Abstract
An abnormal swelling was identified in the distal portion of the right femur in a 1-year-old non-obese diabetic (NOD) mouse. Grossly, a large mass of the distal femur was observed in the right femur. Lesions were poorly marginated, associated with destruction of the cancellous and cortical elements of the bone, and showed ossification within the soft tissue component. Histologically, the tumor was identified as a poorly differentiated sarcoma. Histopathologic examination of the bone masses revealed invasive proliferation of poorly differentiated neoplastic mesenchymal cells forming streams, bundles, and nests, which resulted in destruction of normal bone. Neoplastic cells exhibited random variation in cellular appearance and arrangement, as well as matrix composition and abundance. Haphazard and often intermingling patterns of osteogenic, chondroblastic, lipoblastic, and angiogenic tissues were present. Larger areas of neoplastic bone and hyaline cartilage contained multiple large areas of hemorrhage and necrosis bordered by neoplastic cells. The mass was diagnosed as an osteosarcoma. To our knowledge, this is the first spontaneous osteosarcoma in an NOD mouse.
Osteosarcomas are primary malignant tumors of bone that are characterized by the production of osteoid or immature bone by the malignant cells [1-3]. Osteosarcomas in humans are uncommon tumors. Only about 400 cases are diagnosed each year in the United States, mainly in children and adolescents [4]. Despite their rarity, osteosarcomas are the most common primary malignancy of bone in children and adolescents, and the fifth most common malignancy among adolescents and young adults aged 15 to 19 [5,6]. Osteosarcomas are the most common primary bone tumor in dogs, accounting for more than 80% of malignant bone neoplasms [7-11]. They are most common in middle-aged to older large-breed dogs, with a higher incidence in male dogs [10-13]. In mice, spontaneous osteosarcoma is a rare neoplasm [14-17]. The incidence of skeletal osteosarcoma is 0.2% in both male and female control ICR mice [18] and ranges from 0 to 0.5% and 0 to 6.7% for females in control B6C3F1 mice [19]. Until now, there have been no reports about osteosarcoma in non-obese diabetic (NOD) mice.
This study sought to investigate the abnormal swelling in the right femur of an NOD mouse. A one-year-old male NOD mouse was obtained from the Animal Facilities of the Center for Animal Resources Development, Wonkwang University, Korea. In the previous 3 weeks, an abnormal swelling and growth was identified in the distal portion of the right femur in the animal in question. Urine analysis was conducted with an Urostick and the result was the following: as leucocytes-, nitrogen-, urobilinogen-, protein 30 mg/dL, pH 5.0, blood >200 erythrocytes/µL, specific gravity 1.030, ketones-, bilirubin-, glucose-. Grossly, a large mass on the distal femur was observed in the right femur. Lesions were poorly marginated, associated with destruction of the cancellous and cortical elements of the bone, and showed ossification within the soft tissue component. Necropsy was conducted and the organs were submitted to gross examination and trimmed. The trimmed tissues were fixed in 10% neutral buffered formalin, and embedded in paraffin. Four µm sections were made and stained with hematoxylin and eosin for histopathological examination.
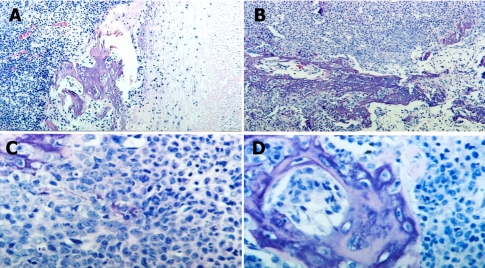
Gross examination of the necropsied animal revealed abnormal swelling of the right femur which had a hard consistency. In the subcutaneous layer of the right femur lesion, jelly-like degenerative materials were found. The cut surface of the 4×5 cm femoral mass was poorly marginated, and white (Figure 1). Also, swelling of inguinal lymph nodes and atrophy of testes were detected. Histologically, the femoral mass was identified as a poorly differentiated sarcoma. Histopathologic examination of the femoral bone mass revealed an invasive proliferation of poorly differentiated neoplastic mesenchymal cells forming streams, bundles, and nests, which resulted in destruction of normal bone (Figure 2). Neoplastic cells exhibited random variation in cellular appearance and arrangement, as well as matrix composition and abundance. Haphazard and often intermingling patterns of osteogenic, chondroblastic, lipoblastic, and angiogenic tissues were present. Larger areas of neoplastic bone and hyaline cartilage contained multiple large areas of hemorrhage and necrosis bordered by neoplastic cells. In cases of inguinal lymph nodes, a major histopathological finding was infiltration of lymphocytes. In the liver, infiltration of lymphocytes was a major lesion. In the testes, major histopathological findings were degeneration of seminiferous tubules and infiltration of lymphocytes. There were no metastatic lesions in the internal organs. The neoplastic cells were found only in the right femoral mass.
The mass in this study was diagnosed as an osteosarcoma. Primary bone sarcomas that include osteosarcomas are divided into two categories, central and peripheral, with central tumors being higher in incidence and involvement [20]. Tumor types are typically named for the matrix they produce; however, when tumors contain multiple cell types and matrices, determination of histologic classification may be problematic. According to the World Health Organization classification of bone and joint tumors, an osteosarcoma is a primary malignant neoplasm of mesenchymal tissue that gives rise to a variety of patterns, but always includes the production of bone by malignant osteoblasts [20]. In animals, dogs have a relatively high occurrence of osteosarcoma [2,10]. However, spontaneous osteosarcoma of mouse is rarely reported [14-17]. Osteosarcomas arise commonly in the proximal humerus, distal radius, distal femur, and proximal and distal tibia in the dog [9-11,21,22]. Primary osteosarcomas rarely cross joints or invade adjacent bones during early tumorigenesis, but as the tumor increases in size, both events occur more commonly [8,20,23]. In domestic animals, osteosarcomas can be classified as poorly differentiated, osteoblastic, chondroblastic, fibroblastic, telangiectatic, and giant cell types [10,20]. Mesenchymal tissue is known to possess high differentiation potential, and this explains the multipotential characteristics of the neoplasm in this case.
The mass in this study was diagnosed as an osteosarcoma. To our knowledge, this is the first spontaneous osteosarcoma in an NOD mouse.
Acknowledgments
This study was supported by the research fund of Wonkwang University in 2011. We wish to acknowledge our appreciation of Sang-Jun Han and Dong-Woo Kim, research assistants in the Center for Animal Resources Development, Wonkwang University, for carrying out the histopathological work.
References
1. Huvos A. Bone Tumors: Diagnosis, Treatment, Prognosis. 1991. 2nd ed. Philadelphia: WB Saunders;p. 784.
2. Sissons HA. The WHO classification of bone tumors. Recent Results Cancer Res. 1976; (54):104–108. PMID: 1070715.

3. McKenna RJ, Schwinn CP, Soong KY, Higinbotham NL. Sarcomas of the osteogenic series (osteosarcoma, chondrosarcoma, parosteal osteogenic sarcoma, and sarcomata arising in abnormal bone): an analysis of 552 cases. J Bone Joint Surg Am. 1966; 48(1):1–26.
4. Gurney JG, Swensen AR, Bulterys M. Ries LA, editor. Malignant bone tumors. Cancer Incidence and Survival Among Children and Adolescents: United States SEER Program 1975-1995. 1999. Bethesda: National Cancer Institute;p. 99–110.
5. Smith MA, Gurney JG, Ries LA. Ries LA, editor. Cancer in adolescents 15 to 19 years old. Cancer Incidence and Survival among Children and Adolescents: United States SEER Program 1975-1995. 1999. Bethesda: National Cancer Institute;p. 111–200.
6. Stiller CA, Bielack SS, Jundt G, Steliarova-Foucher E. Bone tumours in European children and adolescents, 1978-1997. Report from the automated childhood cancer information system project. Eur J Cancer. 2006; 42(13):2124–2135. PMID: 16919776.

7. Brodey RS, Riser WH. Canine osteosarcoma: a clinicopathologic study of 194 cases. Clin Orthop Relat Res. 1969; 62:54–64. PMID: 5251443.
8. Jones RD, Reid R, Balakrishnan G, Barrett A. Multifocal synchonous osteosarcoma: the Scottish bone tumour registry experience. Med Pediatr Oncol. 1993; 21(2):111–116. PMID: 8433676.
9. Moore A. Osteosarcoma in dogs: managing a destructive disorder. Vet Med. 2001; 96(7):539–546.
10. Thompson KG, Pool RR. Meuten DJ, editor. Tumors of bone. Tumors in Domestic Animals. 2002. Ames: Iowa State Press;p. 245–283.
11. Watson C, Lucroy M. Primary appendicular bone tumors in dogs. Comp Cont Ed Pract Vet. 2002; 24(2):128–138.
12. LaRue SM, Withrow SJ, Wrigley RH. Radiographic bone surveys in the evaluation of primary bone tumors in dogs. J Am Vet Med Assoc. 1986; 188(5):514–516. PMID: 3457000.
13. Misdorp W, Hart AA. Some prognostic and epidemiologic factors in canine osteosarcoma. J Natl Cancer Inst. 1979; 62(3):537–545. PMID: 283283.

14. Albassam MA, Courtney CL. Mohr U, editor. Non-neoplastic and neoplastic of the bone. Pathology of the Aging Mouse. 1996. Washington DC: ILSI press;p. 425–437.
15. Wadsworth PF. Mohr U, editor. Classification of bone neoplasms and presentation of data on the incidence and types in C57BL/10J mice. Pathology of the Aging Mouse. 1996. Washington DC: ILSI press;p. 439–443.
16. Frith CH, Johnson BP, Highman B. Osteosarcomas in BALB/c female mice. Lab Anim Sci. 1982; 32(1):60–63. PMID: 6952039.
17. Frith CH, Littlefield NA, Umholtz R. Incidence of pulmonary metastases for various neoplasms in BALB/cStCrlf C3H/Nctr female mice fed N-2-fluorenylacetamide. J Natl Cancer Inst. 1981; 66(4):703–712. PMID: 6262551.
18. Maita K, Hirano M, Harada T, Mitsumori K, Yoshida A, Takahashi K, Nakashima N, Kitazawa T, Enomoto A, Inui K, Shirasu Y. Mortality, major cause of moribundity, and spontaneous tumors in CD-1 mice. Toxicol Pathol. 1988; 16(3):340–349. PMID: 3194656.

19. Long PH, Leininger JR. Maronpot RR, editor. Bones, joints, and synovial. Pathology of the Mouse. 1999. Vienna: Cache River Press;p. 645–678.
20. Slayter MV, Boosinger TR, Pool RR, Dammrich K, Misdorp W, Larsen S. Malignant tumors. World Health Organization Histologic Classification of Bone and Joint Tumors of Domestic Animals. 1994. Washington DC: Armed Forces Institute of Pathology, American Registry of Pathology and the World Health Organization Collaborating Center for Comparative Oncology;p. 7–11.
21. Moore GE, Mathey WS, Eggers JS, Estep JS. Osteosarcoma in adjacent lumbar vertebrae in a dog. J Am Vet Med Assoc. 2000; 217(7):1038–1040. PMID: 11019712.

22. Thrall DE. Thrall DE, editor. Bone tumors versus bone infections. Textbook of Veterinary Diagnostic Radiology. 1998. Philadelphia: WB Saunders;p. 161–168.
23. Vigorita VJ. Vigorita VJ, editor. Bone tumors. Orthopaedic Pathology. 1999. Philadelphia: Lippincott Williams and Wilkins;p. 1–345.
Figure 1
Gross findings for the masses of the right femur. (A) Abnormal swelling of right femur. (B) Necropsy findings. (C) The mass had a hard consistency. (D) The cut surface of the femoral mass.
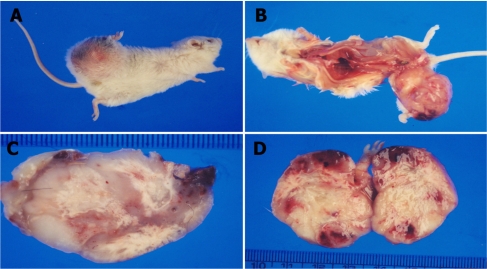
Figure 2
Histopathological findings of the masses of the right femur. (A) Poorly marginated tumor lesions with surrounding tissues. H&E stain. ×40. (B) Malignant tumor cells that originated from osteoblasts. H&E stain, ×100. (C) Poorly differentiated neoplastic mesenchymal cells forming streams, bundles, and nests and causing destruction of normal bone. H&E stain, ×400. (D) Osteoblasts and malignant neoplastic cells. H&E stain, ×400.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download