Abstract
Anti-coccidial effects of the fruits of Tribulus terrestris (Tribuli fructus) ethanol extract (TTE) were studied with animal experiment following per oral administration with Eimeria (E.) tenella. This experiment was performed on the 3-day-old chicks (n=30). The animals were divided with 3 groups; TFE 15mg per animal+infected (n=10), TTE untreated+infected (n=10) and non-infected control (n=10). Animals were administrated with or without TTE during 1 week, and then inoculated with E. tenella. The anti-coccidial activity were evaluated with oocysts shedding numbers in stools, body weights changes and food intake changes. The TTE-inoclated animals revealed significantly decreased stool oocysts numbers (P<0.05) when compared to the TTE untreated animals. Also, TTE-treated animals showed more increased body weight gains (P<0.05) than the TTE untreated animals. These results demonstrate that TTE produce anticoccidial activities against E. tenella. TTE could be a promising treatment for the coccidiosis.
Coccidiosis is an infectious disease by protozoa Eimeria and an important parasitic zoonotic disease [1]. It induced severe losses of mortality, morbidity in the poultry industry. In addition, a lot of anti-coccidial medications to prevent Eimeria infection have revealed the decreased efficacy because some Eimeria species have developed resistance activity to anti-coccidials [2]. Coccidiosis is an important disease in the poultry industry [2]. Eimeria (E.) tenella is one of the most virulent species of Eimeria that causes hemorrhagic cecal coccidiosis in young poultry [3].
Tribulus terrestris is a creeping herb of the family Zygophyllaceae. It is 30 to 70 cm high and has pinnate leaves and yellow flowers. It is widely growing in tropical and moderate regions of the world, including Africa, Western Asia, China, Japan, Korea and southern Europe [4]. It has been used since ancient times in fork medicine for treating tonic, aphrodisiac, analgesic, astringent, stomachic, diuretic, hypertension, edema, eye problems, sexual impotency, inflammation, anti-infective, lithon-triptic, and rheumatoid arthritis [5678]. Previous studies have found that the fruits of T. terrestris (Tribuli fructus) contains steroidal saponins, protodioscin, alkaloids and flavonoids [9].
Recently, many natural herbal compounds have been studied to develop an alternative anti-coccidial drug [1]. However, the effects of Tribuli fructus ethanol extract (TTE) on coccidiosis has not been reported. Therefore, we are intend to evaluate the anticoccidial activity of the TTE in chicks following per oral inoculation with E. tenella.
We bought the dried Tribulus terrestris from an Oriental Pharmacy (Iksan, Korea). It was prepared following to the standard operation procedure as Korean Pharmacopoeia and Korean Herbal Pharmacopoeia, which are the official compendia of standard. Thereafter, we prepare TTE as following procedures. The dried Tribuli fructus was cut into pieces with 100 g. Then, those extracted 2 times with 50% (v/v) ethanol as 600 mL at 80℃ during 3 hours. After filtration with a 400-mesh net, the remnant was retreated with No. 5 Whatman filter paper. Then, it was concentrated with a rotary evaporator (EYELA, Tokyo, Japan) and the concentrated remnant was completely dried with freezing dryer (Labconco, USA). Thereafter, the dried remnant was collected in sterile tube and stored at −20℃.
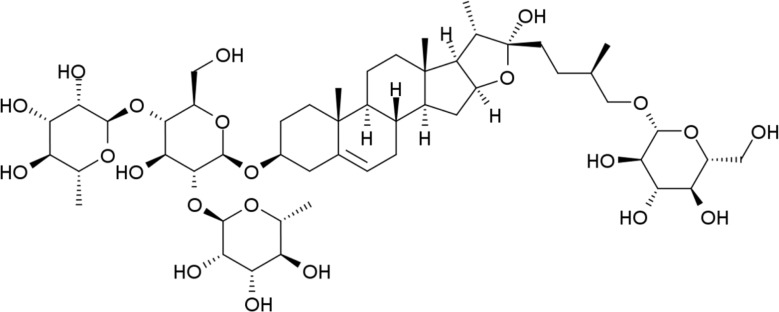
Protodioscin was prepared in order to use the standard compound of TTE. It was obtained from Sigma Aldrich (USA). The chemical structures of protodioscin was presented in Figure 1. We analyzed the protodioscin content in TFE compound with liquid chromatography (Waters Corp., USA). The column of LC was 2.1×50 mm, 1.7 µm C18 type ACQUITY UPLC BEH (Waters Corp., USA). The wavelength of the LC UV detector was adjusted to 300 nm. The temperature of LC column was adjusted to 30℃ with a flow rate of mobile phase at 0.6 mL/min (0.1% H3PO4 /Acetonnitrile).
This experiment was performed on the 3-day-old chicks (n=30) in the animal facility of Center for Animal Resources Development, Wonkwang University, Korea. The chicks were raised and acclimatized in a poultry facility room with standard temperature (28±2℃), humidity (50±5%) and light/dark cycle (12/12 hours). The chicks were provided a post-broiler feed without antibiotics and anti-coccidial compounds (Hanil Feed Co., Yongin, Korea) and tab water ad libitum. The animals were raised in grower wire-floored cages during experimental period. All procedures of animal experiments were conducted in accordance with the IACUC Guidelines by Wonkwang University and approved by the Institutional Animal Care and Use Committee of Wonkwang University (Approval No. WKU16-105). We made every effort to reduce the pain of the animals in this study.
Anti-coccidial activity of TTE were studied in chicks following per oral inoculation with E. tenella. The chicks were divided with 3 groups; TFE 15 mg per animal+infected (n=10), TTE untreated+infected (n=10) and non-infected control (n=10). We decided the dose of TFE following as the recommended feed additive concentration Animals were administrated with or without TTE during 1 week, and then inoculated with E. tenella. The anti-coccidial activity were evaluated with oocysts shedding numbers in stools, body weights changes and food intake changes.
Fecal oocysts were floated and gathered with 5.25% sodium hypochlorite. Then, those samples washed triples with phosphate buffered saline. Chicks were administrated per oral with sonde using a 24 gauge, animal feeding catheter stainless steel sonde for mouse (Popper & Sons, Inc., New York, USA) attached to a 3 mL syringe. The dose of administration for per oral infection has been approximated 104 of E. tenella oocysts in 1 mL of phosphate buffered saline. The animals of control group (n=10) were inoculated orally with phosphate buffered saline.
During the experimental procedure, the chicks were monitored twice daily for clinical signs, morbidity and mortality. Also, body weight gains and diet intake changes were evaluated with experimental animals. During 10 days post-infection, body weight gains and diet intake changes were individually checked.
Stools of the experimental animals were gathered from 6 to 10 days post-infection. The stool specimens were examined for the numbers of Eimeria oocysts using a standard fecal flotation method [10]. Briefly, 5 mL from each specimens was pelleted by centrifugation at 1500 g for 5 min. Thereafter, the pellet remnant was resuspended in aqueous saturated sodium chloride and filtered with a 1 mm size mesh to remove coarse fecal debris. The filtrates were submitted to the fecal flotation using 22 mm×22 mm coverslips. Then, the coverslip was put on a slide glass and observed in its entirety for Eimeria oocysts. Total number of oocysts was obtained using the following formula: [total number of oocysts=oocyst count×dilution factor×(fecal specimen volume/counting chamber volume)/number of chickens per cage].
Differences in mean oocysts secretion and mean weight changes between the 3 groups were evaluated by using one-way analysis of variance (ANOVA; GraphPad InStat; GraphPad Software Inc., San Diego, CA) and considered significant at P<0.05.
The extract yield of Tribuli fructus with 50% ethanol was 20.60% from the first prepared dry mass. We analyzed TTE contents with liquid chromatography. The chemical structure of protodioscin, the standard composition of TTE content, like as Figure 1. The composition of protodioscin in TTE was 310 µg/mL.
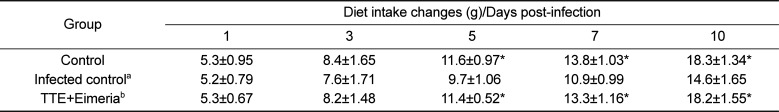
As shown in Table 1, oocyst secreting numbers was significantly higher in the infected animals than in the uninfected control animals (P<0.05). The oocysts number in stool was highest on day 8 post-infection (Table 1). TTE-treated animals revealed significantly decreased shedding oocysts in stool (P<0.05) when compared to TTE untreated animals. Also, TTE-treated animals showed more increased body weight gains (P<0.05) than the TTE untreated animals. TTE prevented the body weight decreases induced by coccidiosis in this study (Table 2). Also, diet intake was decreased in the TTE untreated chickens when compared to TTE-treated chickens (Table 3).
Coccidiosis in poultry industry is an important infectious disease induced by intracellular protozoa Eimeria species. Eimeria infection induce remarkable economic losses in livestock industry. Especially, E. tenella is a pathogenic protozoa causing severe coccidiosis in chickens and known to affect influencing experimental results obtained with infected chickens [1]. Coccidiosis is characterized by variable severe intestinal pathologic lesions, decreasing the enteric function, thus inducing to body weight changes, diarrhea, decreased diet intakes and severe mortality in the infected poultry herds [11].
The results of this study revealed that TTE had a remarkable anti-coccidial activity on Eimeria-infected chickens. The major contents of Tribuli fructus are saponins, protodioscin, diosgenins, alkaloids, and amides [1213]. Protodioscin is a steroidal saponin compound found in a number of plant species, most notably in the Tribulus, Trigonella and Dioscorea families. It is best known as the putative active content of the herbal aphrodisiac plant Tribuli fructus [9]. Several reports have shown that Tribuli fructus extract has an active antimicrobial effects against pathogenic Gram (+) bacteria such as Staphylococcus aureus and Enterococcus faecalis and pathogenic Gram (−) bacteria such as Escherichia coli. It indicates that there is a broadspectrum antibiotic of anti-microbial materials in the extract of Tribuli fructus [14].
In this study, anti-coccidial activities of TTE were studied in chicks following per oral inoculation with E. tenella. The TTE treated animals revealed significantly decreased stool oocysts numbers (P<0.05) when compared to the TTE untreated animals. Also, TTE-treated animals showed more increased body weight gains (P<0.05) than the TTE untreated animals. It is indicated that TTE had excellent anti-coccidial effects against Eimeria infection. It was implied that protodioscin as a steroidal saponin composition among contents of TTE may be provide anti-coccidal effects. TTE could be a promising treatment for the coccidiosis. This is the first study to confirm anticoccidial activity of TTE on Eimeria protozoa parasites.
References
1. Dalloul RA, Lillehoj HS. Poultry coccidiosis: recent advancements in control measures and vaccine development. Expert Rev Vaccines. 2006; 5(1):143–163. PMID: 16451116.

2. Williams RB. Relative virulences of a drug-resistant and a drugsensitive strain of Eimeria acervulina, a coccidium of chickens. Vet Parasitol. 2006; 135(1):15–23. PMID: 16361061.
3. Kaingu F, Liu D, Wang L, Tao J, Waihenya R, Kutima H. Anticoccidial effects of Aloe secundiflora leaf extract against Eimeria tenella in broiler chicken. Trop Anim Health Prod. 2017; 49(4):823–828. PMID: 28315138.
4. Zhang JD, Xu Z, Cao YB, Chen HS, Yan L, An MM, Gao PH, Wang Y, Jia XM, Jiang YY. Antifungal activities and action mechanisms of compounds from Tribulus terrestris L. J Ethnopharmacol. 2006; 103(1):76–84. PMID: 16169173.
5. Chhatre S, Nesari T, Somani G, Kanchan D, Sathaye S. Phytopharmacological overview of Tribulus terrestris. Pharmacogn Rev. 2014; 8(15):45–51. PMID: 24600195.
6. Borran M, Minaiyan M, Zolfaghari B, Mahzouni P. Protective effect of Tribulus terrestris fruit extract on cerulein-induced acute pancreatitis in mice. Avicenna J Phytomed. 2017; 7(3):250–260. PMID: 28748172.
7. Hammoda HM, Ghazy NM, Harraz FM, Radwan MM, ElSohly MA, Abdallah II. Chemical constituents from Tribulus terrestris and screening of their antioxidant activity. Phytochemistry. 2013; 92:153–159. PMID: 23642392.
8. Al-Bayati FA, Al-Mola HF. Antibacterial and antifungal activities of different parts of Tribulus terrestris L. growing in Iraq. J Zhejiang Univ Sci B. 2008; 9(2):154–159. PMID: 18257138.
9. Dinchev D, Janda B, Evstatieva L, Oleszek W, Aslani MR, Kostova I. Distribution of steroidal saponins in Tribulus terrestris from different geographical regions. Phytochemistry. 2008; 69(1):176–186. PMID: 17719068.
10. Lee HA, Hong S, Chung Y, Kim O. Sensitive and specific identification by polymerase chain reaction of Eimeria tenella and Eimeria maxima, important protozoan pathogens in laboratory avian facilities. Lab Anim Res. 2011; 27(3):255–258. PMID: 21998616.
11. Stotish RL, Wang CC, Meyenhofer M. Structure and composition of the oocyst wall of Eimeria tenella. J Parasitol. 1978; 64(6):1074–1081. PMID: 739302.
12. Bourke CA, Stevens GR, Carriqan MJ. Locomotor effects in sheeps of alkaloids identified in Australian Tribulus terrestris. Aust Vet J. 1992; 69(7):163–165. PMID: 1445080.
13. Yan W, Ohtani K, Kasai R, Yamasaki K. Steroidal Saponins from Fruits of Tribulus terrestris. Phytochemistry. 1996; 42(5):1417–1422. PMID: 9397208.
14. Soleimanpour S, Sedighinia FS, Safipour Afshar A, Zarif R, Ghazvini K. Antibacterial activity of Tribulus terrestris and its synergistic effect with Capsella bursa-pastoris and Glycyrrhiza glabra against oral pathogens: an in-vitro study. Avicenna J Phytomed. 2015; 5(3):210–217. PMID: 26101754.
Table 1
Oocyst shedding numbers in the chicks infected with Eimeria tenella with or without the ethanol extract of Tribulus terrestris fruits
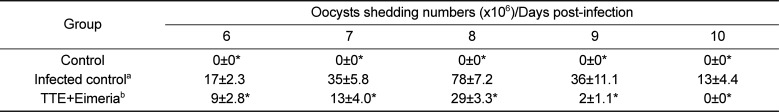
Table 2
Body weight changes (g) of the chicks infected with Eimeria tenella with or without the ethanol extract of Tribulus terrestris fruits
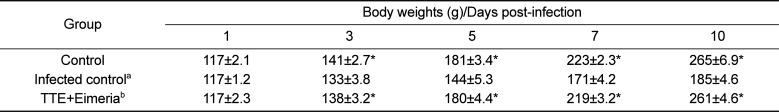
Table 3
Diet intake changes (g) of the chicks infected with Eimeria tenella with or without the ethanol extract of Tribulus terrestris fruits




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download