Abstract
This study was carried out to investigate an enhancing effect of black ginseng extract (BGE) on exercise capacity in an endurance exercising animal model. Fifty Sprague-Dawley rats were assigned to 5 experimental groups including non-training control, training control, and 3 treated groups (BGE at doses of 75, 150 and 300 mg/kg). The animals were treated with BGE for 6 weeks and their exercise ability in the maximal running distance test was determined using a treadmill every week. The blood lactic acid (LA) level and the activity of citrate synthase (CS) in the muscle were also measured after the exercise. The levels of glucose and glucose-6-phosphate (G-6-P) in the liver and muscle were determined using commercial assay kits. BGE treatments at the doses of 150 and 300 mg/kg significantly increased the exercise capacity compared with the non-training control or training control groups (P<0.05). The level of blood LA was decreased but the activity of CS was increased by the treatment of BGE at the dose of 300 mg/kg compared with the training control group. The level of G-6-P in the liver was elevated by the treatment of BGE at the dose of 300 mg/kg, compared to the training group. As compared with non-training control group, the treatments of BGE increased the levels of glucose and G-6-P in the liver and soleus muscle of rats. These results indicate that BGE have a potential for promoting exercise capacity by increasing CS activity in the muscle and decreasing LA in the serum of rats. These results also suggested that BGE can be used as a candidate supplement of health food products for promoting endurance exercise capacity in human athletes.
Go to : 
REFERENCES
Adams G.R.., Haddad F.., Baldwin K.M.1994. Interaction of chronic creatine depletion and muscle unloading: effects on postural locomotor muscles. J. Appl. Physiol. 77(3):1198–1205.

Antonio C.C.., Andrea C.P.., Gracia M.., Julio G.P.., Ana I.A.2001. Protective effects of Panax ginseng on muscle injury and inflammation after eccentric exercise. Comp. Biochem. Physiol. C Pharmacol. 130(3):369–377.
Baek N.I.., Kim D.S.., Lee Y.H.., Park J.D.., Lee C.B.., Kim S.I.1996. Ginsenoside Rh4, a genuine dammarane glycoside from Korean red ginseng. Planta Med. 62(1):86–87.

Carter S.L.., Rennie C.D.., Hamilton S.J.., Tarnopolsky . 2001. Changes in skeletal muscle in males and females following endurance training. Can. J. Physiol. Pharmacol. 79(5):386–392.

Coyle E.F.., Hagberg J.M.., Hurley B.F.., Martin W.H.., Ehsani A.A.., Holloszy J.O.1983. Carbohydrate feeding during prolonged strenous exercise can delay fatigue. J. Appl. Physiol. 55(1):230–235.
Ferrando A.., Vila L.., Cabral J.A.1999. Effects of a standaridized panax ginseng extract on the skeletal muscle of the rat: A comparative study in animals at rest and under exercise. Planta Med. 65(3):239–244.
Fu Y.., Ji L.L.2003. Chronic ginseng consumption attenuates age-associated oxidative stress in rats. J. Nutr. 133(11):3603–3609.

Garber K.2004. Energy boost: The Warburg effect returns in a new theory of cancer. J. Natl. Cancer Inst. 96(24):1805–1806.

Hargreaves M.1998. Skeletal muscle glucose metabolism during exercise: implications for health and performance. Aust. J. Sci. Med. Sport. 1(4):195–202.

Hargreaves M.., Costill D.L.., Coggan A.., Fink W.J.., Nishibata I.1984. Effect of carbohydrate feedings on muscle glycogen utilization and exercise performance. Med. Sci. Sports Exerc. 16(3):219–222.

Hatta T.., Koike M.., Langman P.1994. Laterality of mental imagery generation and operation: tests with brain-damaged patients and normal adults. J. Clin. Exp. Neuropsychol. 16(4):577–588.

Houmard J.A.., Weidner M.L.., Gavigan K.E.., Tyndall G.L.., Hickey M.S.., Alshami A.1998. Fiber type and citrate synthase activity in the human gastrocnemius and vastus lateralis with aging. J. Appl. Physiol. 85(4):1337–1341.
Hu F.., Manson J.., Stampfer M.., Graham C.2001. Diet, lifestyle, and the risk of type 2 diabetes mellitus in women. N. Engl. J. Med. 345(11):790–797.

Ivy J.L.., Costill D.L.., Fink W.J.., Lower R.W.1979. Influence of caffeine and carbohydrate feedings on endurance performance. Med. Sci. Sports. 11(1):6–11.
Jung K.., Kim I.H.., Han C.D.2004. Effect of medicinal plant extract on forced swimming capacity in mice. J. Ethnopharmacol. 93(4):75–81.
Kang K.S.., Kim H.Y.., Pyo J.S.., Yokozawa T.2006. Increase in the free radical scavenging activity of ginseng by heat-processing. Biol. Pharm. Bull. 29(4):750–754.

Kim W.Y.., Kim J.M.., Han S.B.., Lee S.K.., Kim N.D.., Park M.K.., Kim C.K.., Park J.H.2000. Steaming of ginseng at high temperature enhances biological activity. J. Nat. Prod. 63:1702–1704.

Kim S.N.., Ha Y.W.., Shin H.., Son S.H.., Wu S.J.., Kim Y.S.2007a. Simultaneous quantification of 14 ginsenosides in Panax ginseng C.A. meyer (Korean red ginseng) by HPLC-ELSD and its application to quality control. J. Pharm. Biomed. Anal. 45:164–170.
Kim K.T.., Yoo K.M.., Lee J.W.., Eom S.H.., Hwang I.K.., Lee C.Y.2007b. Protective effect of steamed american ginseng (Panax quinquefolius L.) on V79-4 cells induced by oxidative stress. J. Ethnopharmacol. 111(3):443–450.

Lee J.H.., Shen G.N.., Kim E.K.., Shin J.H.., Myung C.S.., Oh H.J.., Kim D.H.., Roh S.S.., Cho W.., Seo Y.B.., Park Y.J.., Kang C.W.., Song G.Y.2006. Preparation of black ginseng and its antitumor activity. Kor. J. Oriental Physiol. Pathol. 20(4):951–956.
Lee K.Y.., Lee Y.H.., Kim S.I.., Park J.H.., Lee S.K.1997. Ginsenoside-Rg5 suppresses cyclin E-dependent protein kinase activity via up-regulating p21Cip/WAF1 and down-regulating cyclin E in SK-HEP-1 cells. Anticancer Res. 17(2):1067–1072.
Lee S.R.., Kim M.R.., Yon J.M.., Baek I.J.., Park C.G.., Lee B.J.., Yun Y.W.., Nam S.Y.2009. Black ginseng inhibits ethanol-induced teratogenesis in cultured mouse embryos through its effects on antioxidant activity. In Vitro Toxicol. 23(1):47–52.

Leung K.W.., Yung K.K.., Mak N.K.., Chan Y.S.., Fan T.P.., Wong R.N.2007. Neuroprotective effects of ginsenoside-Rg1 in primary nigral neurons against rotenone toxicity. Neuropharmacology. 52(3):827–835.

Merry T.L.., McConell G.K.2009. Skeletal muscle glucose uptake during exercise: a focus on reactive oxygen species and nitric oxide signaling. IUBMB Life. 61(5):479–484.

Min Y.K.., Chung S.H.., Lee J.S.., Kim S.S.., Shin H.D.., Lim B.V.., Shin M.C.., Jang M.H.., Kim E.H.., Kim C.J.2002. Red ginseng inhibits exercise-induced increase in 5-hydroxytryptamine synthesis and tryptophan hydroxylase expression in dorsal raphe of rats. J. Pharmacol. Sci. 93(2):218–221.

Oh G.S.., Pae H.O.., Choi B.M.., Seo E.A.., Kim D.H.., Shin M.K.., Kim J.D.., Kim J.B.., Chung H.T.2004. 20(S)-Protopanaxatriol, one of ginsenoside metabolites, inhibits inducible nitric oxide synthase and cyclooxygenase-2 expressions through inactivation of nuclear factor-kappaB in RAW 264.7 macrophages stimulated with lipopolysaccharide. Cancer Lett. 205(1):23–29.
Ohira Y.., Kawano F.., Roy R.R.., Edgerton R.2003. Metabolic modulation of muscle fiber properties unrelated to mechanical stimuli. Jpn. J. Physiol. 53(6):389–400.

Park I.H.., Kim N.Y.., Han S.B.., Kim J.M.., Kwon S.W.., Kim H.J.., Park M.K.., Park J.H.2002. Three new dammarane glycosides from heat processed ginseng. Arch. Pharm. 25(4):428–432.

Patrick T.F.., Sami H.., Deanna P.B.., Carlo M.M.., Richard P.., Markku L.., David H.W.2003. Hexokinase II partial knockout impairs exercise-stimulated glucose uptake in oxidative muscles of mice. Am. J. Physiol. Endocrinol. Metab. 285(5):958–963.
Powers S.K.., Howely E.T.1997. Exercise physiology: Theory and application to fitness and performance. Clin. Exerc. Physiol. 7:624–648.
Roberto B.2001. Cardioprotective function of inducible nitric oxide synthase and role of nitric oxide in myocardial ischemia and preconditioning: an overview of a decade of research. J. Mol. Cell. Cardiol. 33(11):1897–1918.
Roger R.G.., Cristian F.., Mar G.R.., Josep M.F.., Juan C.F.., Joan J.G.2003. Glucose-6-phosphate produced by gluconeogenesis and by glucokinase is equally effective in activating hepatic glycogen synthase. J. Biol. Chem. 278(11):9740–9746.
Rose A.J.., Richter E.A.2005. Skeletal muscle glucose uptake during exercise: How is it regulated. Physiology. 20(4):260–270.

Spina R. J.., Chi M.M.., Hopkins M.G.., Nemeth P.M.., Lowry O.H.., Holloszy J.O.1996. Mitochondrial enzymes increase in muscle in response to 7-10 days of cycle exercise. J. Appl. Physiol. 80(6):2250–2254.

Stampfer M.J.., Hu F.B.., Manson J.E.., Rimm E.B.., Willett W.C.2000. Primary prevention of coronary heart disease in women through diet and lifestyle. N. Engl. J. Med. 343(1):16–22.

Steensberg A.., Hall G.V.., Keller G.., Osada T.., Schjerling P.., Klarlund B.., Pedersen B.K.., Saltin B.., Febbraio M.A.2002. Muscle glycogen content and glucose uptake during exercise in humans: influence of prior exercise and dietary manipulation. J. Physiol. 541(1):273–281.

Song G.Y.., Oh H.J.., Roh S.S.., Seo Y.B.., Park Y.J.., Myung C.S.2006. Effect of black ginseng on body weight and lipid profiles in male rats fed normal diets. Kor. J. Pharmacogn. 50(6):381–385.
Terblanche S.E.., Gohilb K.., Packerb L.., Hendersonc S.., Brooksc G.A.2001. The effects of endurance training and exhaustive exercise on mitochondrial enzymes in tissues of the rat. Comp. Biochem. Physiol. 128(4):889–896.
Voces J.., Alvarez A.I.., Vila L.., Ferrando A.., Cabral D.C.., Oliveira J.G.., Prieto. J.G.1999. Effects of administration of the standardized panax ginseng extract. Comp. Biochem. Physiol. 123(2):175–184.
Vogler B.K.., Pittler M.H.., Ernst E.1999. The efficacy of ginseng. A systematic review of randomised clinical trail. Eur. J. Cin. Phamacol. 55(8):567–575.
Voss B.., Mohr E.., Krzywanek H.2002. Effects of aqua-treadmill exercise on selected blood parameters and on heart-rate variability of horses. J. Vet. Med. A. Physiol. Pathol. Clin. Med. 49(3):137–143.

Yun T.K.2003. Experimental and epidemiological evidence on non-organ specific cancer preventive effect of Korean ginseng and identification of active compounds. Mutat. Res. 523-524. 63–74.
Wang L.C.., Lee T.F.1998. Effect of ginseng saponins on exercise performance in non-trained rats. Planta Med. 64(2):130–133.

Wargovich M.J.2001. Colon cancer chemoprevention with ginseng and other botanicals. J. Korean Med. Sci. 16 (suppl.). S81–86.
Go to : 
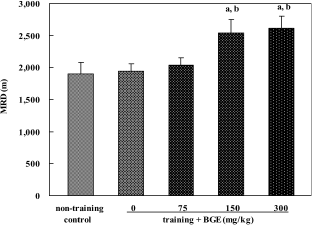 | Figure 1.Maximal running distance (MRD) in rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to rats for 6 weeks. Exercise capacity was assessed as running time and distance to exhaustion on a treadmill. Data were represented as mean± SEM (n=7-10). aSignificantly different from non-training control group at P<0.05. bSignificantly different from training control group at P<0.05. |
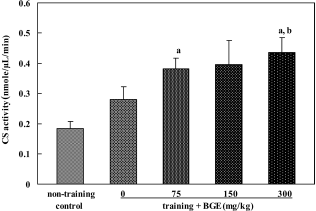 | Figure 2.Citrate synthase (CS) activity in the soleus muscle of rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. Citrate synthase in the soleus muscle was analyzed using an enzyme assay kit. Data were represented as mean± SEM (n=5). aSignificantly different from non-training control group at P<0.05. bSignificantly different from training control group at P<0.05. |
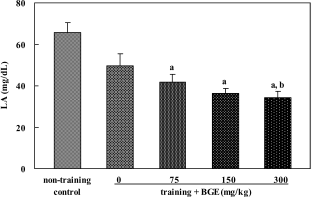 | Figure 3.Serum lactic acid (LA) level of rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. Lactic acid in serum was analyzed using an automatic lactic acid analyzer. Data were represented as mean±SEM (n=5-6). aSignificantly different from non-training control group at P<0.05. bSignificantly different from training control group at P<0.05. |
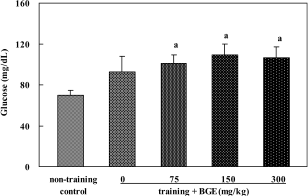 | Figure 4.Serum glucose level of rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. Glucose in blood was analyzed using a biochemical analyzer. Data were represented as mean±SEM (n=5-6). aSignificantly different from non-training control group at P<0.05. |
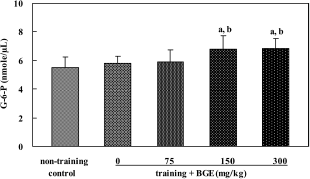 | Figure 5.Glucose-6-phosphate (G-6-P) level in the liver of rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. G-6-P in liver was analyzed with an enzyme assay kit. Data were represented as mean±SEM (n=5-6). aSignificantly different from non-training control group at P<0.05. bSignificantly different from training control group at P<0.05. |
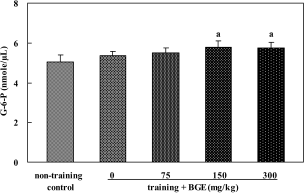 | Figure 6.Glucose-6-phosphate level in the soleus muscle of rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. G-6-P in muscle was analyzed with an enzyme assay kit. Data were represented as mean±SEM (n=6). aSignificantly different from non-training control group at P<0.05. |
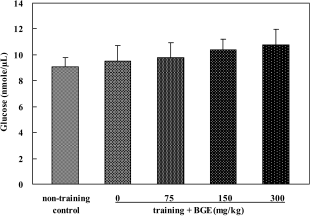 | Figure 7.Glucose level in the liver of rats treated with black ginseng extract (BGE). BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. Glucose concentrations in liver were analyzed using an glucose assay kit. Data were represented as mean±SEM (n=6). |
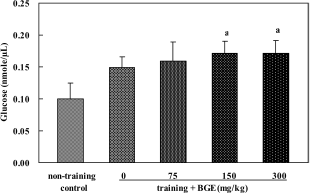 | Figure 8.Glucose level in the soleus muscle of rats treated with black ginseng extract (BGE) for 6 weeks. BGE at the oral doses of 75, 150, or 300 mg/kg was administered to animals for 6 weeks. Glucose concentrations in soleus muscle were analyzed using an glucose assay kit. Data were represented as aSignificantly different from non-training mean±SEM (n=6). control group at P<0.05. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download