Abstract
Objective
Acute subdural hematoma (ASDH) patients are treated conservatively or surgically according to the guidelines for surgical treatment. Many patients with thin ASDH and mild neurologic deficit are managed conservatively, but sometimes aggravation of thin ASDH to chronic subdural hematoma (CSDH) results in exacerbated clinical symtoms and consequently requires surgery. The aim of this study is to evaluate risk factors that indicate progression of initially non-operated ASDH to CSDH.
Methods
We divided 177 patients, presenting with ASDH (managed conservatively initially) between January 2008 to December 2013, into two groups; 'CSDH progression group' (n=16) and 'non-CSDH progression group' (n=161). Patient's data including age, sex, past medical history, medication were collected and brain computed tomography was used for radiologic analysis.
Results
Our data demonstrated that no significant intergroup difference with respect to age, sex ratio, co-morbid conditions, medication history, ischemic heart disease, liver disease and end-stage renal disease was found. However, Hounsfield unit (hematoma density) and mixed density was higher in the 'ASDH progression group' (67.50±7.63) than in the 'non-CSDH progression group' (61.53±10.69) (p=0.031). Midline shifting and hematoma depth in the 'CSDH progression group' were significantly greater than the 'non-CSDH progression group' (p=0.067, p=0.005).
Conclusion
Based on the results of this study, the risk factors that are related to progression of initially non-operated ASDH to CSDH are higher Hounsfield unit and hematoma depth. Therefore, we suggest that ASDH patients, who have bigger hematoma depth and higher Hounsfield unit, should be monitored and managed carefully during the follow-up period.
Go to : 
Acute subdural hematoma (ASDH) is a neurosurgical disease that is most strongly associated with traumatic brain injury and has estimated mortality rate between 40% to 60%.4) It occurs in up to 29% of patients who have traumatic brain injury.7) Based on available guidelines for surgical management, ASDH patients with altered mental status, hematoma depth greater than 10 mm, and midline shifting greater than 5 mm usually need emergency surgical evacuation.1) On the other hand, many patients with thin ASDH and mild neurologic deficit are managed conservatively.5) However, the natural course of ASDH is different among patients so although most resolve spontaneously, a few do not disappear and progress to chronic subdural hematoma (CSDH) requiring surgical treatment. Progression of ASDH to CSDH is a common cause of clinical deterioration in patients with initially non-operated ASDH. Therefore, it is important to evaluate the risk factors related to hematoma progression to CSDH in ASDH patients, who were managed conservatively at the time of admission. The aim of this study is to assess risk factors that might help physicians to predict hematoma progression to CSDH in initially non-operated ASDH patients.
Go to : 
We retrospectively collected 313 patients with ASDH in Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine from January 2008 to December 2013. Of these 313 patients, 122 patients who had urgent craniotomy and hematoma evacuation within 72 hours of admission were excluded. In addition, 14 patients were also excluded due to the following reasons; 1) spontaneous ASDH, 2) expired due to co-morbid conditions or complications, 3) vascular abnormalities, 4) bilateral ASDH, 5) younger than 15 years of age, 6) surgical intervention between 72 hours and 21 days after ASDH event, and 7) discharged or transferred before recovery.
In conclusion, 177 patients met the inclusion criteria for this study. After admission, patients were treated with medications including: osmotic agent such as mannitol, anticonvulsant and hypertensive drugs, if required. Treatment with vitamin K and fresh-frozen plasma and platelet concentrates was given to patients with abnormal coagulation profiles (platelet <50,000 mm3 and international normalized ratio >1.4). If patients were on antiplatelet, anticoagulation therapy, or both, these treatments were discontinued for at least 7 days from the date of diagnosis. These patients were later referred to doctors who diagnosed their comorbid diseases related to coagulation profile abnormality, such as ischemic heart disease and cerebral ischemia.
Repeated follow-up brain computed tomography (CT) scans were performed routinely in all patients at approximately; 24 hours, 1 week, 2 weeks, 1 month, 2 months, and 3 months after admission. Emergency CT scans were taken in patients, who presented with unexpected neurological signs or symptoms.
Patients with stable neurological status without significant increase in hematoma volume maintained the original conservative treatment regime. On the other hand, patients with pathological radiographic features, including hematoma enlargement leading to mass effect, midline shifting or herniation, or those who showed aggravating neurological symptoms that were unresponsive to medical treatment underwent surgery. Emergency burr hole drainage was performed at the possible earliest time and subdural drainage, with standard silicone drains that were connected to soft collection bags, was placed at dependent position for a minimum of 24 hours.
The following patient data were collected and analyzed: sex, age, co-morbid conditions including hypertension, diabetes mellitus, renal disease, ischemic heart disease, liver disease, stroke, cancer, epilepsy, or alcohol abuse, medication history of aspirin, antiplatelet drug, or anticoagulation therapy (e.g., warfarin).
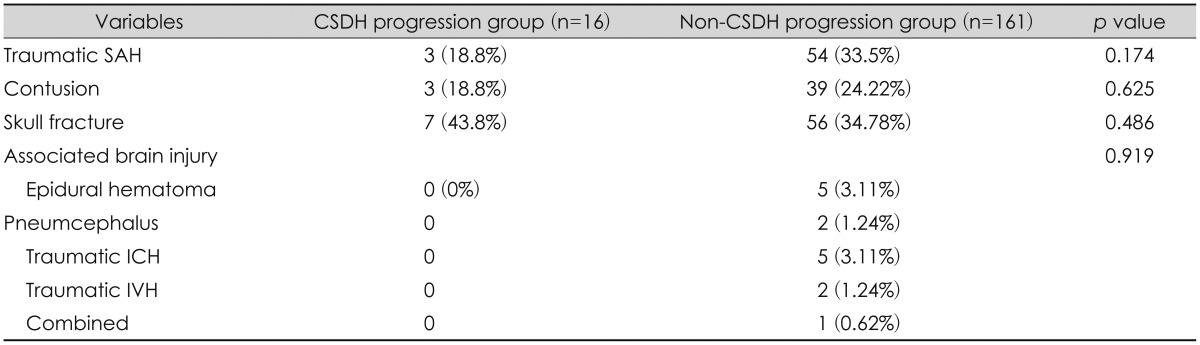
Diagnosis of intracranial injuries including cerebral contusion, subarachnoid hemorrhage (SAH), skull fracture, pneumocephalus, epidural hemorrhage, intraparenchymal hemorrhage (IPH), or intraventricular hemorrhage were gathered from CT scans.
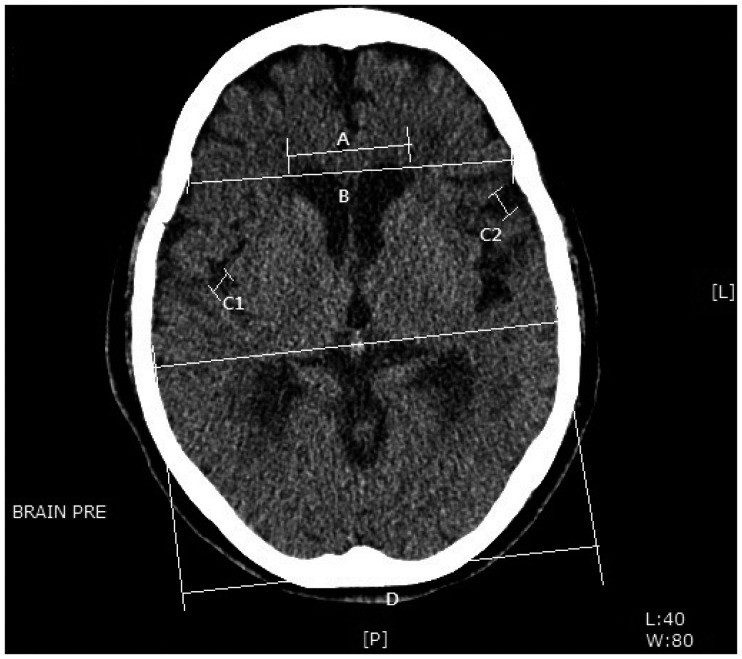
Cerebral contusion was defined as petechial-appearing and ill-defined areas of mixed attenuation on CT scans, and IPH was defined as well-circumscribed, solid-looking, isolated, hematoma. SAHs included both cisternal and sulcal types. Brain atrophy was evaluated with frontal horn index (FHI) and sylvian fissure ration (SFR) (Figure 1).14)
We obtained brain CT scans at initial admission, 2 to 24 hours after admission, within 7 days after admission and at times when symptoms worsened. CT sections (5 mm slices) were obtained in an axial plane parallel to the orbitomeatal line.
The following findings were evaluated from the initial brain CT; location of hematoma (e.g., right or left), maximum thickness of hematoma, and midline shifting. Midline shifting was defined as distance between the most displaced midline structure and the midline of the skull. Initial hematoma density was measured in Hounsfield unit on brain CT scan.
At first, we divided two groups ('CSDH progression group' and 'non-CSDH progression group') according to receiving operation or not. Then, we analyzed variables to find out the risk factors associated with the progression of ASDH to CSDH. Statistical analysis was performed using PASW statistics 18.0 (IBM, Armonk, NY, USA). Data were presented in mean±standard deviation, and statistical significance was defined as p<0.05 for all comparisons. Statistical significance was analyzed with chi-square test or Fisher's exact for categorical variables and t-test or Mann-Whitney test for continuous variables.
Go to : 
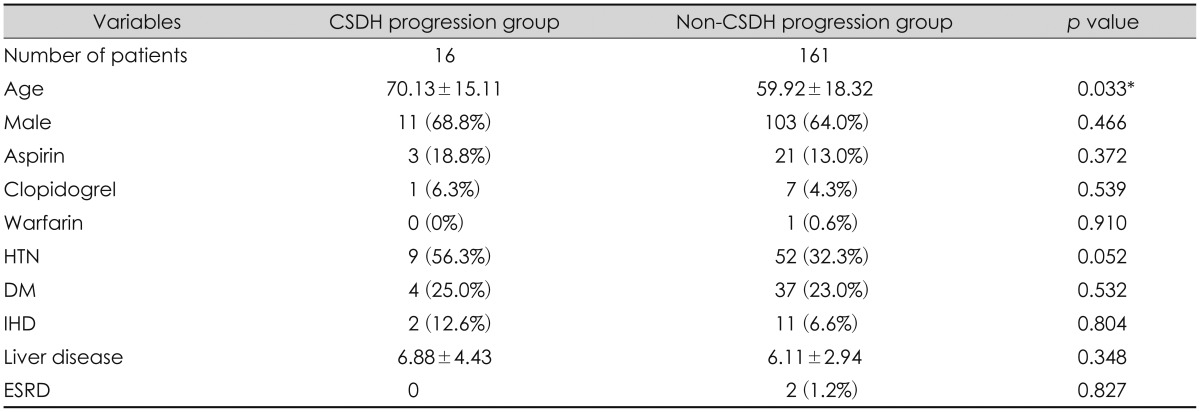
Patient data are listed in Table 1. The study population included 114 men (64.40%) and 63 women (35.60%) and the mean age of the patients was 60.84 years. 'Non-CSDH progression group' included 161 patients, who received initial conservative treatment and did not develop significant CSDH, not requiring surgical evacuation during follow up. 'CSDH progression group' included 16 patients, who were also treated conservatively at first, but progressed to CSDH needing surgical evacuation. Mean age of patients in the 'CSDH progression group' was 70.13 years, and that of patients in the 'non-CSDH progression group' was 59.92 years. No significant intergroup difference was found in age and sex ratio. In terms of co-morbid conditions, such as ischemic heart disease, liver disease and end-stage renal disease, and medication history, no significant intergroup differences were found.
According to the admission lab data, erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) were a little high in the 'CSDH progression group' but were not statistically significant. On the other hand, white blood cell (WBC) was a little higher in the 'non-CSDH group' but this was also not statistically significant.
From CT evaluation, it was evident that many patients also had additional intracranial injuries along with subdural hematoma (SDH) (Table 2).
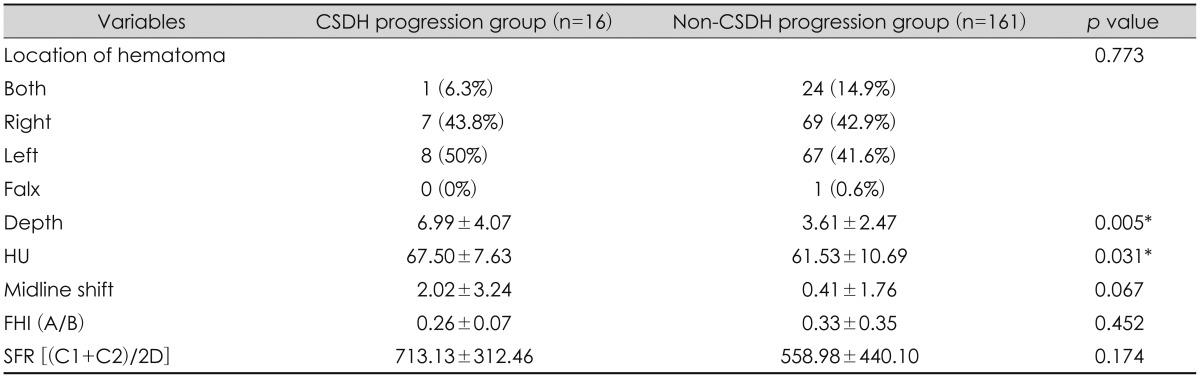
The location of hematoma (right or left) was not significantly different between the two groups. Hounsfield unit (hematoma density) and mixed density were much higher in the 'CSDH progression group' (67.50±7.63) than the 'non-CSDH progression group' (61.53±10.69) (p=0.031) (Table 2). Midline shifting, and hematoma thickness in the 'CSDH progression group' were significantly greater than in the 'non-CSDH progression group' (p=0.067, p=0.005). Hematoma thickness showed significant correlation with CSDH progression, but no significant correlation was seen in midline shifting (Table 3).
When evaluating brain atrophy, SFR and FHI were used and they were higher in CSDH progression group than the other, but were not statistically significant.
Go to : 
ASDH has high reported mortality rate between 40% to 60%.4) Based on patient's clinical and radiologic signs, emergency surgical decompression may be mandatory. Consequent occurrence of intracranial hypertension due to ASDH can be reduced by surgical intervention.13)
Generally, hematoma with thickness greater than 10 mm, or a midline shifting greater than 5 mm are suggested as critical indicators for surgical removal of ASDH regardless of the Glasgow Coma Scale scores.1) However, when these criteria are not met, ASDH does not require surgical treatment as it can resolve spontaneously. Conservative management is possible, if the patient shows mild neurologic deficit or brain CT does not show any mass effect.312) However, of these cases, some of them aggravate to form ASDH in the chronic healing stage or progress to CSDH, requiring surgical intervention.16) Therefore, it is important to evaluate risk factors that indicate progression of ASDH into forming ASDH or CSDH.
According to Yamashima and Yamamoto's study,1617) on average, ASDH in the chronic healing stage was diagnosed 21 days after the initial event while CSDH was diagnosed 71 days after. The two are two different disease entities as they present different histological and clinical signs. However, histological features of ASDH in the chronic healing stage are similar to that of early CSDH, so we assumed that ASDH in the chronic healing stage and CSDH are on the same line. Therefore, cases where surgical intervention was done 3 weeks after initial ASDH event were included and those done within 3 weeks were excluded.
Until today, only few studies have specifically addressed the issue of the rate of CSDH progression after conservative treatment of trauma-related ASDH. Generally, 3% to 26% of patients with ASDH, who were managed conservatively, developed chronic SDHs requiring evacuation.3910)
The results of our study show that over 90.96% of ASDHs resolved spontaneously or had minimal remaining hematoma at the median of 12 weeks post mild head injury event, while 9.04% of the ASDHs increased in size and required hematoma removal. We found that two prognostic factors, including the initial volume of the ASDH and the Hounsfield unit on the initial brain CT scan, were independently associated with necessity of delayed hematoma evacuation in patients with progressive CSDH.
Mathew et al.10) noted that all patients with an acute hematoma (thickness greater than 10 mm), who were initially managed conservatively, required subsequent burr-hole drainage. In our study, we found that higher initial hematoma volume was a risk factor associated with ASDH patients who progressed to CSDH. The mean acute hematoma thickness was 6.99±4.07 mm in the operated group compared to 3.61±2.47 mm in the non-operated group, and this difference remained statistically significant in univariate analysis. Considering the relationship between initial hematoma size and subsequent progression, it has been suggested that a small ASDH is likely to be more stable, whereas a larger one is likely to progress over time. The reason for this is that a large hematoma increases the risk for bridging vein tears, resulting in delayed hematoma progression. Another cause for this might be due to the microbleeding from the hematoma membrane. It is possible that initially large SDHs require more time for resolution and that they may have a higher chance for developing neomembranes, which develop due to inflammatory reaction.
Relatively higher hematoma density was more common in the 'CSDH-progression group' (67.50±7.63) than the 'non-CSDH progression group' (61.53±10.69) (p=0.031) (Table 3). This may be due to the fact that there was more hematoma in the higher density group, resulting in longer and more extensive inflammation response causing more frequent progression to CSDH.
CSDH is known to be caused by trauma but clear history of trauma is evident in only about half of the patients. Many CSDH starts as an acute hematoma but as blood within the subdural space evokes inflammatory response, fibroblasts invade the clot and form neomembranes on the inner (cortical) and outer (dural) surface of the hematoma. This event is followed by ingrowth of neo-capillaries, enzymatic fibrinolysis, and liquefaction of blood clot. Fibrin degradation products are re-incorporated into new clots and further inhibit hemostasis. The course of CSDH is determined by the balance of plasma effusion and/or re-bleeding from the neomembranes and re-absorption of fluid.2) Surgical evacuation of CSDH is indicated for symptomatic lesions, such as focal neurological deficit and mental status change, or for hematoma collections with maximum thickness greater than 1 cm. However, in this study inflammation markers such as WBC, ESR, and CRP did not show any statistical significance between the two groups.
Increased use of anticoagulants and antiplatelet agents may have changed the incidence of intracerebral hemorrhage and subdural hematomas.11) However, the relationship between these drugs and the development of CSDH has been studied mainly in the context of recurrent CSDHs. Rust et al.11) reported that there is 42.5 times higher possibility of developing CSDH when taking warfarin compared to cases where no medication was taken. In another study, while investigating the predictors for recurrent CSDH, Torihashi et al.15) found that out of 343 surgical cases of CSDH, 61 patients had recurrent CSDH. Univariate and multivariate analyses revealed that although antiplatelet and anticoagulant therapy had no significant effect on recurrence of CSDH, the time interval between the injury and the first operation for patients on antiplatelet and/or anticoagulant therapy was shorter than that of patients without it (29.9 days vs. 44.2 days). In the recent Laviv and Rappaport's study,8) there was higher risk of CSDH progression in ASDH patients receiving conservative management who were using antiplatelet or anticoagulative agents. However, our study did not show that the use of aspirin alone or with combination with other anti-coagulant therapy was associated with late development of CSDH requiring surgical evacuation. This may be due to small number of sample size collected for this study.
Regarding other bleeding tendencies and the risk for CSDH, Ko et al.6) found that bleeding tendencies such as in leukemia, liver disease, and chronic renal failure were significantly associated with recurrence of CSDH (p=0.037). Likewise, in Laviv and Rappaport's study,8) clinically the risk of progression to the CSDH group was also higher in the above patient group. However, in this study, we could not find significant association with this progression and various bleeding tendencies in both groups. This is also probably due to the small number of sample size.
Go to : 
Based on the result of this study, the risk factors related to the progression of initially non-operated ASDH to CSDH were identified as; bigger hematoma depth and relatively higher Hounsfield unit. Our study suggests that ASDH patients, who have thick hematoma depth should be carefully observed and managed appropriately to prevent the progression to CSDH and the need for delayed invasive surgical treatment.
Go to : 
References
1. Bullock MR, Chesnut R, Ghajar J, Gordon D, Hartl R, Newell DW, et al. Surgical management of acute subdural hematomas. Neurosurgery. 2006; 58(3 Suppl):S16–S24. discussion Si-SivPMID: 16710968.

2. Drapkin AJ. Chronic subdural hematoma: pathophysiological basis for treatment. Br J Neurosurg. 1991; 5:467–473. PMID: 1764228.

3. Feliciano CE, De Jesús O. Conservative management outcomes of traumatic acute subdural hematomas. P R Health Sci J. 2008; 27:220–223. PMID: 18782966.
4. Kalanithi P, Schubert RD, Lad SP, Harris OA, Boakye M. Hospital costs, incidence, and inhospital mortality rates of traumatic subdural hematoma in the United States. J Neurosurg. 2011; 115:1013–1018. PMID: 21819196.

5. Karibe H, Hayashi T, Hirano T, Kameyama M, Nakagawa A, Tominaga T. Surgical management of traumatic acute subdural hematoma in adults: a review. Neurol Med Chir (Tokyo). 2014; 54:887–894. PMID: 25367584.

6. Ko BS, Lee JK, Seo BR, Moon SJ, Kim JH, Kim SH. Clinical analysis of risk factors related to recurrent chronic subdural hematoma. J Korean Neurosurg Soc. 2008; 43:11–15. PMID: 19096538.

7. Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain injury: a brief overview. J Head Trauma Rehabil. 2006; 21:375–378. PMID: 16983222.
8. Laviv Y, Rappaport ZH. Risk factors for development of significant chronic subdural hematoma following conservative treatment of acute subdural hemorrhage. Br J Neurosurg. 2014; 28:733–738. PMID: 24836959.

9. Lindvall P, Koskinen LO. Anticoagulants and antiplatelet agents and the risk of development and recurrence of chronic subdural haematomas. J Clin Neurosci. 2009; 16:1287–1290. PMID: 19564115.

10. Mathew P, Oluoch-Olunya DL, Condon BR, Bullock R. Acute subdural haematoma in the conscious patient: outcome with initial non-operative management. Acta Neurochir(Wien). 1993; 121:100–108. PMID: 8512003.

11. Rust T, Kiemer N, Erasmus A. Chronic subdural haematomas and anticoagulation or anti-thrombotic therapy. J Clin Neurosci. 2006; 13:823–827. PMID: 16997707.

12. Servadei F, Nasi MT, Cremonini AM, Giuliani G, Cenni P, Nanni A. Importance of a reliable admission Glasgow Coma Scale score for determining the need for evacuation of posttraumatic subdural hematomas: a prospective study of 65 patients. J Trauma. 1998; 44:868–873. PMID: 9603091.
13. Shima K, Aruga T, Onuma T, Shigemori M. members of the Japanese Guidelines Committee on the Management of Severe Head Injury (2nd Edition), and the Japan Society of Neurotraumatology. JSNT-Guidelines for the Management of Severe Head Injury (Abridged edition). Asian J Neurosu. 2010; 5:15–23.
14. Son S, Yoo CJ, Lee SG, Kim EY, Park CW, Kim WK. Natural course of initially non-operated cases of acute subdural hematoma: the risk factors of hematoma progression. J Korean Neurosurg Soc. 2013; 54:211–219. PMID: 24278650.
15. Torihashi K, Sadamasa N, Yoshida K, Narumi O, Chin M, Yamagata S. Independent predictors for recurrence of chronic subdural hematoma: a review of 343 consecutive surgical cases. Neurosurgery. 2008; 63:1125–1129. discussion 1129PMID: 19008766.
16. Yamashima T, Yamamoto S. Clinicopathological study of acute subdural haematoma in the chronic healing stage. Clinical, histological and ultrastructural comparisons with chronic subdural haematoma. Neurochirurgia (Stuttg). 1984; 27:98–105. PMID: 6483072.
17. Yamashima T, Yamamoto S. How do vessels proliferate in the capsule of a chronic subdural hematoma? Neurosurgery. 1984; 15:672–678. PMID: 6209593.

Go to : 




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download