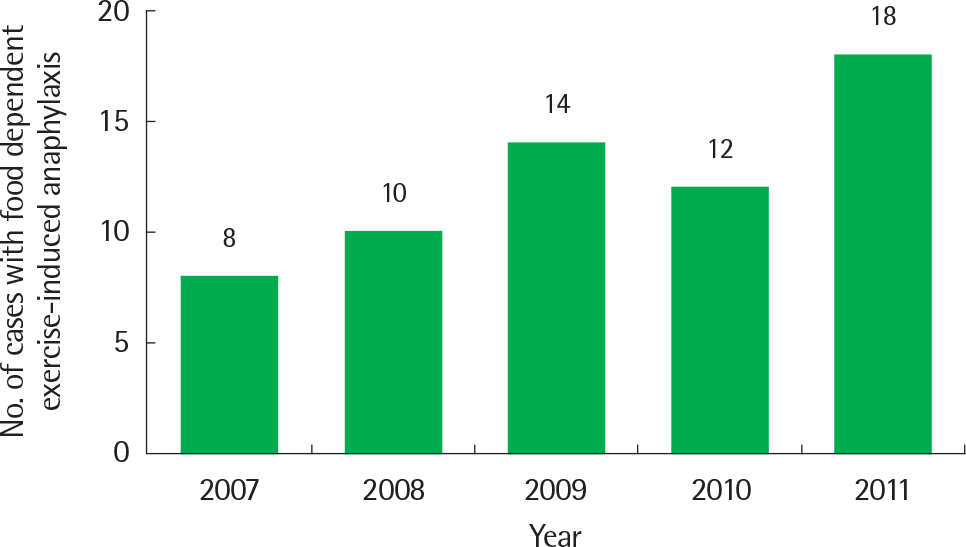 | Fig. 1.The number of patients diagnosed with food-dependent exercise-induced anaphylaxis from 2007 to 2011. Although small drop was noted in 2010, increased tendency in the number of patients diagnosed with food-dependent exercise-induced anaphylaxis was observed during this period. |
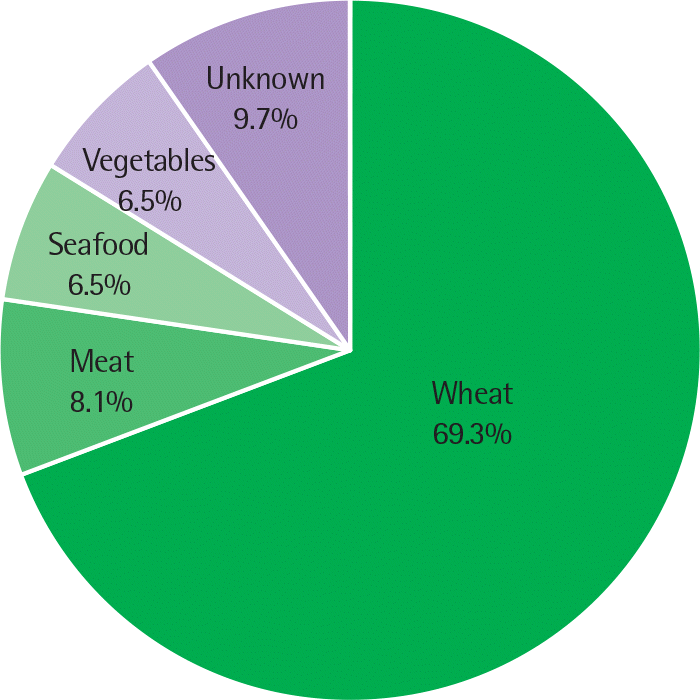 | Fig. 2.The causative foods for patients with food-dependent exercise-induced anaphylaxis. Wheat was the most common cause of food-dependent exercise-induced anaphylaxis in Korea. |
Table 1.
Clinical characteristics of subjects
Table 2.
Clinical manifestation of food dependent exercise-induced anaphylaxis
Table 3.
Treatment and prognosis of food dependent exercise-induced anaphylaxis
| Variable | No. of subjects (%) |
|---|---|
| Hospital visit and care | |
| Emergency room | 26/58 (44.8) |
| Ward admission | 6/58 (10.3) |
| ICU care | 1/58 (1.7) |
| Outpatient clinic | 25/58 (43.1) |
| Treatment with | |
| Epinephrine injection | 6/44 (13.6) |
| Systemic steroid | 33/47 (70.2) |
| H1 receptor antagonist | 36/47 (76.6) |
| H2 receptor antagonist | 16/45 (35.6) |
| Prescription of epinephrine autoinjector∗ | 10/58 (17.2) |
| Recurrence during follow-up period | 6/32 (18.8) |
Table 4.
Comparison of clinical parameters between severe and mild-moderate anaphylaxis
| Variable | Mild to moderate anaphylaxis (n=37) | Severe anaphylaxis (n=25) | P-value |
|---|---|---|---|
| Age (yr) | 30.2±11.6 | 37.2±16.1 | 0.069 |
| Sex (male/female) | 29/8 | 16/9 | 0.213 |
| Log (total immunoglobulin E) (IU/mL) | 2.37±0.55 | 2.38±0.55 | 0.972 |
| Past history of allergic disease | 14/25 (56.0) | 14/21 (66.7) | 0.460 |
| Latent period (min)∗ | 72.7±80.5 | 51.6±44.9 | 0.370 |
| Clinical manifestation | |||
| Cutaneous symptom | 37 (100) | 25 (100) | |
| Respiratory symptom | 26 (70.3) | 14 (56.0) | 0.249 |
| Cardiovascular symptom | 14 (37.8) | 24 (96.0) | 0.001 |
| Gastrointestinal symptom | 4 (10.8) | 2 (8.0) | 1.000 |
| General symptom | 9 (24.3) | 6 (24.0) | 0.977 |
| Wheat-dependency | 26 (70.3) | 17 (68.0) | 0.849 |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download