Abstract
Background and Objectives
Off-thyroxine serum thyroglobulin (Tg) level is important to predict metastatic disease (MD) in papillary thyroid cancer (PTC); however, it is unclear whether a single off-thyroxine Tg level is sufficient for predicting MD. In this study, we determined whether serial measurement of off-thyroxine serum Tg level can predict metastasis in PTC patients after total thyroidectomy.
Materials and Methods
We enrolled 140 PTC patients in whom serum thyroid-stimulating hormone (TSH) and Tg levels were measured 7 days before radio-iodine (RAI) treatment (TSHA and TgA) and on the day of RAI treatment (TSHB and TgB) with withholding L-thyroxine for 4 weeks before RAI treatment. The values of TSHinc (TSHB-TSHA) and Tginc (TgB-TgA), Tgratio (TgB/TgA), Tginc/TSHinc and Tgratio/TSHinc were calculated. Tginc/TSHinc and Tgratio/TSHinc were tested if those parameters can predict MD in patients with TSHA>30 µIU/mL and TgA<10 ng/mL.
Results
Forty-four patients had MD and 96 had no evidence of MD (non-MD). MD group showed higher levels of TgA, TgB, Tginc, Tginc/TSHinc and Tgratio/TSHinc compared with non-MD group. A significant correlation was found between TSHinc and Tgratio (r=0.669) in MD group. In 43 patients with TSHA>30 µIU/mL and TgA<10 ng/mL (MD, 9; non-MD, 34), both Tginc/TSHinc (100%) and Tgratio/TSHinc (89%) had higher sensitivities for predicting MD than TgB (78%).
Thyroglobulin (Tg), a large glycoprotein stored in the follicular colloid of the thyroid gland, is produced only by the normal thyroid gland or well-differentiated thyroid cancer (DTC) cells, including papillary thyroid cancer (PTC) cells.1,2) Serum Tg is a very specific tumor marker for patients with DTC and shows high specificity for detecting DTC recurrence.3-5) Although DTC patients who were considered clinically free of tumor had serum Tg levels of <1 ng/mL with a suppressed thyroid-stimulating hormone (TSH), 21% of these patients had serum Tg levels of >2 ng/mL after recombinant human TSH (rhTSH) stimulation.6) Moreover, one-third of this patient population had metastatic DTC lesions.6) Hence, it is important to measure the serum Tg level after appropriate serum TSH elevation, the level of which is yet to be determined, however, commonly accepted as a TSH level of more than 30 µIU/mL after thyroid hormone withdrawal (off-thyroxine) or rhTSH stimulation in order to effectively detect DTC recurrence. The American Thyroid Association guidelines suggest that further investigation for DTC recurrence is required when the serum Tg level is >2 ng/mL following serum TSH elevation.7) Further, empirical radio-iodine (RAI) treatment should be considered in patients with Tg levels of >5 ng/mL after rhTSH stimulation or >10 ng/mL after thyroid hormone withdrawal, even if the results of diagnostic RAI scan and neck ultrasonography are negative.8,9)
However, it is unknown whether a serum TSH level of 30 µIU/mL is adequate for measuring the off-thyroxine Tg level and whether a single off-thyroxine serum Tg level is sufficient for determining further RAI treatment. In this study, we evaluated the relationship between the increments of serum TSH and Tg levels in PTC patients whose off-thyroxine serum TSH and Tg levels were measured serially, and, then, evaluated the clinical utility of serial measurements of off-thyroxine serum TSH and Tg levels for predicting PTC metastasis as compared with single measurement of off-thyroxine Tg level.
This retrospective study was approved by the Institutional Review Board in our medical center (IRB No. 2012-02-002). A total of 140 patients with PTC in whom serial measurements of off-thyroxine serum TSH and Tg levels were obtained before high-dose RAI treatment were found in electric medical data base of our medical center and enrolled in this study. Of 140 enrolled patients, 100 patients underwent RAI treatment for ablation after total thyroidectomy, 25 patients for treating recurrent or metastatic lesions, and 15 patients for adjuvant treatment after surgical resection of neck recurrence. All patients had undergone a total thyroidectomy before RAI treatment. The first measurement of off-thyroxine serum TSH (TSHA) and Tg (TgA) levels was performed 7±1 days (median 7 days; range, 5-14 days) before the RAI treatment and the second measurement of serum TSH (TSHB) and Tg (TgB) levels was performed on the day of RAI treatment before the administration of RAI. Patients with a high serum anti-Tg antibody level (>25 IU/mL) and patients with a history of a malignant tumor other than PTC were excluded from this study. The therapeutic RAI dose ranged from 3.7 GBq (100 mCi) to 7.4 GBq (200 mCi). The replacement of L-thyroxine (T4) was discontinued for 4 weeks before RAI treatment, and patients received replacement L-triiodothyronine (T3) up to 2 weeks before the RAI treatment. In addition, all patients received a low-iodine diet for at least 2 weeks before the RAI treatment.
After RAI treatment, all patients underwent clinical follow-up that included blood tests and diagnostic imaging studies. Each patient was clinically assessed and classified as a patient with local and/or systemic metastatic disease (MD) or a patient with only thyroid remnant or no evidence of disease (non-MD). The inclusion criteria for MD group are 1) distant metastatic lesions which showed positive uptake on RAI scan, 2) positive neck lymph node uptake on RAI scan which were also found on other imaging modalities such as ultrasonography or 18F-fluorodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT), and 3) recurred or metastatic lesions which were histopathologically confirmed. Non-MD group was defined as no evidence of recurrence or metastatic lesion on at least a year clinical follow-up after RAI treatment. Furthermore, patients who showed only high serum Tg level with negative finding on imaging studies were excluded from the study.
Serum TSH, Tg and anti-Tg antibody were measured using an electrochemiluminescence immunoassay (ECLIA) on the Roche Modular Analytics E170 system (Roche Diagnostics, Indianapolis, IN, USA). Interassay coefficients of variation (CV) were <2.5% for TSH (1.61 and 9.64 µIU/mL), <3.0% for Tg (25.57 and 78.19 ng/mL) and <5.4% for anti-Tg (81.80 and 216.17 IU/mL).
The increase of serum TSH (TSHinc=TSHB-TSHA) and Tg (Tginc=TgB-TgA), and the Tg increment ratio (Tgratio=TgB/TgA) were calculated in all patients. Because the TSH level influences serum Tg release, TSH can be a corrected factor for Tg production;10) hence, Tginc and Tgratio values corrected for TSHinc (Tginc/TSHinc and Tgratio/TSHinc, respectively) were also calculated. However, the values of Tginc/TSHinc and Tgratio/TSHinc were not calculated in patients with TSHA>100 µIU/mL, because the upper limit of our serum TSH assay is 100 µIU/mL. Moreover, the values of Tginc/TSHinc and Tgratio/TSHinc were also not calculated for the statistical analysis in patients with negative TSHinc (TSHA>TSHB) and in patients with TSHinc less than CV value of serum TSH (2.5%) whose TSH level could be considered unchanged between TSHA and TSHB. The values of TSHA, TgA, TSHB, TgB, TSHinc, Tginc, Tgratio, Tginc/TSHinc, and Tgratio/TSHinc were compared between the MD group and the non-MD group using the Mann-Whitney test. Furthermore, Spearman's correlation coefficients were calculated for Tginc and Tgratio with regard to TSHinc.
Afterwards, we performed subgroup analysis. First, a subgroup of patients with TSHA>30 µIU/mL was selected. The ratios of patients with MD according to the value of TgA and TgB were calculated in order to evaluate whether the value of TgA is sufficient to detect MD in patients with TSHA>30 µIU/mL.
Second, the clinical role of Tginc/TSHinc and Tgratio/TSHinc in patients who showed an increase of off-thyroxine serum TSH level during serial measurement was evaluated. We selected another subgroup of patients with TSHA>30 µIU/mL and TgA<10 ng/mL who showed sufficient serum levels of TSH but insufficient serum levels of Tg for empirical RAI treatment 7 days before RAI treatment.8) Patients with TSHA levels greater than 100 µIU/mL were excluded from this subgroup analysis because their serum TSHA levels had already reached the upper limit of our serum TSH assay and we could not evaluate their Tginc/TSHinc and Tgratio/TSHinc values. Furthermore, patients with TSHinc less than CV value of serum TSH (2.5%) were also excluded from the statistical analysis. In the subgroup patients, the values of TSHA, TgA, TSHB, TgB, TSHinc, Tginc, Tgratio, Tginc/TSHinc and Tgratio/TSHinc were compared between the MD group and the non-MD group by using the Mann-Whitney test, and receiver-operating characteristic (ROC) curves were drawn for Tginc/TSHinc and Tgratio/TSHinc to determine the cut-off value to differentiate the MD group from the non-MD group. SPSS software for Windows (SPSS Inc., Chicago, IL, USA) was used for all statistical tests and p values less than 0.05 were considered statistically significant.
Of 140 enrolled patients, 44 patients (31%) were classified into the MD group, and the remaining 96 patients (69%) were classified into the non-MD group. The characteristics of all patients, MD group, and non-MD group are shown in Table 1. Of 44 patients in the MD group, 37 were diagnosed with neck lymph node metastases; 4, with pulmonary metastases; 2, with mediastinal lymph node metastases; and 1, with lung, mediastinal, and neck lymph node metastases. Of 140 patients, 83 patients (59%) had a serum TSHA level greater than 30 µIU/mL and all patients had a serum TSHB level greater than 30 µIU/mL. During the last 7 days before RAI treatment, the average serum TSH level of all patients had increased to over 40 µIU/mL. Furthermore, 21 patients (15%) had already reached serum TSHA levels greater than 100 µIU/mL and 6 patients (4%) had negative TSHinc or TSHinc levels less than CV value of serum TSH (2.5%). The values of Tginc/TSHinc and Tgratio/TSHinc were calculated in the remaining 113 patients except those 27 patients.
The comparison between the MD and the non-MD groups revealed that the values of TgA, TgB, Tginc, Tginc/TSHinc and Tgratio/TSHinc were significantly higher in the MD group than in the non-MD group (Table 1; p<0.05 each). The Tgratio value tended to be higher in the MD group than in the non-MD group, but the significance was marginal (p=0.05).
There were significant correlation between TSHinc and Tgratio in both MD and non-MD patients. In MD group patients, a significant moderate correlation was found between TSHinc and Tgratio (p<0.0001; Spearman's correlation coefficient r=0.669; 95% confidence interval 0.465-0.806). Linear regression of Tgratio (YA) versus TSHinc (XA) in MD group yielded the equation YA=0.106XA+0.425 (Fig. 1A). However, no significant correlation was found between TSHinc and Tginc in this group (p=0.2; r=0.197). In non-MD group patients, a weak but significant correlation was found between TSHinc and Tgratio (p<0.0001; r=0.419; 95% confidence interval 0.239-0.572) (Fig. 1B). Linear regression of Tgratio (YB) versus TSHinc (XB) in non-MD group yielded the equation YB=0.063XB+1.432. Similar to MD group patients, there was no significant correlation between TSHinc and Tginc in this group (p=0.4; r=0.216).
In a subgroup of 83 patients with TSHA>30 µIU/mL, the ratios of patients with MD according to the value of TgA and TgB were evaluated (Table 2). Of these 83 patients, 27 patients were MD group and the remaining 56 patients were non-MD group. As shown in Table 2, 41% (11 out of 27 patients) of patients with MD showed TgA<10 ng/mL, although the level of TSHA in those patients were more than 30 µIU/mL which is commonly accepted as a appropriate serum TSH level to evaluate off-thyroxine Tg level. Furthermore, in 11 patients with MD who showed TgA<10 ng/mL, 7 patients (64%) showed TgB>10 ng/mL.
We also evaluated the clinical role Tginc/TSHinc and Tgratio/TSHinc in a subgroup of 43 patients with TSHA>30 µIU/mL and TgA<10 ng/mL who seemed to have insufficient serum levels of TgA for empirical RAI treatment. Of the 43 subgroup patients, 9 patients were from the MD group and the remaining 34 patients were from the non-MD group. All 9 patients in MD group were diagnosed with neck lymph node metastases at follow-up (as in Fig. 2). The data of MD and non-MD subgroup patients are summarized in Table 3. Furthermore, the characteristics of 9 patients with MD in subgroup patients are shown in Table 4. The values of TgA, TgB, Tginc, Tgratio, Tginc/TSHinc and Tgratio/TSHinc were significantly higher for MD patients than for non-MD patients (p<0.05 each). Of the 9 patients from the MD subgroup, 7 patients had TgB>10 ng/mL whereas only 3 out of 34 non-MD patients had TgB>10 ng/mL.
Tginc/TSHinc and Tgratio/TSHinc showed high diagnostic performance in these subgroup patients for predicting MD. Because Tg stimulation is related to the intensity of TSH receptor activation,11,12) and the serum TSH levels varied among the subgroup patients, only Tginc/TSHinc and Tgratio/TSHinc were used as parameters for ROC curve analysis in detecting patients with MD. The area under the ROC curve (AUC) for Tginc/TSHinc was 0.944 (95% confidence interval 0.829-0.991; Fig. 3A), and the AUC for Tgratio/TSHinc was 0.794 (95% confidence interval 0.643-0.902; Fig. 3B). The sensitivities of Tginc/TSHinc (100%) and Tgratio/TSHinc (89%) for predicting patients with MD were higher than that of TgB (78%; Table 5).
In this study, we retrospectively examined the increments of serum TSH and Tg in PTC patients by serial measurements of serum TSH and Tg levels 7 days before RAI treatment and on the day of RAI treatment. Patients with MD showed significantly higher levels of Tginc, and TSHinc-corrected Tginc and Tgratio than non-MD patients. Also, the increment in serum Tg was significantly correlated with the increment in serum TSH in MD patients. Furthermore, we could predict MD patients with high sensitivity by using Tginc/TSHinc and Tgratio/TSHinc in patients with TSH>30 µIU/mL and Tg<10 ng/mL 7 day before RAI treatment.
The serum Tg level in DTC patients is related to tumor burden and is influenced by the serum TSH level. A previous study showed that the serum Tg/TSH level after thyroid hormone withdrawal was significantly linked with the total volume of metastatic lymph nodes of DTC.13) Furthermore, Robbins et al.11) showed that the basal Tg level was correlated with the number of metastatic lesions and patients with bone or lung metastases showed higher Tg levels than those with cervical metastases. However, they also revealed that the fold increase in the serum Tg after rhTSH stimulation was not influenced by tumor volume or the site of metastatic lesions. In addition to the mass of metastatic lesions, serum Tg value was partially accounted for by serum TSH level.14) Furthermore, the magnitude and chronicity of TSH stimulation and TSH receptor status also influence the magnitude of the serum Tg response to TSH stimulation.12) The results of our study also showed a moderate correlation between the increments of TSH and Tg in MD patients, supporting the idea that serum Tg increment is influenced by the serum TSH level as well as by the mass of the metastatic lesion. A previous study by Spencer, et al. showed similar increase in the Tg level (10- to 20-fold increases) in both normal thyroid remnants and DTC lesions.12) In contrast, the results of our study showed that the Tg increment was higher and more strongly correlated with increment of the TSH increment in MD patients than in non-MD patients. This difference may be because the patients in our study had higher levels of serum TSH (1-100 µIU/mL) than the patients in the study from Spencer, et al. (0.01-1 µIU/mL).12) Therefore, the response to TSH stimulation in tumors and thyroid remnants may be different when the serum level of TSH is extremely high.
Although empirical RAI treatment in patients with a high Tg level but a negative RAI scan is still somewhat controversial,8,9,15-17) empirical RAI treatment is routinely considered in patients with a off-thyroxine serum Tg level >10 ng/mL.8,9,15) A serum Tg level with serum TSH level of more than 30 µIU/mL is commonly accepted as off-thyroxine serum Tg level. However, 41% of patients with MD in our study showed TgA<10 ng/mL, even though the level of TSHA was more than 30 µIU/mL. Moreover, 64% of these patients showed Tg>10 ng/mL after further increase of serum TSH level, suggesting that serum TSH level of 30 µIU/mL might be inadequate to evaluate off-thyroxine Tg level and further increase of serum TSH level with longer period of TSH stimulation could be needed in some patients with MD.
Because the increment of serum Tg in MD patients was more strongly correlated with the increment of serum TSH with higher value of the slope in linear regression than in non-MD patients in our study, differences in the serum Tg level or the serum Tg increment might become evident after further elevation of serum TSH. Furthermore, a previous study reported that because of the variability of serum TSH levels, repeated measurement of serum Tg level after rhTSH stimulation might be needed in DTC patients with persistent disease and low Tg levels.14) Based on these results, we hypothesized that we could predict more patients with MD by using Tginc/TSHinc and Tgratio/TSHinc than by using a single off-thyroxine Tg measurement. Hence, we selected patients who showed TSHA>30 µIU/mL and TgA<10 ng/mL to evaluate the further role of Tginc/TSHinc and Tgratio/TSHinc for predicting MD. These patients showed a sufficient serum TSH elevation but insufficient off-thyroxine Tg levels for consideration for RAI treatment. According to the results of our study, Tginc/TSHinc and Tgratio/TSHinc had high sensitivities for detecting MD patients among the selected patients. This suggests that further increase of serum TSH level could be needed to exclude the possibility of presence of metastatic lesion even in patients with a low off-thyroxine Tg level, and the increment in the TSHinc-corrected Tg level could be used as a diagnostic tool in those patients.
Moreover, the results of our study showed that Tginc/TSHinc and Tgratio/TSHinc had higher sensitivity than TgB for detecting MD patients in the subgroup patients. Previous studies have already demonstrated that a significant number of DTC patients with metastatic lesions were serum Tg and anti-Tg antibody negative, suggesting that off-thyroxine serum Tg level alone cannot be used to determine the RAI treatment.18,19) The results of subgroup analysis in our study also revealed that 2 of 9 patients with MD had off-thyroxine serum Tg level of less than 10 ng/mL. The sensitivity of off-thyroxine serum Tg level (TgB) for predicting patients with MD was 78%, which was lower than that of Tginc/TSHinc (100%) and Tgratio/TSHinc (89%). However, because our study was a retrospective study with a small number of patients, a further prospective study with more patients will be necessary to elucidate the role of Tginc/TSHinc and Tgratio/TSHinc.
In this study, we enrolled only patients with PTC and excluded other histological types of DTC such as follicular thyroid cancer (FTC) and Hürthle cell carcinoma. Over 90% of thyroid cancers in Korea are PTC, whereas FTC is relatively rare subtype, less than 2%.20) Furthermore, a previous study showed that the median fold increase in Tg after TSH stimulation was higher in PTC than FTC, suggesting that the histological type of DTC influences the Tg increment after TSH stimulation.11) Although a small number of patients in our medical center had FTC, we excluded patients with FTC to avoid the effect of histological type.
Our study had several limitations. First, the study was retrospectively performed and the enrolled patients with MD had clinically evident metastatic lesions. Therefore, the use of serial measurement of off-thyroxine serum TSH and Tg levels in the clinical management of patients with high off-thyroxine Tg levels but negative diagnostic image tests should be evaluated in further studies. Second, because the upper limit of our serum TSH assay is 100 µIU/mL, all serum TSH levels of more than 100 µIU/mL were reported as 100 µIU/mL, which could affect the value of Tginc/TSHinc and Tgratio/TSHinc. Third, the first measurement of off-thyroxine serum TSH and Tg levels was obtained over a wide range of days for different patients (5-14 days). Finally, the MD group in our study had metastatic lesions on various organs (lung, neck, and mediastinum). The site of the metastatic lesion might affect the increment in serum Tg, although a previous study showed that the location of metastatic lesions did not influence Tg responsiveness to rhTSH.11)
In summary, PTC patients with metastatic lesions showed a high level of the serum Tg increment on serial measurements obtained after thyroid hormone withdrawal. A significant correlation was observed between the increment in serum TSH and Tg in patients with metastases. Furthermore, the increment in serum Tg corrected for the increment in serum TSH had higher sensitivity than a single off-thyroxine serum Tg level for predicting patients with metastases who had serum TSH>30 µIU/mL and serum Tg<10 ng/mL 7 days before RAI treatment. Serial measurement of off-thyroxine serum TSH and Tg levels can be helpful for predicting metastases in PTC patients.
Figures and Tables
Fig. 1
Scatter plot showing the correlation between TSHinc and Tgratio in the MD group (A) and the non-MD group (B). A moderate correlation was observed in the MD group (A, p<0.001; r=0.669) and a weak correlation was observed in the non-MD group (B, p<0.0001; r=0.419).

Fig. 2
A 41-year-old female with PTC underwent 5.6 GBq radio-iodine (RAI) treatment after surgical resection of neck lymph node recurrence. TSHA, TgA, TSHB, and TgB levels were 34.7 µIU/mL, 5.4 ng/mL, 100 µIU/mL, and 25.4 ng/mL, respectively. The Tginc/TSHinc and Tgratio/TSHinc were 0.31 and 0.07, respectively, which were higher than our cut-off values. A post-therapeutic RAI scan (A) did not show abnormal RAI uptake; however, FDG PET/CT (B) performed on the day of RAI treatment showed focal intense 18F-FDG uptake in the left neck area (arrows), which was histopathologically confirmed as metastatic PTC.
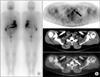
Fig. 3
ROC curves of Tginc/TSHinc (A) and Tgratio/TSHinc (B) for detecting patients with MD in a subgroup of patients with TSHA>30 µIU/mL and TgA<10 ng/mL.

Table 2
The distribution of subgroup patients with TSHA>30 µIU/mL (n=83 MD group=27) according to serum TgA and TgB levels

Table 3
The values of TSHA, TgA, TSHB, TgB, Tginc, Tgratio, Tginc/TSHinc and Tgratio/TSHinc for MD and non-MD groups in subgroup patients with TSHA>30 µIU/mL and TgA<10 ng/mL (n=43)

References
1. Iervasi A, Iervasi G, Carpi A, Zucchelli GC. Serum thyroglobulin measurement: clinical background and main methodological aspects with clinical impact. Biomed Pharmacother. 2006. 60(8):414–424.

2. Francis Z, Schlumberger M. Serum thyroglobulin determination in thyroid cancer patients. Best Pract Res Clin Endocrinol Metab. 2008. 22(6):1039–1046.

3. Van Herle AJ, Uller RP, Matthews NI, Brown J. Radioimmunoassay for measurement of thyroglobulin in human serum. J Clin Invest. 1973. 52(6):1320–1327.

4. Torrens JI, Burch HB. Serum thyroglobulin measurement. Utility in clinical practice. Endocrinol Metab Clin North Am. 2001. 30(2):429–467.
5. Eustatia-Rutten CF, Smit JW, Romijn JA, van der Kleij-Corssmit EP, Pereira AM, Stokkel MP, et al. Diagnostic value of serum thyroglobulin measurements in the follow-up of differentiated thyroid carcinoma, a structured meta-analysis. Clin Endocrinol (Oxf). 2004. 61(1):61–74.

6. Mazzaferri EL, Robbins RJ, Spencer CA, Braverman LE, Pacini F, Wartofsky L, et al. A consensus report of the role of serum thyroglobulin as a monitoring method for low-risk patients with papillary thyroid carcinoma. J Clin Endocrinol Metab. 2003. 88(4):1433–1441.

7. Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, et al. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009. 19(11):1167–1214.

8. Mazzaferri EL. Empirically treating high serum thyroglobulin levels. J Nucl Med. 2005. 46(7):1079–1088.
9. Silberstein EB. The problem of the patient with thyroglobulin elevation but negative iodine scintigraphy: the TENIS syndrome. Semin Nucl Med. 2011. 41(2):113–120.

10. Lin Y, Li T, Liang J, Li X, Qiu L, Wang S, et al. Predictive value of preablation stimulated thyroglobulin and thyroglobulin/thyroid-stimulating hormone ratio in differentiated thyroid cancer. Clin Nucl Med. 2011. 36(12):1102–1105.

11. Robbins RJ, Srivastava S, Shaha A, Ghossein R, Larson SM, Fleisher M, et al. Factors influencing the basal and recombinant human thyrotropin-stimulated serum thyroglobulin in patients with metastatic thyroid carcinoma. J Clin Endocrinol Metab. 2004. 89(12):6010–6016.

12. Spencer CA, LoPresti JS, Fatemi S, Nicoloff JT. Detection of residual and recurrent differentiated thyroid carcinoma by serum thyroglobulin measurement. Thyroid. 1999. 9(5):435–441.

13. Bachelot A, Cailleux AF, Klain M, Baudin E, Ricard M, Bellon N, et al. Relationship between tumor burden and serum thyroglobulin level in patients with papillary and follicular thyroid carcinoma. Thyroid. 2002. 12(8):707–711.

14. Pellegriti G, Scollo C, Regalbuto C, Attard M, Marozzi P, Vermiglio F, et al. The diagnostic use of the rhTSH/thyroglobulin test in differentiated thyroid cancer patients with persistent disease and low thyroglobulin levels. Clin Endocrinol (Oxf). 2003. 58(5):556–561.

15. Ma C, Xie J, Kuang A. Is empiric 131I therapy justified for patients with positive thyroglobulin and negative 131I whole-body scanning results? J Nucl Med. 2005. 46(7):1164–1170.
16. Kim WG, Ryu JS, Kim EY, Lee JH, Baek JH, Yoon JH, et al. Empiric high-dose 131-iodine therapy lacks efficacy for treated papillary thyroid cancer patients with detectable serum thyroglobulin, but negative cervical sonography and 18F-fluorodeoxyglucose positron emission tomography scan. J Clin Endocrinol Metab. 2010. 95(3):1169–1173.

17. Pacini F, Agate L, Elisei R, Capezzone M, Ceccarelli C, Lippi F, et al. Outcome of differentiated thyroid cancer with detectable serum Tg and negative diagnostic (131)I whole body scan: comparison of patients treated with high (131)I activities versus untreated patients. J Clin Endocrinol Metab. 2001. 86(9):4092–4097.

18. Robbins RJ, Chon JT, Fleisher M, Larson SM, Tuttle RM. Is the serum thyroglobulin response to recombinant human thyrotropin sufficient, by itself, to monitor for residual thyroid carcinoma? J Clin Endocrinol Metab. 2002. 87(7):3242–3247.

19. Park EK, Chung JK, Lim IH, Park do J, Lee DS, Lee MC, et al. Recurrent/metastatic thyroid carcinomas false negative for serum thyroglobulin but positive by posttherapy I-131 whole body scans. Eur J Nucl Med Mol Imaging. 2009. 36(2):172–179.

20. Ministry of Health and Welfare. The Korea central cancer registry. Annual report of cancer statistics in Korea in 2008. 2010. Seoul, Korea: Ministry of Health and Welfare.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download