Abstract
Objectives
The etiology for Kawasaki disease (KD) remains unknown, but several studies have suggested the involvement of immune dysregulation and genetic factors. The purpose of this study is to compare gene expressions before and after an infusion of intravenous immunoglobulin (IVIG) in KD patients.
Methods
Blood was obtained from both acute and sub-acute phases of 4 patients with KD and febrile control children. Blood was collected in PAXgene blood RNA tubes and RNA was extracted using a PAXgene blood RNA isolation kit. Labeled RNAs were analyzed using Roche NimbleGen human whole genome 12-plex array.
Results
KD patients prior to IVIG injection showed more than a two-fold increase in the expression of 88 genes and more than a two-fold decrease in the expression of 98 genes compared to the control group. They also showed more than two-fold increase in the expression of 226 genes and more than a two-fold decrease in 117 genes in KD patients after IVIG treatment compared to the patients before IVIG injection. Through microarray evaluation, the expressions of genes involved in proliferation, translation, inflammatory response, immune response, cell adhesion, cell migration, cell differentiation, apoptosis, cell growth, transport, cell cycle, transcription, signal transduction and metastasis were observed.
Kawasaki disease (KD) is an acute febrile disorder characterized by systemic vasculitis of infants and children, manifested as prolonged fever and signs of mucocutaneous inflammation which are polymorphous skin rashes, injected conjunctiva, erythematous edema in the palms and soles [1,2]. Coronary artery lesions are the most important complication of KD [3,4]. As the current first-line therapy, IVIG, at a dose of 2 g/kg in combination with aspirin, has been shown to reduce the risk of coronary artery complications [5-7]. Even though treatment with IVIG reduces the development of aneurysm or dilatation, these are critical complication to be solved [3,8].
The etiology for KD remains unknown, however, infection, immune response, or genetic susceptibility is considered in the development of KD. The acute phase of KD demonstrates elevated serum levels of proinflammatory cytokines such as tumor necrosis factor (TNF)-α, interleukins (ILs) and endothelial growth factor [6,9]. The degree of elevation of these cytokines may be correlated with coronary aneurysms and subsequent stricture formation. Elevated levels of IL-1 have been reported in acute patients and have been correlated to vascular endothelial cell damage [6,10]. Also, single-nucleotide polymorphisms of inflammatory genes such as C-reactive protein (CRP) and TNF-α are associated with predisposition to KD disease and increased carotid arterial stiffness and intima-media thickness in the long-term [11]. Several studies have suggested the involvement of a genetic factor [12,13]. Matrix metalloproteinases (MMP) is related to focal destruction of the internal elastic lamina of coronary artery and influence recruitment of inflammatory cells. Therefore, MMPs play important roles in both inflammation and tissue remodeling [12,14].
The advantage of DNA microarray analysis is that it can evaluate changes in relative expression of thousands of genes simultaneously [15-17].
The purpose of this study was to investigate the changes of gene expressions by microarray analysis in KD patients after IVIG therapy.
The study group included acute phases for four KD patients and four febrile control children who were admitted to the Ewha Womans University Hospital. All patients met the criteria for the diagnostic guidelines of KD (http://www.kawasaki-disease.org/diagnostic/index.html). Clinical characteristics are fever lasting 5 days and complete blood count (CBC), erythrocyte sedimentation rate (ESR), platelet count, CRP, pro-brain natriuretic peptide (BNP) were significantly higher in the KD group compared to the control group. All KD patients had coronary artery lesions such as dilated coronary artery or coronary artery aneurysm by echocardiography.
All patients were treated with IVIG (2 g/kg/day for 1 day) as a single infusion over 10~12 hours. Fresh whole blood samples were obtained from KD patients pre- and post-IVIG treatment as well as febrile control group who had been febrile (body temperature >38℃) for at least 3 days. Laboratory data were obtained from each child, including CBC, ESR, platelet count, CRP, pro-BNP. And echocardiography was performed by pediatric cardiologists to detect the presence of coronary artery lesions.
Total RNA was extracted from the blood sample that stored for 24hr at room temperature and then in the fridge (-20℃) using a PAXgene blood RNA extraction kit according to the manufacturer's instructions. Each total RNA sample (1 µg) was labeled and amplified using Universal Linkage System (ULS) aRNA labeling kit (Kreatech diagnostics, Amsterdam, Netherlands).
The Cy3-labeled aRNAs were resuspended in 10 µL of hybridization solution (GenoCheck, Ansan, Korea). After labeled aRNA were placed on Roche Nimblegen Human whole genome 12-plex array (Roche Nimble-Gen, Inc., Madison, USA). The slides were hybridized for 12 hr at 42℃ MAUI system (Biomicro systems, Inc., Salt Lake City, USA).
The Roche NimbleGen Human genome 12-plex arrays were analyzed using an Axon GenePix 4000B scanner with associated software (Molecular Devices Corp., Sunnyvale, USA). Gene expression levels were calculated with NimbeScan Version 2.4 (Roche NimbleGen, Inc., Madison, USA). Relative signal intensities for each gene were generated using the Robust Multi-Array Average algorithm. And then the data was analyzed using GeneSpring GX 7.3.1 (Agilent technologies, Santa Clara, USA). Genes were grouped as increased or decreased in the acute phase and also before and after an injection of IVIG. The color red indicated an over expression while green indicated a down expression.
An unpaired two-tailed t-test and a Mann-Whitney test were used, and a P value<0.05 was considered statistically significant. SPSS 14.0 for windows (SPSS, Chicago, USA) was used for all statistical analyses. The two-tailed t-test was used to compare patients' samples obtained before and after IVIG therapy, and ANOVA was used to compare pre- and post-IVIG patients with KD and the control patients.
Expressions of 393 genes in the KD group pre-IVIG treatment were significantly different to those of the control group. The KD group pre-IVIG treatment showed a 1.5-fold increase in the expression of 203 genes (two-fold increase in the expression of 88 genes) and 1.5-fold decrease in the expressions of 190 genes (two-fold decrease in the expression of 96 genes) compared to the control group (Table 1).
Among up-regulated genes (Table 2), three genes (interkeukin-32, leukocyte specific transcript 1, complement component 4 binding protein) were related to immune responses and two genes (chemokine ligand 5, IL-5) were related to inflammation responses. Fourteen genes (chemokine ligand 5, leukocyte specific transcript 1, protocadherin beta 16, roundabout, angiopoietin like 3, tubulin tyrosine ligase, POU domain class 4, transcription factor 3, EPH receptor A2, neurofibromin 2, activating transcription factor 3, GATA zinc finger domain containing 1, CCAAT enhancer binding protein delta, mitogen activated protein kinase 3, dystrobrevin, alpha) are related to cell proliferation process. Among them, POU domain class 4, transcription factor 3 and mitochondrial protein 18 kDa are related to apoptosis. Three genes (nitric oxide synthase (NOS) 1, IL-17A, IL-12A) are nitric oxide (NO) related genes.
Eighteen genes are down-regulated compared with the control group (Table 3). Among down-regulated genes, four genes (major histocompatibility complex, class II, DP beta 1, phosphoprotein associated with glycosphingolipid microdomains 1, activation-induced cytidine deaminase, proteasome subunit, beta type 8) are related to immune response. Neurotrophin 3 and protein disulfide isomerase family are related to apoptosis. Thirteen genes (integrin alpha 7, neurotrophin 3, activation-induced cytidine deaminase, protein disulfide isomerase family A, member 3, N-ethylmaleimide-sensitive factor attachment protein, beta, endosulfine alpha, chloride intracellular channel 5, polycomb group ring finger 5, zinc finger protein 692, zinc finger protein 10, DENN/MADD domain containing 4A, c-myc binding protein, oxoglutarate receptor 1) are related to cell proliferation process (Table 3).
In microarray analysis, 226 genes showed more than two fold up expression and 117 genes were down-regulated in post-IVIG treatment group compared with the pre-IVIG treatment group (Table 1).
The expressed genes that had a 1.5-fold increase are summarized in Table 4. The genes that showed decreased expressions are summarized in Table 5.
Six gene expressions (cytoskeleton associated protein 2, matrix metallopeptidase 3, matrix metallopeptidase 16, CD44 molecules, formyl peptide receptor receptor1 and angiotensinogen peptidase inhibitor) increased pre- and post-IVIG treatment in the KD patients (Table 4).
The genes that showed differential expressions by more than 1.5-fold (P<0.05) in at least one sample are shown in Fig. 1. The red color indicates an over expression while green indicates a down expression (Fig. 1). Through microarray analysis, the changes of gene expressions associated with proliferation, translation, inflammatory response, immune response, cell adhesion, cell migration, cell differentiation, apoptosis, cell growth, transport, cell cycle, transcription, signal transduction and metastasis were observed.
Several studies have suggested that immune activation and the secretion of cytokines contribute to the pathogenesis of KD. Although the etiology of KD remains unknown despite extensive investigations, the incidence of KD patients continues to increase in many countries [1,2,10,18].
There is no doubt that IVIG is a therapeutic utility in KD now. Infusion of high dose IVIG effectively reduces systemic inflammation and prevents coronary artery lesion in KD. Several mechanisms may explain the anti-inflammatory effects of IVIG in this disease [7, 8]. They include modification of the cytokine balance and alteration on both the differentiation and the function of monocyte/macrophages, neutrophils and lymphocytes [9,19,20]. However, the long term consequences of the cardiovascular sequelae in KD remain uncertain and therefore, KD is a leading cause of acquired heart disease in children [3,4,11].
To investigate the mechanisms underlying the therapeutic effects of IVIG, we examined gene expression profiles of fresh whole blood obtained in an acute stage before and in a subacute stage after IVIG therapy. The advantage of DNA microarrays is that it can evaluate changes in relative expression of thousands of genes simultaneously [15,21]. To gain further insight into the mechanism of KD related to immune processes and genetic factors, we investigated the difference of gene expression between KD patients and the control group. Also, we compared the difference of gene expression levels after IVIG therapy to identify potential candidate genes that might link the systemic immune response to the development of vasculitis and coronary artery disease by examining the gene expression patterns between acute and subacute stages in KD patients.
In the present study, many immunologic processes and genetic factors are attributed to the pathogenesis of KD. Immunologic abnormalities during the acute phase of KD reflect marked activation of the immune system leading to increased cytokine production. Chemokine ligand 5 (CCL5) and IL-5, which are related to the inflammatory response are over expressed compared to the control group in the acute state of KD. Also, over expressed CCL5, IL-32 protocadherin beta 16, angiopoietin like 3 are related to cell adhesion and migration. It has been reported that CCL5 is highly expressed in various tumors and stimulate tumor growth and metastasis by inducing tumor cell proliferation, angiogenesis, or the expression of MMPs [22,23]. We cannot find out a definite correlation between CCL5 and MMP but increased MMP genes, CCL5 and IL are potential candidates to understand this pathway.
After IVIG treatment, MMP related genes which belongs to MMP-2, MMP-28, MMP-25, MMP-15, are decreased. MMPs especially MMP-2, and 9, have been considered to play pathophysiologic roles in the development of coronary artery lesions [24,25]. Many studies that find out MMP-28 are over expressed in several disease states. MMP-28, stimulated by TNF, is a potential novel therapeutic target for prevention and treatment of metastasis of gastric cancer. MMP-28 is frequently over expressed during the progression of gastric cancer and contributes to tumor cell invasion and metastasis of tumor cells [26,27]. In this study, MMP-2 and 9 are insignificant compared to the control group. However, the expression of MMP-28 decreased after IVIG treatment.
NO is secreted by immune and vascular endothelial cells, NO has several roles such as regulating vascular tone and the maintenance of the integrity of the vasculature [28]. In the KD group, NOS 1, IL-12A, IL-17A, which are related to NO related genes, are over expressed before IVIG treatment. These genes are not down regulated after IVIG infusion so further studies are needed.
Caspase (apoptosis related cysteine peptidase, CASP) 1 and cytoskeleton associated protein 2 (CKAP2) are also over expressed in this study. Endothelial cell dysfunction and apoptosis are related to endothelial cell damage of the coronary artery. We can't detect direct change after IVIG infusion among over expressed genes. However, estrogen receptor 1, dimethylarginine dimethylaminohydrolase 2 (DDAH2) GTP cyclohydrolase I feedback regulator, endothelial NOS decreased in expression after IVIG compared to the control group. One study showed endothelial progenitor cell (EPC) participated in the process of arterial repair. The number of EPC increased significantly in the subacute phase of KD. Especially, the number of circulating EPC positively correlated with the level of NO and negatively correlated with the levels of TNF-α and CRP [29]. IVIG suppresses induced NOS expression of mononuclear leukocytes in patients with KD, thus decreasing NO-mediated inflammatory responses and coronary artery dilation [28,30].
Severe vasculitis leading to coronary artery lesions are noted in the refractory KD group resistant to IVIG [31]. We expect down regulation of the up regulated genes among inflammatory related genes after IVIG infusion but we didn't find different expression levels in this study. It seems this result is skewed because of small group size or IVIG resistant patients. TNF-α blockade (infliximab) has been reported to benefit KD patients with initial IVIG treatment failure. Further analysis of these genes in IVIG resistant group after infliximab is also attributed to understand pathogenesis of KD [31,32].
In conclusion, the data indicated that there are several genes which have different expressions in the KD group than in the control group. We also confirmed that expression levels change in several genes after an IVIG infusion. These genes are related to inflammatory response, immune response, cell adhesion, cell migration, cell differentiation, apoptosis, cell growth, transport, cell cycle, transcription, signal transduction, and metastasis. We found out a different expression pattern before and after IVIG treatment but there is lack of consistency in all KD patients. High dose IVIG is definitely the gold standard treatment. There is no doubt that IVIG has a therapeutic utility in treating KD now.
However, many studies estimate that 10-20% of patients do not respond to single dose IVIG, and the risk of aneurysms formation is higher in the unresponsive group than among patients who defervesce completely after a single dose of IVIG.
The limitation of our study is as follows. The sample size is small in number. Further analysis with larger samples of other independent set and specific sample such as peripheral blood T cell, monocytes/macrophages would be needed to find confirmative results in KD treatment.
Figures and Tables
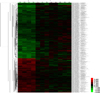 | Fig. 1Gene heat map in KD patients Pre- and Post-IVIG treatment. The color red indicates over expression while green indicates down expression. KD, Kawasaki disease; IVIG, intravenous immunoglobulin. |
Table 2
Up-regulated genes expressed by over 50 percent between the KD patients Pre-IVIG treatment and the control group

Table 3
Down-regulated genes expressed by over 50 percent between Pre-IVIG in KD patients and the control group

References
1. Park AH, Batchra N, Rowley A, Hotaling A. Patterns of Kawasaki syndrome presentation. Int J Pediatr Otorhinolaryngol. 1997. 40:41–50.
2. Shulman ST, Rowley AH. Etiology and pathogenesis of Kawasaki disease. Prog Pediatr Cardiol. 1997. 6:187–192.
3. Fulton DR, Newburger JW. Long term cardiac sequelae of Kawasaki disease. Curr Rheumatol Rep. 2000. 2:324–329.
4. Crystal MA, Manhiot C, Yeung RSM, Smallhorn JF, McCrindle BW. Coronary artery dilation after Kawasaki disease for children within the normal range. Int J Cardiol. 2009. 136:27–32.
5. Newburger JW. Takahashi M, Beiser AS, Burns JC, Bastian J, Chung KJ, et al. Single infusion of intravenous gamma globulin compared to four daily doses in the treatment of acute Kawasaki syndrome. N Engl J Med. 1991. 324:1633–1639.
6. Leung DY, Cortran RS, Kurt-Jones E, Burns JC, Newburger JW, Pober JS. Endothelial cell activation and high interleukin-1 secretion in the pathogenesis of acute Kawasaki disease. Lancet. 1989. 334:1298–1302.
7. Galeotti C, Bayry J, Kone-Paut I, Kaveri SV. Kawasaki disease: Aetiopathogenesis and therapeutic utility of intravenous immunoglobulin. Autoimmun Rev. 2010. 9:441–448.
8. Dajani AS, Taubert KA, Takahashi M, Bierman FZ, Freed MD, Ferrieri P, et al. Guidelines for long term management of patients with Kawasaki disease. Circulation. 1994. 89:916–922.
9. Matsuda A, Morita H, Unno H, Saito H, Matsumoto K, Hirao Y, et al. Anti-inflammatory effects of high-dose IgG on TNF-α-activated human coronary artery endothelial cells. Eur J Immunol. 2012. 42:2121–2131.
10. Rowley AH, Shulman ST. Pathogenesis and management of Kawasaki disease. Expert Rev Anti Infect Ther. 2010. 8:197–203.
11. Cheung YF, Huang GY, Chen SB, Liu XQ, Xi L, Liang XC, et al. Inflammatory gene polymorphisms and susceptibility to Kawasaki disease and its arterial sequelae. Pediatrics. 2008. 122:e608–e613.
12. Ikeda K, Ihara K, Yamaguchi K, Muneuchi J, Ohno T, Mizuno Y, et al. Genetic analysis of MMP gene polymorphisms in patients with Kawasaki disease. Pediatr Res. 2008. 63:182–185.
13. Wang CL, Wu YT, Liu CA, Kuo HC, Yang KD. Kawasaki disease infection, immunity and genetics. Pediatr Infect Dis J. 2005. 24:998–1004.
14. Shimizu C, Matsubara T, Onouchi Y, Jain S, Sun S, Nievergelt CM, et al. Matrix metalloproteinase haplotypes associated with coronary artery aneurysm formation in patients with Kawasaki disease. J Hum Genet. 2010. 55:779–784.
15. Abe J, Hibiki T, Noma S, Nakajima T, Saito H, Terai M. Gene expression profiling of the effect of high dose intravenous Ig in patients with Kawasaki disease. J Immunol. 2005. 174:5837–5845.
16. Nomura I, Abe J, Noma S, Saito H, Gao B, Wheeler G, et al. Adrenomedullin is highly expressed in blood monocytes associated with acute Kawasaki disease: A microarray gene expression study. Pediatr Res. 2005. 57:49–55.
17. Popper SJ, Shimizu C, Shike H, Kanegaye JT, Newburger JW, SundelRP , et al. Gene expression patterns reveal underlying biological processes in Kawasaki disease. Genome Biol. 2007. 8:R261.
18. Nakamura Y, Yashiro M, Uehara R, Sadakane A, Tsuboi S, Aoyama Y, et al. Epidemiologic features of Kawasaki disease in Japan: results of the 2009-2010 nationwide survey. J Epidemiol. 2012. 22:216–221.
19. Matsubara T, Ichiyma T, Furukawa S. Immunological profile of peripheral blood lymphocytes and monocytes/macrophages in Kawasaki disease. Clin Exp Immunol. 2005. 141:381–387.
20. Gupta M, Noel GJ, Schaefer M, Friedman D, Bussel J, Johann-Liang R. Cytokine modulation with immune gamma-globulin in peripheral blood of normal children and its implications in Kawasaki disease treatment. J Clin Immunol. 2001. 21:193–199.
21. Ikeda K, Yamaguchi K, Tanaka T, Mizuno Y, Hijikata A, Ohara O, et al. Unique activation status of peripheral blood mononuclear cells at acute phase of Kawasaki disease. Clin Exp Immunol. 2009. 160:246–255.
22. Chang LY, Lin YC, Mahalingam J, Huang CT, Chen TW, Kang CW, et al. Tumor derived chemokine CCL5 enhances TGF-β mediated killing of CD8+ T cells in colon cancer by T regulatory cells. Cancer Res. 2012. 72:1092–1102.
23. Mamtani M, Matsubara T, Shimizu C, Furukawa S, Akagi T, Onouchi Y, et al. Association of CCR2-CCR5 haplotypes and CCL3L1 copy number with Kawasaki disease, coronary artery lesions, and IVIG responses in Japanese Children. PLoS One. 2010. 5:e11458.
24. Furuno K, Takada H, Yamamoto K, Ikeda K, Ohno T, Khajoee V, et al. Tissue inhibitor of metalloproteinase 2 and coronary artery lesions in Kawasaki disease. J Pediatr. 2007. 151:155–160.
25. Lau AC, Duong TT, Ito S, Yeung RSM. Matrix metalloproteinase 9 activity leads to elastin breakdown in an animal model of Kawasaki disease. Arthritis Rheum. 2008. 58:854–863.
26. Jian P, Yanfang T, Zhuan Z, Jian W, Xueming Z, Jian Ni. MMP 28(epilysin) as a novel promoter of invasion and metastasis in gastric cancer. BMC Cancer. 2011. 11:200–208.
27. Overall CM, Kleifeld O. Towards third generation matrix metalloproteinase inhibitors for cancer therapy. Br J Cancer. 2006. 94:941–946.
28. Khajoee V, Kariyazono H, Ohno T, Ihara K, Mizuno Y, Kusuhara K, et al. Inducible and endothelial constitutive nitric oxide synthase gene polymorphisms in Kawasaki disease. Pediatr Int. 2003. 45:130–134.
29. Xu MG, Men LN, Zhao CY, Zhao X, Wang UX, Meng XC, et al. The number and function of circulating endothelial progenitor cells in patients with Kawasaki disease. Eur J Pediatr. 2010. 169:289–296.
30. Wang CL, Wu YT, Lee CJ, Liu HC, Huang LT, Yang KD. Decreased nitric oxide production after intravenous immunoglobulin treatment in patients with Kawasaki disease. J Pediatr. 2002. 141:560–565.
31. Blaisdell LL, Hayman JA, Moran AM. Infliximab treatment for pediatric refractory Kawasaki disease. Pediatr Cardiol. 2011. 32:1023–1027.
32. Kuo HC, Yang KD, Chang WC, Ger LP. Hsieh KS. Kawasaki disease: An update on diagnosis and treatment. Pediatr Neonatol. 2012. 53:4–11.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download