Abstract
Background:
Iguratimod is a new type of disease modifying anti-rheumatic drug, which reduced the production of inflammatory cytokines. The purpose of this study was to evaluate pharmacokinetic characteristics and safety profiles of iguratimod after a single oral administration in healthy Korean volunteers.
Methods:
A randomized, double-blind, placebo-controlled, parallel group, single oral dose study was conducted in 24 healthy male volunteers. Three groups of eight subjects each received 25 mg, 50 mg, or 100 mg dosage, respectively. Two subjects in each dose group were administered matching placebo. Plasma concentrations of iguratimod were measured till 72 hours after drug administration. Tolerability was evaluated by monitoring adverse events, clinical laboratory tests, and 12-lead electrocardiograms.
Results:
The mean area under the concentration-time curve from 0 to 72 hours (AUClast) were 11.9, 25.2, and 51.8 mg × h/L and the maximum plasma concentration (Cmax) were 1.15, 2.33, and 4.78 mg/L in 25, 50 and 100 mg dose groups, respectively. All doses of iguratimod were well tolerated without serious adverse events or clinically meaningful changes.
Go to : 
REFERENCES
1. Koch AE. Review: angiogenesis: implications for rheumatoid arthritis. Arthritis Rheum. 1998; 41(6):951–962.
4. Sawada T, Hashimoto S, Tohma S, Nishioka Y, Nagai T, Sato T, Ito K, Inoue T, Iwata M, Yamamoto K. Inhibition of L-leucine methyl ester mediated killing of THP-1, a human monocytic cell line, by a new anti-inflammatory drug, T614. Immunopharmacology. 2000; 49(3):285–294.

5. Tanaka K, Kawasaki H, Kurata K, Aikawa Y, Tsukamoto Y, Inaba T. Iguratimod, a novel antirheumatic drug, inhibits both the activity and induction of cyclooxygenase-2 (COX-2) in cultured fibroblasts. Jpn J Pharmacol. 1995; 67(4):305–314.
6. Scrivo R, Di Franco M, Spadaro A, Valesini G. The immunology of rheumatoid arthritis. Ann N Y Acad Sci. 2007; 1108:312–322.

7. van der Linden MP, van der Woude D, Ioan-Facsinay A, Levarht EW, Stoeken-Rijsbergen G, Huizinga TW, Toes RE & van der Helm-van Mil AH. Value of anti-modified citrullinated vimentin and third-generation anti-cyclic citrullinated peptide compared with second-generation anti-cyclic citrullinated peptide and rheumatoid factor in predicting disease outcome in undifferentiated arthritis and rheumatoid arthritis. Arthritis Rheum. 2009; 60(8):2232–2241.

8. Guidelines for the management of rheumatoid arthritis: 2002 Update. Arthritis Rheum. 2002; 46(2):328–346.
9. Smolen JS, Landewe R, Breedveld FC, Dougados M, Emery P, Gaujoux-Viala C, Gorter S, Knevel R, Nam J, Schoels M, Aletaha D, Buch M, Gossec L, Huizinga T, Bijlsma JW, Burmester G, Combe B, Cutolo M, Gabay C, Gomez-Reino J, Kouloumas M, Kvien TK, Martin-Mola E, McInnes I, Pavelka K, van Riel P, Scholte M, Scott DL, Sokka T, Valesini G, van Vollenhoven R, Winthrop KL, Wong J, Zink A, van der Heijde D. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Ann Rheum Dis. 2010; 69(6):964–975.

10. Carmona L, Cross M, Williams B, Lassere M, March L. Rheumatoid arthritis. Best Pract Res Clin Rheumatol. 2010; 24(6):733–745.

11. Smolen JS, Aletaha D, Koeller M, Weisman MH, Emery P. New therapies for treatment of rheumatoid arthritis. Lancet. 2007; 370(9602):1861–1874.

12. Kawakami A, Tsuboi M, Urayama S, Matsuoka N, Yamasaki S, Hida A, Aoyagi T, Furuichi I, Nakashima T, Migita K, Kawabe Y, Nakashima M, Origuchi T, Eguchi K. Inhibitory effect of a new anti-rheumatic drug Iguratimod on costimulatory molecule expression, cytokine production, and antigen presentation by synovial cells. J Lab Clin Med. 1999; 133(6):566–574.
13. Kuriyama K, Higuchi C, Tanaka K, Yoshikawa H, Itoh K. A novel anti-rheumatic drug, Iguratimod, stimulates osteoblastic differentiation in vitro and bone morphogenetic protein-2-induced bone formation in vivo. Biochem Biophys Res ommun. 2002; 299(5):903–909.
14. Lu LJ, Bao CD, Dai M, Teng JL, Fan W, Du F, Yang NP, Zhao YH, Chen ZW, Xu JH, He PG, Wu HX, Tao Y, Zhang MJ, Han XH, Li XF, Gu JR, Li JH, Yu H. Multicenter, randomized, double-blind, controlled trial of treatment of active rheumatoid arthritis with Iguratimod compared with methotrexate. Arthritis Rheum. 2009; 61(7):979–987.
15. Hara M, Abe T, Sugawara S, Mizushima Y, Hoshi K, Irimajiri S, Hashimoto H, Yoshino S, Matsui N, Nobunaga M. Long-term safety study of Iguratimod in patients with rheumatoid arthritis. Mod Rheumatol. 2007; 17(1):10–16.

16. Hara M, Abe T, Sugawara S, Mizushima Y, Hoshi K, Irimajiri S, Hashimoto H, Yoshino S, Matsui N, Nobunaga M, Nakano S. Efficacy and safety of Iguratimod compared with placebo and salazosulfapyridine in active rheumatoid arthritis: a controlled, multicenter, double-blind, parallel-group study. Mod Rheumatol. 2007; 17(1):1–9.

17. Nakashima K, Zhou X, Kunkel G, Zhang Z, Deng JM, Behringer RR, de Crombrugghe B. The novel zinc finger-containing transcription factor osterix is required for osteoblast differentiation and bone formation. Cell. 2002; 108(1):17–29.

18. Tanaka K, Urata N, Mikami M, Ogasawara M, Matsunaga T, Terashima N, Suzuki H. Effect of Iguratimod and other anti-rheumatic drugs on adenocarcinoma colon 26-induced cachexia in mice. Inflamm Res. 2007; 56(1):17–23.

19. Lu LJ, Teng JL, Bao CD, Han XH, Sun LY, Xu JH, Li XF, Wu HX. Safety and efficacy of Iguratimod in the treatment of patients with active rheumatoid arthritis: a double blind, randomized, placebo-controlled and multicenter trial. Chin Med J (Engl). 2008; 121(7):615–619.
Go to : 
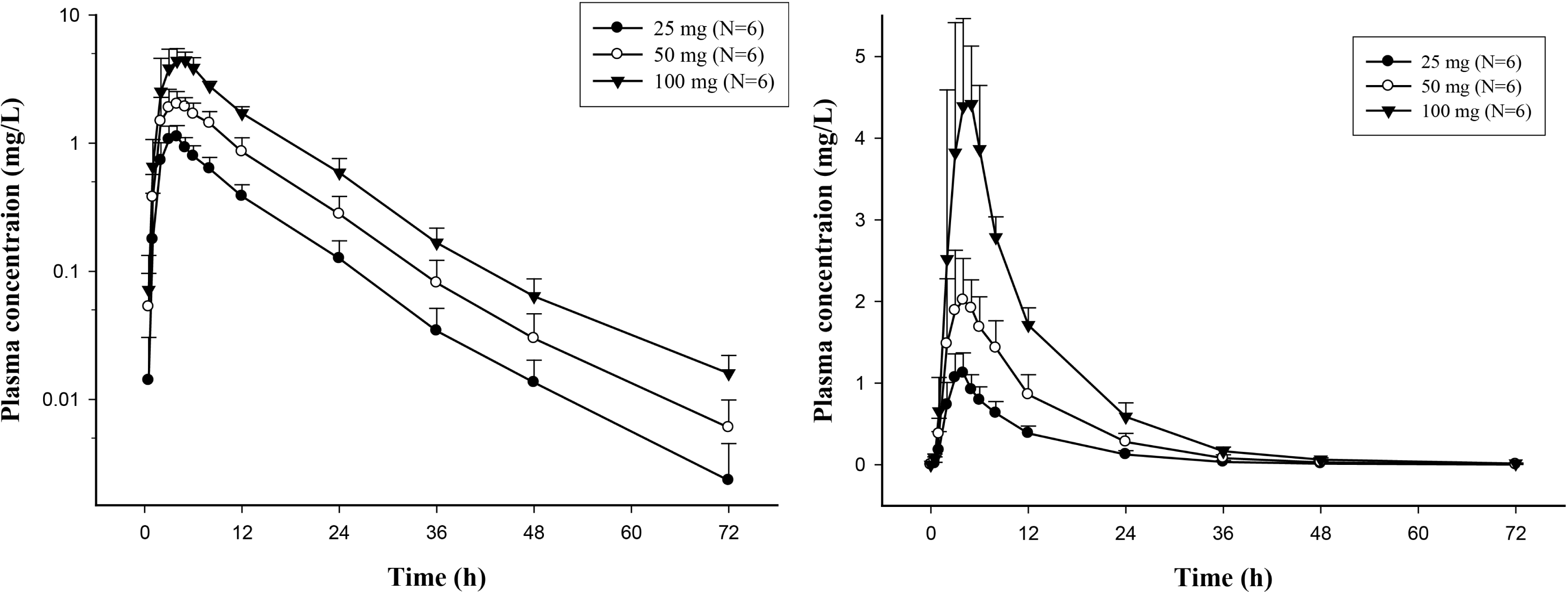 | Figure 1.Mean plasma concentration-time profiles after oral administration of Iguratimod 25, 50 and 100 mg in healthy Korean subjects. Left panel, semi-log scale; right panel linear scale. Bars represent standard deviations. |
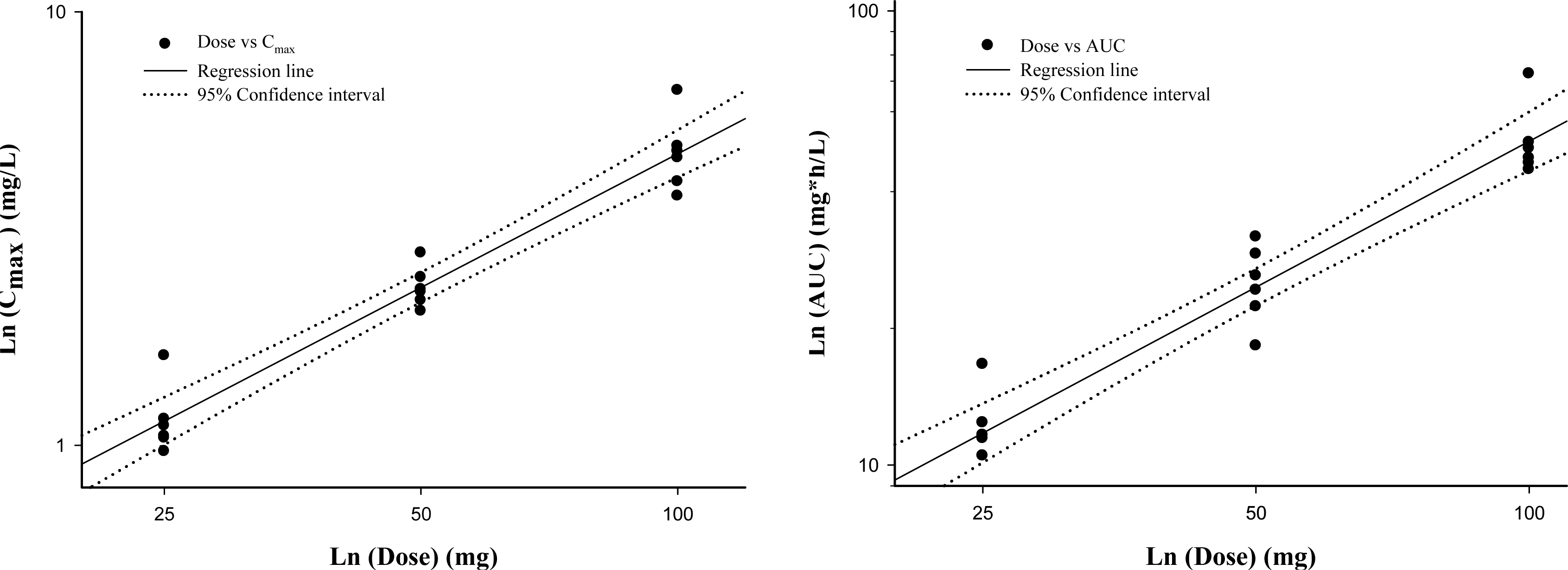 | Figure 2.Relationship between pharmacokinetic parameters and dose following single oral Iguratimod doses of 25, 50 and 100 mg. Left panel, log-transformed Cmax-dose relationship and right panel, log-transformed AUClast-dose relationship of Iguratimod. |
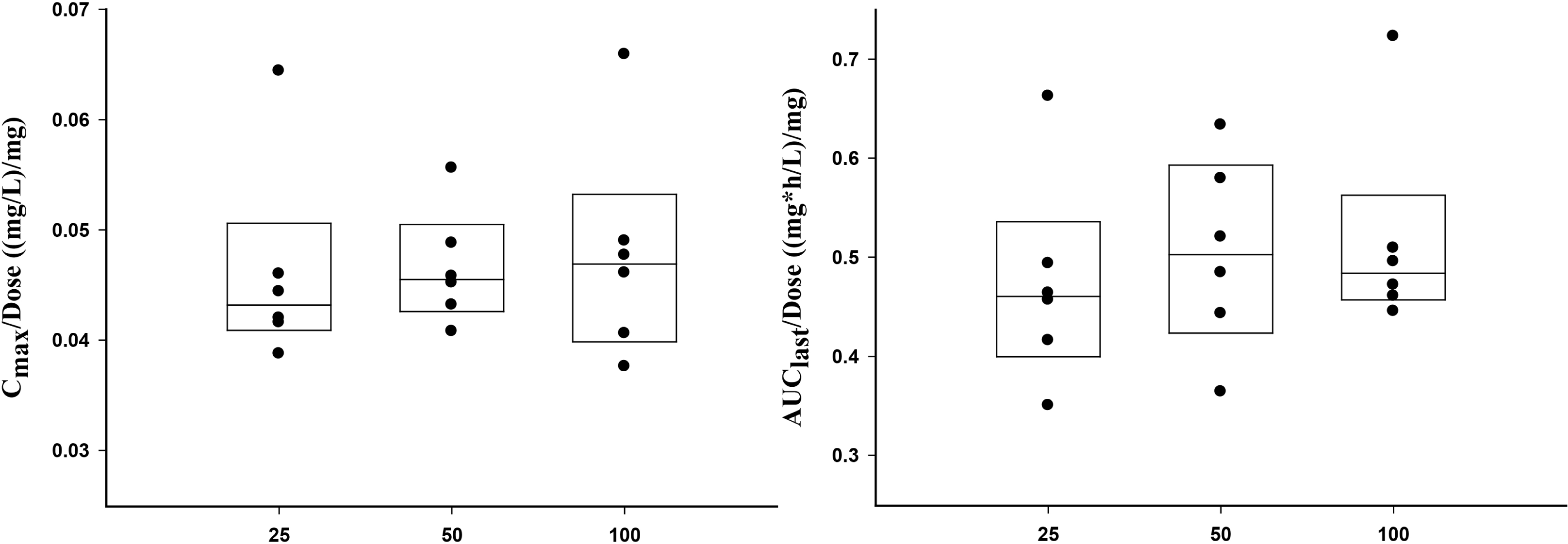 | Figure 3.Comparison of dose-normalized Cmax and AUClast after single oral administration of Iguratimod 25, 50 and 100 mg. The box edges show lower (25 th) and upper (75 th) quartiles and the line in the middle indicates the median value. Left panel, dose-normalized Cmax and right panel, dose-normalized AUClast. |
Table 1.
Demographic characteristics of the 24 healthy volunteers
Table 2.
Pharmacokinetic parameters of Iguratimod 25 mg, 50 mg and 100 mg




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download