Abstract
Pulmonary mucoepidermoid carcinoma (MEC) is a rare form of lung cancer that originates from submucosal glands of tracheobronchial tree. Unlike low-grade tumor with benign nature, high-grade case is even rarer and has aggressive clinical features with no definite treatment option. Here, we report a case of high-grade pulmonary MEC with fulminant clinical course. A 74-year-old man presented with cough, sputum and mental change. Chest imaging showed massive mediastinal lymphadenopathy with obstructive pneumonia, and multiple metastases in lung and adrenal gland. Bronchoscopy showed polypoid masses obstructing right main bronchus and bronchus intermedius. Histopathology revealed a mixture of glandular structure lined with mucussecreting cells and nests of squamoid cells with nuclear atypia and pleomorphism, which is compatible with high-grade MEC. We intensively treated the patient with combination antibiotics and ventilator care. However, the patient did not respond to the treatment and rapidly deteriorated, and finally expired a month after diagnosis.
Mucoepidermoid carcinoma (MEC) of lung is a rare form of lung tumor and accounts for less than 0.2% of all lung malignancies [1]. It is believed to originate from serous and mucus glands of tracheobronchial tree, and most cases arise from trachea and proximal bronchi [2]. Its histological features are similar to those of MEC of salivary glands, which are characterized by a mixture of mucus-secreting cells, squamoid or epidermoid cells and intermediate cells [3].
Pulmonary MEC patients typically present with symptoms related to bronchial irritation or obstruction, such as cough, hemoptysis, wheezing and obstructive pneumonia [4]. Most cases are low-grade disease and are curable with surgical resection. However, the high-grade tumor, which occurs less frequently than low-grade tumor, often requires adjuvant chemotherapy or radiation therapy. In contrast to the low-grade tumor whose prognosis is excellent, the high-grade tumor has poor prognosis even with an aggressive treatment [5]. We experienced a patient of high-grade MEC with obstructive pneumonia and multiple metastases at presentation, who expired a month after the diagnosis despite intensive treatment. We report this case with a review of the pertinent literature.
A 74-year-old male visited our emergency department via ambulance with a complaint of mental change which developed in the morning. The patient was a former smoker (1 pack per day for 30 years) who had quit smoking 20-years ago and had no other specific medical history. According to his wife, the patient had been complaining of cough and sputum for several months and had taken over-the-counter medication without hospital visit. At the emergency room, the initial blood pressure was 100/70 mmHg; body temperature was 38.5℃; pulse rate was 121 per minute; and respiration rate was 24 per minute. The patient was stuporous and disoriented. Decreased lung sound and rhonchi were heard in chest auscultation. On the laboratory test, hemoglobin level was 10.5 g/dL; white blood cell count was 42,200/µL (neutrophil 89%); platelet count was 254,000/µL; and high-sensitivity C-reactive protein was 170.5 mg/L (normal range, 0 to 10 mg/L). Arterial blood gas analysis checked in room air showed pH 7.41, PaCO2 41.7 mmHg, PaO2 46.9 mmHg, HCO3- 24.5 mmol/L, and SaO2 84.5%. Chest radiography showed collapsed right lower lobe (Fig. 1). Chest computed tomography (CT) scan revealed hugely enlarged upper paratracheal and subcarinal lymph nodes, an endobronchial mass associated with atelectasis of right lower lobe and pleural effusion at right hemithorax. Additionally two nodules in left lung and 2 cm-sized right adrenal mass suspicious of metastases were detected (Fig. 2). Brain CT only showed mild ischemic changes of periventricular white matter and age-related atrophic changes. Considering the provisional diagnosis of lung cancer with sepsis due to obstructive pneumonia, a broad spectrum of antibiotics (piperacillin-tazobactam and moxifloxacin) was immediately administered with a supply of oxygen via mask. Bronchoscopy was then performed and demonstrated right vocal cord palsy and extrinsic compression of upper trachea. There were two endobronchial masses at right main bronchus, 3-cm apart from carina and at the orifice of right bronchus intermedius. The masses were about 1-2 cm sized, well-circumscribed hypervascular polypoid tumors (Fig. 3). Pathologic examination revealed a mixture of glandular structure lined with mucussecreting cells and nests or sheets of squamoid cells. The nuclei showed pleomorphism, and about 4-5 mitoses were noted per 10 high power field. Immunohistochemical staining was positive in cytokeratin 5 and 7 and negative in thyroid transcription factor 1 (Fig. 4). The final diagnosis was high-grade pulmonary MEC and the clinical stage of the patient was IV (T4N2M1b). For mutation in the epidermal growth factor receptor (EGFR), the study revealed negative result. Even with the combination antibiotics treatment, the obstructive pneumonia was not improved, and hypoxemia was aggravated. The patient deteriorated to septic shock with multiple organ failure. At hospital day 14, he was moved to intensive care unit with ventilator care and underwent tube thoracostomy due to consecutive pneumothorax developed at right hemithorax. Despite the intensive treatment with antibiotics, vasopressors and continuous renal replacement therapy, the patient's condition worsened, and he was finally expired at hospital day 34.
Pulmonary MEC is a rare form of lung tumor, which comprises 0.1%-0.2% of all lung cancers [4]. Radiologically, the solitary nodule or mass, pneumonic consolidation, and atelectasis may be seen, but such findings are not helpful in discriminating it from benign lung disease or other lung cancers [26]. Chest CT scan helps to identify small endobronchial or endotracheal mass, local invasion, and metastasis. Bronchoscopy is essential for identifying the endobronchial lesion and obtaining the tissue samples for the diagnosis.
Histologically, MEC comprises of a mixture of mucussecreting cells, intermediate cells, and squamoid or epidermoid cells, which are organized into glandular or tubular structure [7]. In contrast to the low-grade tumor, which is characterized by a high proportion of mucus-secreting cells, low proportion of squamoid cells, and an absence of necrosis or nuclear pleomorphism, the high-grade tumor has a high proportion of nest or sheet of squamoid cells with nuclear pleomorphism and mitosis [3]. According to the Armed Forces Institute of Pathology (AFIP) grading system, which is based on a point-score for histopathologic features described above, our case was indicative of high-grade MEC [8].
The high-grade MEC occurs less frequently compared to the low-grade MEC, and it has an aggressive clinical course [6]. High-grade tumors are frequently associated with local invasion and are rarely presented as metastatic disease [9]. In a previous report reviewing 31 pulmonary MEC cases of Korea, only three cases were shown to be high-grade and none had metastatic disease [10]. All three patients underwent surgical resection and adjuvant treatment, but they died several months after the surgery due to disease progression. Another case series of Chinese patients reported that 7 out of 32 pulmonary MEC cases were high-grade and only three had metastatic disease [9]. All of the 7 patients high-grade tumors were treated with surgery plus postoperative chemotherapy or radiotherapy. The chemotherapy regimens used were vinorelbine/cisplatin and docetaxel/cisplatin. Although the prognosis of those patients was poor (5-year survival was 28.6%), no patients died during treatment in the hospital. Compared to the previous reports, our case showed more aggressive biological behavior. Our patient had an obstructive pneumonia with multiple metastases at presentation, which rapidly developed to sepsis; and the patient was not a candidate for surgery or even chemotherapy. To our knowledge, this is the first report demonstrating a fulminant clinical course of pulmonary high-grade MEC which was intractable. Clinicians should be reminded that the high-grade MEC may show more aggressive behavior than previously reported.
The upmost obstacle in the management of high-grade MEC is that there is no consensus for the proper treatment. Previous case series have reported that postoperative chemotherapy and radiation therapy were used, but the clinical usefulness and outcome of adjuvant therapy is not established [9]. Recently, the detection of EGFR mutation in the pulmonary MEC has shed light on the possible use of targeted therapy for this aggressive disease. Han et al. [11] reported that EGFR mutation (L858R) was found in three out of five patients with pulmonary MEC, with one patient showing partial response to EGFR-tyrosine kinase inhibitor (TKI). Two more cases of pulmonary MEC with a good response to EGFR-TKI have been reported [712]. Interestingly, those three cases, which showed clinical response to EGFR-TKI, did not have activating mutations. These data suggest that pulmonary MEC may have additional mechanisms that make the tumor sensitive to EGFR-TKI other than EGFR mutation [11]. In our case, EGFR mutation study was performed but no mutation was found. Retrospectively, the molecular targeted therapy would be the only therapeutic option that could be considered in the present case. Further large study is needed to elucidate if pulmonary MEC is a true candidate target for the treatment with EGFR-TKI.
In conclusion, a high-grade MEC can be presented as a metastatic disease and it may show a highly malignant clinical course. Although there has been no established treatment strategy, recent reports have suggested that EGFR-TKI could be a therapeutic option for metastatic tumors. The present report shows an aggressive clinical feature of high-grade MEC and emphasizes establishing a proper treatment strategy, including targeted therapy for advanced disease, to improve its clinical outcome.
Figures and Tables
Fig. 1
Chest radiography at presentation. It shows increased opacity at right lower lung field and hyperlucent left lung, suggestive of collapsed right lower lobe.
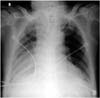
Fig. 2
Chest computed tomography scan. It reveals hugely enlarged upper paratracheal lymph node compressing trachea (A), and subcarinal lymph node and an endobronchial mass arrow associated with atelectasis of right lower lobe and pleural effusion (B). About 2 cm-sized right adrenal mass (C) and a metastatic nodule in left lower lung were also noted (D).
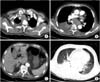
Fig. 3
Bronchoscopic findings. It shows two endobronchial masses arising at right main bronchus and the orifice of right bronchus intermedius. The masses are 1-2 cm-sized, well-circumscribed hypervascular polypoid tumors.
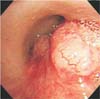
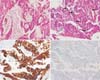
Fig. 4
Histopathologic images. Hematoxylin and eosin staining shows glandular structures with mucus-secreting cells (A), nests of squamoid and intermediated cells (B). Immnuohistochemistry shows positive staining in cytokeratin 7 (C) and negative staining in thyroid transcription factor-1 (D). The nuclei are pleomorphic and show frequent mitosis (arrows), compatible with high-grade mucoepidermoid carcinoma (×200).
References
1. Miller DL, Allen MS. Rare pulmonary neoplasms. Mayo Clin Proc. 1993; 68:492–498.
2. Yousem SA, Hochholzer L. Mucoepidermoid tumors of the lung. Cancer. 1987; 60:1346–1352.
3. Seethala RR. An update on grading of salivary gland carcinomas. Head Neck Pathol. 2009; 3:69–77.
4. Heitmiller RF, Mathisen DJ, Ferry JA, Mark EJ, Grillo HC. Mucoepidermoid lung tumors. Ann Thorac Surg. 1989; 47:394–399.
5. Vadasz P, Egervary M. Mucoepidermoid bronchial tumors: a review of 34 operated cases. Eur J Cardiothorac Surg. 2000; 17:566–569.
6. Chin CH, Huang CC, Lin MC, Chao TY, Liu SF. Prognostic factors of tracheobronchial mucoepidermoid carcinoma: 15 years experience. Respirology. 2008; 13:275–280.
7. Rossi G, Sartori G, Cavazza A, Tamberi S. Mucoepidermoid carcinoma of the lung, response to EGFR inhibitors, EGFR and KRAS mutations, and differential diagnosis. Lung Cancer. 2009; 63:159–160.
8. Goode RK, Auclair PL, Ellis GL. Mucoepidermoid carcinoma of the major salivary glands: clinical and histopathologic analysis of 234 cases with evaluation of grading criteria. Cancer. 1998; 82:1217–1224.
9. Song Z, Liu Z, Wang J, Zhu H, Zhang Y. Primary tracheobronchial mucoepidermoid carcinoma: a retrospective study of 32 patients. World J Surg Oncol. 2013; 11:62.
10. Ha SY, Han J, Lee JJ, Kim YE, Choi YL, Kim HK. Mucoepidermoid carcinoma of tracheobronchial tree: clinicopathological study of 31 cases. Korean J Pathol. 2011; 45:175–181.
11. Han SW, Kim HP, Jeon YK, Oh DY, Lee SH, Kim DW, et al. Mucoepidermoid carcinoma of lung: potential target of EGFRdirected treatment. Lung Cancer. 2008; 61:30–34.
12. Lee KW, Chan AB, Lo AW, Lam KC. Erlotinib in metastatic bronchopulmonary mucoepidermoid carcinoma. J Thorac Oncol. 2011; 6:2140–2141.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download