Abstract
The C57BL/6J-fe/fe mouse is a coat color mutant. The coat color of the homozygote mouse becomes progressively lighter with advancing age. The faded gene (fe) of C57BL/6J-fe/fe was mapped in a 2.0 cM distal to D10mit191 by our group. To make a high-resolution map, we used the Korean wild mouse (KWHM) for a backcross panel, which was captured in 1995 and has been maintained as an inbred line by our laboratory. In the inter-specific backcross panel (N=400), the fe gene was mapped to 1.0 cM distal to D10mit156. The gene order was defined: centromere -D10mit3/85 (1.3±0.6 cM)-D10mit155 (1.3±0.6 cM)-D10mit191 (2.0±0.7 cM)-D10mit156 (1.0±0.5 cM)-fe-D10mit193 (1.3±0.6 cM)-D10mit54 (1.0±0.5 cM)-D10mit44 (8.5±1.4 cM)-D10mit42 (10.0±1.5 cM). The measured distance between D10mit191 and D10mit 44 differed in both inter-specific (DBA/2) and intra-specific (KWHM) backcross panels (14.2 vs 13.8 cM). Taken together, our high-resolution linkage map of the fe locus from an intra-specific backcross panel will provide a good entry point to isolate the fe gene.
The mouse is a useful mammalian model for coat color research because of its high number of coat color mutations. Nearly 130 different genes with 1,000 different alleles in the mouse are known to affect coat color [1]. These coat color mutations are useful as visible genetic markers for genetic analysis and the generation of mouse knockout mutations. Variations of coat color are determined by combinations of four basic loci (A-D) in the mouse [2].
A number of coat color genes that correspond to human genetic diseases have now been cloned [3]. These mouse mutations have improved our knowledge of the mechanisms of pigmentary abnormalities in humans Dominant spotting (W) [4], microphthalmia (mi) [5], piebald (s) [6], and lethal spotting (ls) [7] mutants are animal model for piebaldism [8], Waardenburg syndrome 2 [9], Hirschsprung disease [10], and Shah-Waardenburg disease [11] in humans, respectively. Mutations at albino (c) locus are responsible for Oculocutaneous albinism I [12] in humans. Brown (b) [13], pink-eyed dilution (p) [14], and recessive yellow (e) [15] mouse mutants are useful animal model for Oculocutaneous albinism III [16], Oculocutaneous albinism II [17], and red hair [18] respectively in humans.
The C57BL/6J-fe/fe mouse is a coat color mutant that was first described by Oh [19,20]. It occurred as a spontaneous mutation in the KSB inbred strain at Nagoya University. An autosomal recessive gene transmits this mutation with 100% penetrance. Homozygotes can be distinguished at 14 days of age by a slight dilution of coat color but are more easily classified at 30 days. The coat color appears faded (black becoming gray) and the under fur is white (the proximal region of hairs is white). Their eyes are black. Faded shows no different dilution effects in black and agouti background mice. Loss of pigment occurs progressively with age, so that homozygotes are almost white by 1 year of age [19].
The fe gene was mapped to a region between D10mit191 and D10mit44 on mouse chromosome 10 by our group [21]. The nearest marker, D10mit191, was located 2.1 cM proximal regions from the fe gene on chromosome 10. This distance between the D10mit191 and fe genes makes it hard to identify the fe gene by chromosome working.
In this study, we have established a high-resolution genetic map in the vicinity of the fe gene by generating a backcross panel penetrant for the faded mutation. We also were compared genetic distances between intra-specific and inter-specific backcross panels.
C57BL/6J-fe/fe, C57BL/6J, and the Korean wild mouse (KWHM) were used in this study. KWHM were captured in 1995, and subsequently maintained in our laboratory by brother sister mating. The 19th generation of KWHM was used for an inter-specific backcross panel. The animals were maintained at 22±2℃ temperature, in 55±10% relative humidity, and under 12 hr artificial lighting from 08:30 to 20:30. The mice were given commercial laboratory chow made for mouse (Jeiljedang, South Korea) and water ad libitum. The animal study was conducted in accordance with and the approval of the Institutional Animal Care and Use Committee of Hallym University.
For histological characterization of hair shafts and hair bulbs, C57BL/6J-fe/fe and control (C57BL/6J) mice at 1 and 12 weeks of age were sacrificed under ether anesthesia. To investigate their hair bulbs, mid-dorsal skin was fixed in 10% neutral buffered formalin solution for 24 hr, and hair was removed with scissors. Then the skin tissue was fixed in 10, 20, and 30% sucrose phosphate buffer solutions over one day. The tissue was embedded in Tissue-tek (4583, Miles Inc., USA), quickly frozen, and cut into 10-µm sections using a cryostat-microtome. After rinsing in 0.1 M phosphate buffer for 10 min three times, they were stained with hematoxylin and eosin. Tissues were viewed under a Zeiss Axioskop light microscope (Carl Zeiss, Germany). Affected faded mice were carefully confirmed by their faded coat color at about 4 weeks of age.
C57BL/6J-fe/fe mice were crossed with the KWHM mice to produce F1. F1 mice were then backcrossed to C57BL/6J-fe/fe mice, and four hundred inter-specific progeny were analyzed. Linkage analyses were performed using microsatellite markers by using a modification of a previously described method [22]. Six MIT markers over a 27-cM region between D10mit3/D10mit85 and D10mit42 were used for a high-resolution linkage test. Genomic DNA was prepared from the tail of N2 progeny by standard SDS/proteinase K lysis and the phenol/chloroform extraction method.
PCR reactions using 400 ng of template DNA were carried out in a total volume of 25 µL containing 10 mM Tris-HCl (pH 8.3), 50 mM KCl, 1.5 mM MgCl2, 0.2 mM dNTPs, 2 pmol of each primer, and 1.75 U Taq polymerase (Takara, Japan). Target DNA was amplified in a Cyclogene Dri-Block® cycler (Techne Cambridge Ltd., U.K.), with denaturation at 94oC for 30 s, annealing at 57-62℃ for 1 min, extension at 72℃ for 2 min, followed by a final extension at 72℃ for 7 min at the end of 30 cycles. Modifications in annealing temperature and/or magnesium concentration were made as needed to optimize the results with some primer pairs. The amplification products were separated on 15% polyacrylamide gel and detected by ethidium bromide staining. Percentage recombination and standard error were calculated according to Green [23]. Genetic distances are represented as cM±standard error, and order was established by minimizing putative double crossover events.
The coat color of a homozygote mouse (C57BL/6J-fe/fe) becomes progressively lighter with advancing age (Figure 1). The outer fur and under fur are white from the proximal region of the hair and show an age-related pigment change toward white. However, these shafts appear normal in structure. Cleared hairs of a faded mouse display various clumps of pigment granules, which occur sporadically in the medulla of the shaft. The hair bulbs of C57BL/6J mouse show normal pigmentation (Figure 2A). Homozygotes of C57BL/6J-fe/fe can be distinguished at 2 weeks of age by a slight dilution of coat color. However, the hair bulbs of a C57BL/6J-fe/fe mouse are clearly abnormal from 1 week of age (Figure 2B). Melanin of the hair of a C57BL/6J-fe/fe mouse completely disappears by 12 weeks of age (Figure 2C).
To create high-resolution mapping data from the analysis of the intra-specific backcross, we used the Korean wild mouse (KWHM) for a backcross panel. The KWHM mice were captured in 1995 and have been maintained in an inbred line by our laboratory. The KWHM strain was chosen over others because it is divergent enough to be distinguished from the inbred strains with microsatellite markers. Four hundred interspecific backcross progeny were analyzed at 6 MIT markers over a 27-cM region between D10mit3/D10mit85 and D10mit42. Twenty-two mice showed a recombination between fe and D10mit3/D10mit85 in the proximal region of the fe locus, yielding a genetic distance of 5.6±1.1 cM between these two loci. Twelve mice exhibited a haplotype in which the recombination occurred between fe and D10mit191, indicating that D10mit191 is 3.0±0.9 cM proximal to fe. For the D10mit156 marker, 4 of the 400 N2 animals showed recombination with fe, giving an estimated distance between D10mit156 and fe of 1.0±0.5 cM (Figure 3). This results show that D10mit156 provides a useful anchor for the isolation of YACs containing the fe gene.
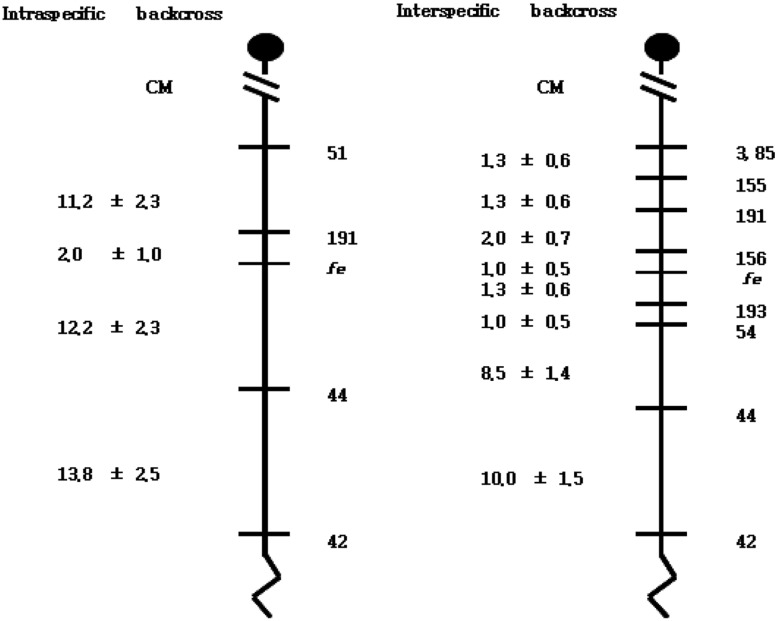
In the distal region of the fe locus, 83 and 44 mice showed a recombination between fe and D10mit42 and between fe and D10mit44, respectively. Estimated distances between fe and D10mit44, and between D10mit44 and D10mit42 are 10.8±1.6 and 10.0±1.5 cM, respectively. In addition, 8 mice showed recombination between fe and D10mit54, placing these markers at a distance of 2.3±0.7 cM. Two double recombination events between fe and D10mit54 were found in the 400 N2 animals. Five mice exhibited a haplotype in which recombination occurred between fe and D10mit193, indicating that D10mit193 is 1.3±0.6 cM distal to fe. Since D10mit3/D10mit85 has been shown to be proximal to D10mit42, the gene order was defined as follows: centromere-D10mit3/D10mit85-D10mit155-D10mit191-D10mit156-fe-D10mit193-D10mit54-D10mit44-D10mit42 (Figure 4). The linkage maps from both backcrosses are shown in Figure 4. The order of the loci analyzed was identical between the two maps. The measured distance between D10mit191 and D10mit44 differed in these two crosses (14.2 vs.13.8 cM).
Because the C57BL-fe/fe mouse is a useful animal model for pigmentary abnormalities in humans, the identification of the mutated gene in C57BL-fe/fe mice will help in the identification of the mechanism of pigmentary abnormalities. The fe gene was mapped to a region between D10mit191 and D10mit44 on mouse chromosome 10 by our group [21]. The nearest marker, D10mit191, was located 2.1 cM proximal regions from the fe gene on chromosome 10. This distance between D10mit191 and the fe genes makes it hard to identify the fe gene by chromosome working. The gene product and function of the fe gene are not evident based on previous studies. For the first step to identify the fe gene, we have established a high-resolution genetic map in the vicinity of the fe gene by generating a backcross panel penetrant for the faded mutation. From the inter-specific backcross, (C57BL/6J-fe/fe X KWHM)F1 X C57BL/6J-fe/fe, 400 progenies were used for high-resolution mapping of the fe gene. The fe gene was mapped to 1.0 cM distal to D10mit156. Therefore, the gene order was defined: centromere-D10mit3/85 (1.3±0.6 cM)-D10mit155 (1.3±0.6 cM)-D10mit191 (2.0±0.7 cM)-D10mit156 (1.0±0.5 cM)-fe-D10mit193 (1.3±0.6 cM)-D10mit54 (1.0±0.5 cM)-D10mit44 (8.5±1.4 cM)-D10mit42 (10.0±1.5 cM).
In this study, intra-specific and inter-specific genetic distances were compared between each pair of adjacent markers common to both backcrosses. The measured distance between D10mit191 and D10mit44 differed in these two crosses (14.2 vs 13.8 cM). Although Mus musculus species originated from same ancestral Mus musculus one million years ago, the Mus musculus domesticus (C57BL/6J and CBA/J) and Mus musculus molossinus (Korean wild mouse) line diverged [24] almost 2,000 years ago. Because of this, structural rearrangements in the organization of genetic information probably represent an important mechanism for the establishment or maintenance of speciation. In a previous study it was concluded that the use of inter-specific crosses do not introduce any systemic bias into measurement of either linkage or gene order [24]. Our mapping results of the fe gene in intra- and inter-specific backcross panels, therefore, may be thought reasonable.
From mapping information obtained from the Mouse Genome Informatics (The Jackson Laboratory), there are many genes between D10mit156 and D10mit193 on mouse chromosome 10. However, there is no gene related to coat color to our knowledge. The mouse map location of the fe locus appears to place it in a region homologous to human 6q21. No human candidate genes have yet been localized in this interval, which may potentially be the human homology of the fe gene.
The hair bulbs of the C57BL/6J-fe/fe mouse are abnormal from 1 week of age. Melanin of the hair of C57BL/6J-fe/fe mouse completely disappears by 12 weeks of age and shows a faded coat color. Mutation in the C57BL/6J-fe/fe mouse seems to differ with previously described mutations in mice that reduce the intensity of hair pigmentation or age-related pigment change (for example, d, ln, bg, p, Ga, rp, gl, gr, mh, Sl, si) [19,20].
In conclusion, we made a high-resolution map that flanks the fe gene region on mouse chromosome 10. The fe gene was mapped to 1.0 cM distal to D10mit156. Thus, the gene order was defined: centromere -D10mit3/85 (1.3±0.6 cM)-D10mit155 (1.3±0.6 cM)-D10mit191 (2.0±0.7 cM)-D10mit156 (1.0±0.5 cM)-fe-D10mit193 (1.3±0.6 cM)-D10mit54 (1.0±0.5 cM)-D10mit44 (8.5±1.4 cM)-D10mit42 (10.0±1.5 cM). The nearest marker of the fe gene, D10mit156, is useful to isolate the fe gene using a candidate gene approach and chromosome walking. Taken together, our high-resolution linkage map of the fe locus from an intra-specific backcross panel will provide a good entry point to isolate the fe gene.
Acknowledgments
This work was supported by Priority Research Centers Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012R1A6A1048184).
References
1. Steingrímsson E, Copeland NG, Jenkins NA. Mouse coat color mutations: from fancy mice to functional genomics. Dev Dyn. 2006; 235(9):2401–2411. PMID: 16691561.

2. Green MC. Genetic variants and strains of the laboratory mouse. 1981. Stuttgart: Gustav Fischer Verlag.
3. Marks PW, Bandura JL, Shieh DB, Foernzler D, Beier DR, Kwiatkowski DJ. The spontaneous coat color mutant white nose (wn) maps to murine chromosome 15. Mamm Genome. 1999; 10(7):750–752. PMID: 10384053.
4. Kunisada T, Yoshida H, Yamazaki H, Miyamoto A, Hemmi H, Nishimura E, Shultz LD, Nishikawa S, Hayashi S. Transgene expression of steel factor in the basal layer of epidermis promotes survival, proliferation, differentiation and migration of melanocyte precursors. Development. 1998; 125(15):2915–2923. PMID: 9655813.

5. O'Reilly FM, Brat DJ, McAlpine BE, Grossniklaus HE, Folpe AL, Arbiser JL. Microphthalmia transcription factor immunohistochemistry: a useful diagnostic marker in the diagnosis and detection of cutaneous melanoma, sentinel lymph node metastases, and extracutaneous melanocytic neoplasms. J Am Acad Dermatol. 2001; 45(3):414–419. PMID: 11511840.
6. Lamoreux ML. Strain-specific white-spotting patterns in laboratory mice. Pigment Cell Res. 1999; 12(6):383–390. PMID: 10614578.

7. Gariepy CE, Williams SC, Richardson JA, Hammer RE, Yanagisawa M. Transgenic expression of the endothelin-B receptor prevents congenital intestinal aganglionosis in a rat model of Hirschsprung disease. J Clin Invest. 1998; 102(6):1092–1101. PMID: 9739043.

8. Richards KA, Fukai K, Oiso N, Paller AS. A novel KIT mutation results in piebaldism with progressive depigmentation. J Am Acad Dermatol. 2001; 44(2):288–292. PMID: 11174389.
9. Wang C, Kim E, Attaie A, Smith TN, Wilcox ER, Lalwani AK. A PAX3 polymorphism (T315K) in a family exhibiting Waardenburg Syndrome type 2. Mol Cell Probes. 1998; 12(1):55–57. PMID: 9584079.
10. Shanske A, Ferreira JC, Leonard JC, Fuller P, Marion RW. Hirschsprung disease in an infant with a contiguous gene syndrome of chromosome 13. Am J Med Genet. 2001; 102(3):231–236. PMID: 11484199.

11. Edery P, Attié T, Amiel J, Pelet A, Eng C, Hofstra RM, Martelli H, Bidaud C, Munnich A, Lyonnet S. Mutation of the endothelin-3 gene in the Waardenburg-Hirschsprung disease (Shah-Waardenburg syndrome). Nat Genet. 1996; 12(4):442–444. PMID: 8630502.

12. Tomita Y, Miyamura Y, Kono M, Nakamura R, Matsunaga J. Molecular bases of congenital hypopigmentary disorders in humans and oculocutaneous albinism 1 in Japan. Pigment Cell Res. 2000; 13(Suppl 8):130–134. PMID: 11041370.

13. Johnson R, Jackson IJ. Light is a dominant mouse mutation resulting in premature cell death. Nat Genet. 1992; 1(3):226–229. PMID: 1303241.

14. Fukuta K, Imamura K, Goto N. Pink-eyed dilution, a coat color mutation in the Japanese field vole (Microtus montebelli). Jikken Dobutsu. 1991; 40(3):375–379. PMID: 1915604.
15. Wada A, Okumoto M, Tsudzuki M. Tawny: a novel light coat color mutation found in a wild population of Mus musculus molossinus, a new allele at the melanocortin 1 receptor (Mc1r) locus. Exp Anim. 1999; 48(2):73–78. PMID: 10374067.
16. Toyofuku K, Wada I, Valencia JC, Kushimoto T, Ferrans VJ, Hearing VJ. Oculocutaneous albinism types 1 and 3 are ER retention diseases: mutation of tyrosinase or Tyrp1 can affect the processing of both mutant and wild-type proteins. FASEB J. 2001; 15(12):2149–2161. PMID: 11641241.

17. Brilliant MH. The mouse p (pink-eyed dilution) and human P genes, oculocutaneous albinism type 2 (OCA2), and melanosomal pH. Pigment Cell Res. 2001; 14(2):86–93. PMID: 11310796.
18. Voisey J, Box NF, van Daal A. A polymorphism study of the human Agouti gene and its association with MC1R. Pigment Cell Res. 2001; 14(4):264–267. PMID: 11549109.

19. Oh YS, Tomita T. Linkage of faded gene (fe) to chromosome 6 of the mouse. Jikken Dobutsu. 1987; 36(1):73–77. PMID: 3816991.
20. Oh YS, Tomita T, Kondo K. Faded, a mutation in the KSB strain of mouse which shows age-related pigment changes. Jikken Dobutsu. 1986; 35(2):131–138. PMID: 3732403.
21. Oh SH, Nam Y, Suh JG. Mapping of the Faded (fe) Gene to a Region between D10mit191 and D10mit44 on Mouse Chromosome 10. Lab Anim Res. 2011; 27(1):41–46. PMID: 21826159.
22. Suh JG, Yamanishi T, Matsui K, Tanaka K, Wada K. Mapping of the gracile axonal dystrophy (gad) gene to a region between D5Mit197 and D5Mit113 on proximal mouse chromosome 5. Genomics. 1995; 27(3):549–551. PMID: 7558041.

23. Green EL. Linkage, recombination and mapping. Genetics and probability in animal breeding experiments. 1981. New York: Oxford University Press;p. 77–133.

24. Moriwaki K, Shiroishi T, Yonekawa H. Genetics in wild mice: Its application to biomedical research. 1994. Japan: Japan Scientific societies press;p. 141–295.
Figure 1
Age-related coat color change in C57BL/6J-fe/fe mice. The coat color of homozygous mice becomes progressively lighter with advancing age (black becoming gray).
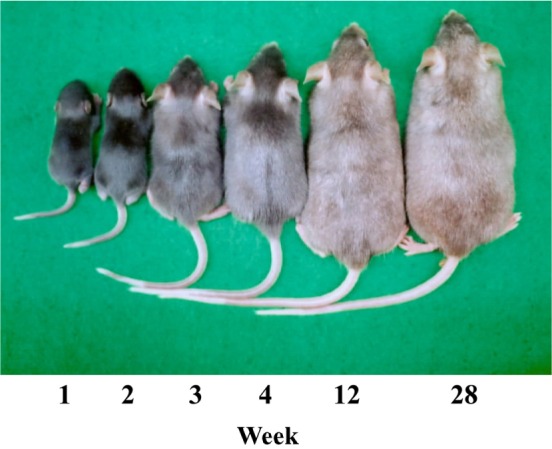
Figure 2
The skin of the C57BL/6J (A) and C57BL/6J-fe/fe mice (B,C). In faded mice, the reduction of melanin content in hair bulbs with age is shown. H&E staining (× 400). M, Melanin; MC, Melanocyte; H, Hair.
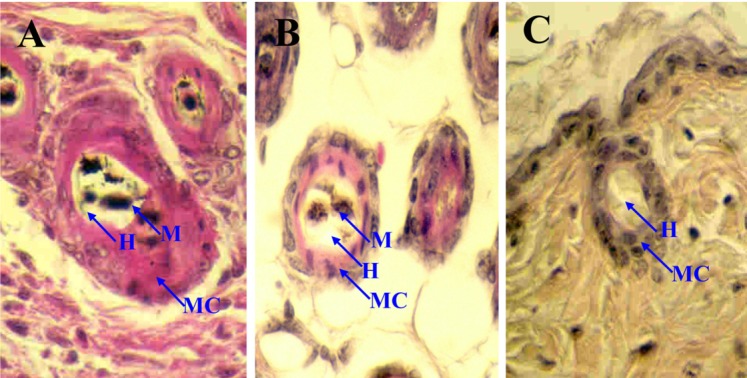
Figure 3
Haplotype analysis of an interspecific backcross between fe and KWHM mice (C57BL/6J-fe/fe X KWHM)F1 X C57BL/6J-fe/fe. Each column represents a chromosomal haplotype identified in the backcross and inherited from the F1 parent. The number of individuals is listed beneath each column. Black boxes represent fe alleles, and white boxs represent KWHM alleles as determined by SSLP scoring with markers indicated at the left.

Figure 4
Genetic linkage maps of proximal chromosome 10 as derived from an interspecific backcross panel (C57BL/6J-fe/fe X KWHM)F1 X C57BL/6J-fe/fe) generated in this study. The distances of the intervals between loci and/or markers are not drawn to the same scale, and the locations of mapped human homologs are taken from the Mouse Genome Informatics (The Jackson Laboratory), Distances between intervals are shown in centimorgans±standard error to the left of the map. The linkage map of intra-specific backcross panel came from Oh et al., 2011[21].




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download