Abstract
Figures and Tables
 | Fig. 1Depiction of the elements of decision making used to determine appropriate efforts to achieve glycemic targets. Adapted from Inzucchi et al. Diabetes Care 2012;35:1364-79 [1]. |
 | Fig. 2Antihyperglycemic therapy in type 2 diabetes: general recommendations. Moving from the top to the bottom of the figure, potential sequences of antihyperglycemic therapy. Adapted from Inzucchi et al. Diabetes Care 2012;35:1364-79 [1]. |
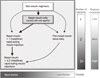 | Fig. 3Sequential insulin strategies in type 2 diabetes. Basal insulin alone is usually the optimal initial regimen. In patients willing to take more than one injection and who have higher HbA1c levels (≥ 9.0%), twice-daily premixed insulin or a more advanced basal plus mealtime insulin regimen could also be considered (curved dashed arrow lines). When basal insulin has been titrated to an acceptable fasting glucose but HbA1c remains above target, consider proceeding to basal plus mealtime insulin, consisting of one to three injections of rapid-acting analogs. A less studied alternative-progression from basal insulin to a twice-daily premixed insulin-could be also considered (straight dashed arrow line); if this is unsuccessful, move to basal plus mealtime insulin. Adapted from Inzucchi et al. Diabetes Care 2012;35:1364-79 [1]. |
Table 1

aLimited use in the U.S./Europe. bNotlicensed in the U.S. cPrescribing highly restricted in the U.S.;with drawn in Europe. dNotlicensed in Europe. eTo be available as a generic product in 2012, with expected significant reductions in cost. fDepends on type (analogs > human insulins) and dosage;CKD,chronickidneydisease;CVD, cardiovascular disease;DPP-4, dipeptidyl peptidase 4;GIP, glucose-dependent insulinotropic peptide; GLP-1, glucagon-like peptide1;HDL-C, HDL-cholesterol;LDL-C, LDL-cholesterol;PPAR, peroxisome proliferator-activated receptor;ProACTIVE, ProspectivePioglitazone Clinical Trialin Macrovascular Events(60); STOP-NIDDM, Study to Prevent Non-Insulin-Dependent Diabetes Mellitus (134);UKPDS, UK Prospective DiabetesStudy (29-33).




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download