Abstract
Nephrotic syndrome is most commonly observed in membranous lupus nephritis in patients with systemic lupus erythematosus (SLE). However, other forms of idiopathic nephrotic syndrome rarely occur in these patients. Here, we report a case of SLE complicated by minimal change disease (MCD). A 24-year-old woman with SLE visited our hospital for generalized edema and heavy proteinuria. Laboratory tests did not support immunological exacerbation of lupus, while renal biopsy revealed diffusely effaced foot processes without electron-dense deposits that were consistent with MCD. Administration of high-dose corticosteroids and 6 subsequent cycles of monthly intravenous cyclophosphamide resulted in complete remission. Although nephrotic-range proteinuria recurred 1 month after switching to maintenance therapy with mycophenolate mofetil, complete remission was reestablished after a 6-month treatment with corticosteroids and cyclosporine. Physicians should be cautious in assessment and management of such a rare renal manifestation.
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease that can affect multiple organs. Approximately 60% of patients with SLE develop clinically evident kidney disease and renal involvement is a major cause of morbidity and mortality [1]. Undoubtedly, the pathological classification of lupus nephritis is of paramount importance for providing treatment choice and prognostic information. The classification system proposed by the International Society of Nephrology/Renal Pathology Society (ISN/RPS) has been widely used to date. Of note, one-third of patients with lupus nephritis experience nephrotic syndrome, which is most commonly seen in patients with membranous lupus nephritis (class V) followed by diffuse proliferative lupus nephritis (class IV) [2]. However, other forms of idiopathic nephrotic syndrome rarely occur in patients with SLE. Here, we report a case of SLE complicated by minimal change disease (MCD).
A 24-year-old woman visited our hospital because of generalized edema, weight gain of 9 kg over 2 months, and seizure. Nine months before her visit, she was diagnosed with SLE using the American College of Rheumatology criteria. At that time, the patient presented with a malar rash, arthralgia, and neurologic symptoms such as headache, cognitive dysfunction, and lower extremities weakness. In addition, positive tests for anti-nuclear antibodies (ANA) and anti-double stranded DNA (anti-dsDNA) antibodies were observed on serum analysis, but renal involvement was not evident. Complement 3 and 4 levels were 95 and 26 mg/dL. She had immediately received high-dose corticosteroids and 4 cycles of monthly intravenous cyclophosphamide, and subsequently, she had been switched to low-dose corticosteroids and azathioprine. At our hospital, the patient's laboratory tests showed a white blood cell count (WBC) of 8.14×103/µL (absolute neutrophil count of 3.76×103/µL, absolute lymphocyte count of 1.94×103/µL), hemoglobin of 8.5 g/dL, platelets of 513×103/µL, total cholesterol of 475 mg/dL, serum albumin of 1.0 g/dL, serum creatinine of 1.27 mg/dL (corresponding to an estimated glomerular filtration rate [eGFR] of 63 mL/min/1.73 m2, using the Chronic Kidney Disease Epidemiology Collaboration equation), serum glucose of 88 mg/dL, serum total calcium of 7.0 (corrected 9.4) mg/dL, serum sodium/potassium/chloride/total CO2 of 138/5.0/106/22 mmol/L, erythrocyte sedimentation rate of 13 mm/hr, serum C-reactive protein of 4.85 mg/L. The patient was tested positive for ANA (1 : 160, nucleolar pattern), but negative for anti-dsDNA, anti-Smith, anti-ribonucleoprotein, anti-Sjögren' s syndrome A/Ro, anti-Sjögren's syndrome B/La, lupus anticoagulant, and anti-cardiolipin antibodies. Serum complement levels were within normal limits. Urinalysis had revealed proteinuria (4+) and microhematuria. The spot urine protein-creatinine ratio (UPCR) was 36.4 g/g and 24-hr urinary protein excretion was 15.3 g/day with non-selective glomerular proteinuria on urine electrophoresis. Chest X-ray scan showed pleural effusion on both right and left sides, while a considerable amount of ascites was observed on abdominal ultrasound. Peritoneal fluid examination revealed WBC of 47/mm3 (mononuclear cells 98%), total protein of 100 mg/dL, and albumin not detected. There was leptomeningeal enhancement on brain magnetic resonance imaging (MRI) study, but cerebrospinal fluid analysis did not reveal a remarkable finding. To confirm renal involvement of lupus, a kidney biopsy was performed. Light microscopy showed 7 normal glomeruli without mesangial or endocapillary proliferation. No tubular or interstitial lesions were observed (Fig. 1A). Immunofluorescence study revealed weak staining of immunoglobulin G (IgG) along the capillary wall but no positive staining for IgA, IgM, C1q, or C3 on the glomeruli (Fig. 1B). On examination of 2 glomeruli by electron microscopy, no electron-dense deposits but diffuse effacement of the epithelial foot processes were observed (Fig. 1C). The patient was given a daily high-dose prednisolone of 1 mg/kg. After 6 weeks, however, her UPCR was 11.52 g/g and edema persisted. Considering the possibility of central nervous system involvement, monthly intravenous cyclophosphamide was administered. Six cycles of treatment resulted in complete remission with a UPCR of 0.11 g/g and a serum creatinine of 0.42 mg/dL. Mycophenolate mofetil (MMF) at a dose of 1.0 g/day was prescribed for maintenance therapy. One month later, however, the patient's UPCR increased to 4.45 g/g, but serological tests showed no evidence of increased lupus activity. A follow-up MRI study showed resolution of the leptomeningeal lesion. Daily oral prednisolone of 0.5 mg/kg in combination with cyclosporine of 5 mg/kg was administered, and complete remission was reestablished after 6 months. She has been maintained on low-dose oral prednisolone and cyclosporine without recurrence. The changes in kidney function and proteinuria during the disease course are shown in Fig. 2.
In general, 6 ISN/RPS classification categories can explain almost all kidney lesions involved in SLE. Although proliferative lupus nephritis such as class II, III, or IV comprise most cases, 10% to 15% of patients have pure membranous lupus nephritis characterized by heavy proteinuria and edema that is typically observed in idiopathic nephrotic syndrome [3]. Therefore, physicians usually suspect class V lupus nephritis when they encounter patients with SLE who present with such features. In this report, we describe an unusual renal manifestation of SLE. In our case, the patient presented with typical clinical symptoms and signs of nephrotic syndrome and showed normal appearing glomeruli on light microscopy and immunofluorescence study; however, diffuse foot process effacement without electron-dense deposits were observed on electron microscopy, all of which are compatible with MCD. Such pathologic findings are in sharp contrast with those of class V lesions, which show subepithelial immune deposits along with occasional mesangial or subendothelial immune deposits. Nevertheless, membranous lupus nephritis can develop without other clinical or serological manifestations of lupus; therefore, it is difficult to diagnose MCD without performing a kidney biopsy.
Because MCD is rare in patients with lupus, its clinical outcome and responsiveness to immunosuppressive treatments have not yet been clearly defined. A literature review identified several reports of idiopathic nephrotic syndrome in association with SLE to date [2,4-9]. Most cases involved MCD or focal segmental glomerulosclerosis. According to these reports, MCD can occur at the initial diagnosis, during flare, or several years after the onset of SLE even without any apparent lupus activity [9]. Although a few cases had been treated with MMF [8] or rituximab [2], most of them responded well to corticosteroids, and all cases eventually achieved complete remission. In our case, MCD occurred 9 months after the diagnosis of SLE, and the patient has since been on maintenance treatment with low-dose corticosteroids and azathioprine. The patient experienced a relapse but attained complete remission after the prolonged use of immunosuppressive drugs. These findings taken together suggest that responsiveness to corticosteroids or other immunosuppressants is favorable in these patients; therefore, they presumably have good prognosis.
Similar to many cases of class V lupus nephritis, immunological exacerbation at the time of MCD was not clearly evident on laboratory tests. However, it is uncertain whether lupus activity was indeed low because the patient developed seizure, which corresponds to a high score of SLE disease activity index. Interestingly, it is reported that such a neuropsychiatric manifestation can also occur in a stable state without immunological exacerbation of lupus [10]. Nevertheless, in this patient, proteinuria and hematuria, which are also included in the SLE disease activity index, were revealed to be not relevant to typical lupus nephritis.
The pathogenic association between SLE and MCD remains under debate because MCD is considered a non-inflammatory process, while lupus nephritis is an inflammatory process. Several reports can explain a possible link between the 2 diseases. SLE is an autoimmune disease characterized by autoantibody overproduction, complement activation, and immune complex deposition. In contrast, the pathogenesis of MCD has not yet been clearly defined, although glomerular permeability factor has been reported to be the main culprit in the pathogenesis. Interestingly, altered T-lymphocyte function is present in both disorders. In fact, the helper/suppressor T-cell ratio is well correlated with a number of clinical parameters of patients with SLE [11]. In addition, an increased number of helper T cells and lack of T-cell suppression can lead to polyclonal B-cell activation and excess antibody production in these patients [12]. Furthermore, regulatory T-cell (Treg)-associated cytokines are involved in the development of SLE [13]. Of note, in MCD, glomerular permeability factor is reported to be derived from T-cells, which can cause filtration barrier alterations and ultimately result in heavy proteinuria [14]. Some authors also suggested the presence of dysregulated helper/suppressor T-cells and unbalanced Th17/Treg cells in adult patients with MCD [15,16]. Such T-cell abnormalities may comprise a pathophysiological link between MCD and SLE. Nevertheless, this cannot fully explain the pathogenesis of MCD in SLE because the immune complex deposits that play a major role in SLE were not seen in our case.
In summary, here we reported a rare case of SLE complicated by MCD. In this case, it can be clinically confused with membranous lupus nephritis, thus physicians should be cautious in assessment and management of such a rare renal manifestation. Although T-cell dysfunction might create an immunological association between the 2 diseases, further studies are required to clarify whether this is another manifestation of lupus nephritis.
Figures and Tables
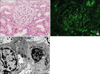 | Fig. 1Pathologic findings. (A) Renal biopsy specimen shows normal glomeruli on light microscopy (original magnification, ×400). (B) Direct immunofluorescence shows weakly positive staining of IgG along the capillary wall. (C) Electron microscopy shows no electron-dense deposits within the glomeruli. Diffusely effaced foot processes are noted. |
References
1. Cameron JS. Systemic lupus erythematosus. In : Neilson EG, Couser WG, editors. Immunologic renal diseases. 2nd ed. Philadelphia: Lippincott Williams & Wilkins;2001. p. 1057–1104.
2. Shahane A, Khasnis A, Gota C. Rituximab-responsive nephrotic syndrome due to minimal change disease in systemic lupus erythematosus. J Clin Rheumatol. 2012; 18:199–202.
3. Lupus nephritis: prognostic factors and probability of maintaining life-supporting renal function 10 years after the diagnosis: Gruppo Italiano per lo Studio della Nefrite Lupica (GISNEL). Am J Kidney Dis. 1992; 19:473–479.
4. Perakis C, Arvanitis A, Sotsiou F, Emmanouel DS. Nephrotic syndrome caused by minimal-change disease in a patient with focal proliferative SLE nephritis (WHO III) in remission. Nephrol Dial Transplant. 1998; 13:467–470.
5. Hertig A, Droz D, Lesavre P, Grunfeld JP, Rieu P. SLE and idiopathic nephrotic syndrome: coincidence or not? Am J Kidney Dis. 2002; 40:1179–1184.
6. Seo DB, Lee SW, Song JH, Lee KJ, Han JY, Kim MJ. Acute renal failure associated with a minimal change nephrotic syndrome in a systemic lupus erythematosus patient. Yonsei Med J. 2002; 43:114–118.
7. Horino T, Takao T, Morita T, Ito H, Hashimoto K. Minimal change nephrotic syndrome associated with systemic lupus erythematosus. Nephrol Dial Transplant. 2006; 21:230.
8. Hong YH, Yun DY, Jung YW, Oh MJ, Kim HJ, Lee CK. A case of minimal change disease treated successfully with mycophenolate mofetil in a patient with systemic lupus erythematosus. Korean J Intern Med. 2011; 26:470–473.
9. Redondo-Pachon MD, Enriquez R, Sirvent AE, Andrada E, Millan I, Amoros F. Minimal-change nephropathy in systemic lupus erythematosus. Nefrologia. 2012; 32:412–413.
10. Zhou HQ, Zhang FC, Tian XP, Leng XM, Lu JJ, Zhao Y, et al. Clinical features and outcome of neuropsychiatric lupus in Chinese: analysis of 240 hospitalized patients. Lupus. 2008; 17:93–99.
11. Smolen JS, Chused TM, Leiserson WM, Reeves JP, Alling D, Steinberg AD. Heterogeneity of immunoregulatory T-cell subsets in systemic lupus erythematosus. Correlation with clinical features. Am J Med. 1982; 72:783–790.
12. Morimoto C, Steinberg AD, Letvin NL, Hagan M, Takeuchi T, Daley J, et al. A defect of immunoregulatory T cell subsets in systemic lupus erythematosus patients demonstrated with anti-2H4 antibody. J Clin Invest. 1987; 79:762–768.
13. Okamoto A, Fujio K, Okamura T, Yamamoto K. Regulatory T-cell-associated cytokines in systemic lupus erythematosus. J Biomed Biotechnol. 2011; 2011:463412.
14. Koyama A, Fujisaki M, Kobayashi M, Igarashi M, Narita M. A glomerular permeability factor produced by human T cell hybridomas. Kidney Int. 1991; 40:453–460.
15. Ozaki T. Two-color analysis of lymphocyte subpopulations in membranous nephropathy and minimal change nephrotic syndrome. Nihon Jinzo Gakkai Shi. 1989; 31:797–806.
16. Fiser RT, Arnold WC, Charlton RK, Steele RW, Childress SH, Shirkey B. T-lymphocyte subsets in nephrotic syndrome. Kidney Int. 1991; 40:913–916.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download