Abstract
Purpose
Recurrent respiratory tract infection is a common manifestation of primary immunodeficiency disease, and respiratory viruses or bacteria are important triggers of asthma exacerbations. Asthma often coexists with humoral immunodeficiency in adults, and some asthmatics with immunoglobulin (Ig) G subclass deficiency (IgGSCD) suffer from recurrent exacerbations. Although some studies suggest a benefit from Ig replacement, others have failed to support its use. This study aimed to assess the effect of Ig replacement on asthma exacerbation caused by respiratory infection as well as the asthma control status of adult asthmatics with IgGSCD.
Methods
This is a multi-center, open-label study of adult asthmatics with IgGSCD. All patients received monthly intravenous immunoglobulin (IVIG) for 6 months and were evaluated regarding asthma exacerbation related to infection, asthma control status, quality of life, and lung function before and after IVIG infusion.
Results
A total of 30 patients were enrolled, and 24 completed the study. Most of the patients had a moderate degree of asthma severity with partly (52%) or uncontrolled (41%) status at baseline. IVIG significantly reduced the proportion of patients with asthma exacerbations, lowered the number of respiratory infections, and improved asthma control status, compared to the baseline values (P<0.001). The mean asthma-specific quality of life and asthma control test scores were improved significantly (P=0.009 and P=0.053, respectively); however, there were no significant changes in lung function.
Asthma affects nearly 250 million people worldwide, of whom approximately 20%–25% have moderate-to-severe uncontrolled disease.1 Despite the benefits of standard inhaled corticosteroid-based treatment, patients with inadequately controlled asthma remain at high risk of asthma exacerbations. Given the socioeconomic costs of asthma exacerbations, the identification of at-risk subjects is important.2
There are various precipitants of acute asthma exacerbation, such as viral infection, exercise, and exposure to allergens, occupational substances, drugs, or air pollutants.1 However, respiratory tract viruses, such as rhinovirus, are the most common triggers and can lead to bacterial infections.3 The host response to viral or bacterial infection is likely to influence susceptibility to asthma exacerbation. During exacerbation, airway inflammation is amplified by intrinsic host factors, such as bronchiectasis, diminished lung function, deficiency of interferon production by epithelial cells, and immunodeficiency.4
Most adult-onset primary immunodeficiency diseases (PID) are humoral immune deficiencies, such as common variable immunodeficiencies, hypogammaglobulinemia, immunoglobulin (Ig) G subclass deficiency (IgGSCD), and selective IgA deficiency.5 IgGSCD is common in adult asthmatic patients6 and associated with increased susceptibility to sinopulmonary infections.78 Such patients suffer from recurrent upper respiratory infections (rhinosinusitis and otitis media) as well as lower respiratory infections (bronchitis and pneumonia), which can cause structural changes in their airways. In fact, obstructive airway diseases, including asthma, bronchiolitis, and bronchiectasis, are common in patients with PID.910
Management of primary antibody deficiencies includes infection control and Ig replacement in selected cases.11 Although few reports have suggested the efficacy of Ig replacement therapy in patients with IgGSCD, intravenous immunoglobulin (IVIG) significantly improved quality of life, reduced the number of infections, and decreased the need for antibiotics and hospitalization.712 Several open trials suggested that Ig replacement had corticosteroid-sparing effects in severe asthmatics1314; however, randomized controlled studies failed to demonstrate the efficacy of Ig replacement in asthmatics.1516
Therefore, we hypothesized that IVIG would reduce the frequency of respiratory infections/asthma exacerbations and control asthma status in asthmatics associated with IgGSCD as well as with histories of recurrent upper and lower respiratory infections.
This is an open-label, single-arm, phase III multicenter clinical trial conducted at 4 sites between January 2013 and February 2016. The study was performed in accordance with the International Conference on Harmonization Good Clinical Practice (ICH GCP) and applicable legal requirements, and registered at ClinicalTrials.gov (NCT01992328). The study protocols and informed consent forms were reviewed and approved by the appropriate ethics committees. Written informed consent was obtained from all subjects and/or their legally authorized representatives prior to performing any study-related procedures.
The subjects ranged in age from 16 to 75 years and had been diagnosed with asthma more than 6 months before enrollment in the study based on clinical symptoms (such as cough, wheezing, breathlessness, chest tightness, and dyspnea), airway reversibility (defined as an increase of forced expiratory volume in 1 second (FEV1) >12% or 200 mL from pre-bronchodilator use), and airway hyperresponsiveness (PC20<16 mg/mL of methacholine). They met the criteria for IgGSCD of the International Union of Immunological Societies. IgGSCD was defined as a pre-IVIG level 2 standard deviations below the mean of that subclass on at least 2 separate occasions. IgG subclasses 1, 2, 3, and 4 were assayed by a turbidimetric enzyme immunoassay (Green Cross Corp, Yongin, South Korea). The normal ranges for IgG subclasses are: 382.4–928.6 mg/dL for IgG1, 241.8–700.3 mg/dL for IgG2, 21.8–176.1 mg/dL for IgG3, and 3.9–86.4 mg/dL for IgG4. All patients had a history of at least 2 episodes of steroid bursts or antibiotic therapies for asthma exacerbation due to respiratory infections in the previous year. In addition, patients were required to undergo normal complete blood count, routine chemistry, urinalysis, and electrocardiography at the time of screening. Exclusion criteria included treatment with IVIG 6 months before study entry, hypersensitivity to a component of IVIG, and concomitant use of drugs, such as systemic steroids or immunomodulatory agents which affect asthma control.
During the 6-month observation period, medical data, including infectious complications, were collected by the study physicians. After the observation period, patients received IVIG (400 mg/kg/month, Green Cross Corp) as an adjunct to their standard asthma therapy. The treatment comprised 6 infusions at monthly intervals, followed by a post-treatment observation period (Fig. 1). Pulmonary function test (PFT) results, asthma control test results (ACT), asthma-specific quality of life (AQOL), and safety variables were assessed every 4 weeks at scheduled clinic visits. Sputum and blood samples for the measurement of cytokines were obtained at baseline and week 21. The trough levels of IgG subclasses were measured at baseline, and at weeks 5 and 25.
The primary endpoint was the proportion of subjects experiencing asthma exacerbations related to respiratory infections during the treatment period compared to that in the prior 6 months. Asthma exacerbation was defined a condition in which: (1) a treating physician elected to administer systemic glucocorticoids (prednisolone 10 mg equivalent dose per day for at least 3 consecutive days) or (2) a patient was either hospitalized or visited an emergency department/a physician's office (unscheduled). Secondary endpoints included the change in FEV1 from baseline, use of systemic glucocorticoids, use of antibiotics and treatment duration, the number of acute exacerbations, AQOL and ACT scores, and serum levels of IgG subclasses and cytokines. Asthma severity at diagnosis and asthma control level at enrollment were assessed based on the Expert Panel Report 3 and the Global Initiative for Asthma (GINA) guidelines, respectively. Adverse events were assessed at each visit and collected its severity, duration, and causality assessment by the criteria of WHO-UMC (World Health Organization-The Uppsala Monitoring Centre).
Serum samples were obtained at visit 1 (pre-treatment) and visit 7 (post-treatment). Cytokines—interleukin 4, 5, 6, 8, 9, and 12p70, interferon gamma (IFN)-induced protein 10 (IP-10), and IFN-γ—were quantified using a Luminex bead-based multiplex assay (R&D Systems, Minneapolis, MN, USA).
A sample size of 21 had 80% power at a 5% level of significance to detect differences in the proportion of asthma exacerbations before and after treatment, based on previous data.17 To allow for the possibility of up to 30% of participants withdrawing early from the study, a recruitment target of 30 participants was set. The intention-to-treat (ITT) population comprised all subjects who received at least 1 IVIG infusion; the per-protocol (PP) population comprised the subjects in the ITT population without full protocol deviations.
Analysis of the primary efficacy—the reduction in the proportion of subjects in the ITT and PP populations experiencing asthma exacerbations related to respiratory infection before and after treatment—was performed by the one sample t test. The analysis of secondary outcomes was performed using Cochran's Q test for categorical variables and repeated ANOVA for continuous variables. Paired analysis was performed using the McNemar-Bowker test for categorical variables and the paired t test for continuous variables. Differences in serum cytokine levels before and after treatment were evaluated using Wilcoxon's signed rank test. Safety data are reported only as descriptive statistics.
All analyses were two-sided and performed at a 5% significance level. The results were analyzed using SPSS (ver. 22; SPSS Inc., Chicago, IL, USA) and R software (ver. 3.2.3.; R Development Core Team, Vienna, Austria).
In total, 30 patients were enrolled and 24 completed the study. The demographic and clinical features of the study subjects are listed in Table 1. There was a significant female predominance (female: male, 6.5:1.0), and the mean age at diagnosis was 54.5±13.4 years. The mean duration of asthma and PID was 10.4±10.3 years and 513.2±573.8 days, respectively. Most patients had moderate (45.0%) to severe (44.0%) asthma and were in a partly controlled (52.0%) or uncontrolled (41.0%) state. At baseline, the mean FEV1 of the study subjects was 73.4%±23.1%. Among the 4 IgG subtypes, deficiency in IgG3 was most common (n=23, 77%).
Ig replacement was associated with a reduction in the proportion of patients with asthma exacerbations caused by respiratory infections compared to the pre-treatment value (Table 2). The proportion of asthma exacerbation by respiratory infections was significantly reduced at each visit compared to baseline (Fig. 2). The overall asthma exacerbation rate, including both infections and noninfectious causes, was also reduced during the treatment versus pre-treatment period. The number of infectious complications was significantly reduced, from 2.46±1.96 during the previous 6 months to 1.04±1.46 during the treatment period (Table 3). We noted significant reductions in the frequency of asthma exacerbation events requiring unscheduled physician visits and hospital admission (P=0.029 and P=0.008, respectively).
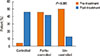
No significant changes in lung function were noted at any visit during the treatment period compared to baseline. However, there was a significant increase in the proportion of patients categorized as controlled during the study period compared to those partly or fully controlled at baseline (Fig. 3). In addition, IVIG treatment resulted in significant improvements in ACT and total AQOL scores compared to those at baseline (P=0.009 and P=0.053, respectively). Improvements in AQOL and ACT scores were observed as early as week 8 and sustained up to week 25 (P=0.009, and P=0.053, respectively) (Supplementary Figure and Table 3). Following the 6 months of IVIG, the serum level of IgG1 (P=0.023), IgG2 (P<0.001), and IgG3 (P=0.001) were significantly increased. However, IgG4 was not increased (P=0.479, Table 3). The total inflammatory cells in sputum comprised primarily neutrophils (65.8%±27.4%) rather than eosinophils (19.6%±25.7%) at baseline. IVIG was associated with a significant decrease in the number of neutrophils in sputum compared to baseline, but, there was no significant change in the number of eosinophils (Fig. 4). IL-4 and IL-9 levels were significantly lower at the end of the study compared to the baseline values (P=0.004, and P=0.033, respectively; Fig. 4 and Supplementary Table).
A total of 85 adverse events (AEs) in 23 patients were reported during the study period (Table 4). Six cases of serious AEs were reported: acute gastroenteritis, anorexia, asthma exacerbation, general weakness, herpes simplex infection, and rhinitis/sinusitis. However, none of the severe AEs were related to IVIG treatment according to the criteria of WHO-UMC causality assessment. Most of the AEs were mild (84.7%) or moderate (12.9%). Thirty-nine cases of acute asthma exacerbation in 13 patients were reported, 25 of which were related to respiratory infections; the remaining had non-infectious causes. IVIG-related adverse drug reactions were headache (n=5), skin rash (n=2), and myalgia (n=2); however, none of these patients discontinued treatment due to the reactions.
The results of this study showed that IVIG improved outcomes in patients with uncontrolled, moderate-to-severe asthma. They had a history of recurrent respiratory infections and had been diagnosed with IgGSCD. In the primary analysis, IVIG significantly reduced the proportion of patients with asthma exacerbations due to respiratory infections compared to baseline. During the 24-week treatment, the number of respiratory infections was significantly decreased, and the proportion of patients with controlled asthma status was increased. These results were achieved despite the continued use of previous asthma treatments throughout the study.
Most of the asthmatic patients have rare or intermittent exacerbations; however, there is evidence that a subset of asthmatics is ‘exacerbation-prone’18 Several epidemiologic studies have reported a strong correlation between FEV1 and the risk of asthma exacerbation.192021 Acute severe exacerbations in susceptible asthmatics activate pathways of inflammation and remodeling, resulting in the deterioration of lung function. Accelerated loss of lung function in turn increases the risk of exacerbation, resulting in a vicious cycle and the exacerbation-prone phenotype. Moreover, longitudinal studies of a PID cohort show that permanently diminished lung function due to recurrent respiratory infections leads to the exacerbation of underlying asthma or an increase in asthma symptoms.222324 In the present study, all subjects had histories of more than 2 recurrent respiratory infections per year, and their mean FEV1 was 73.4%. This suggests the presence of a subset of asthmatics with an exacerbation-prone phenotype as well as considerable overlap between asthma and PIDs.
Several open trials using high doses (1–2 g/kg) of monthly Ig replacement showed that IVIG had a systemic steroid-sparing effect in children and adults with steroid-dependent severe asthma.131425 However, randomized controlled studies failed to demonstrate the efficacy of IVIG in the same population, although the steroid-sparing effect was noted in a subgroup that required high daily doses of oral corticosteroids.151626 In contrast, IVIG has been shown to be effective and is used extensively in patients with primary humoral immunodeficiencies.27 Regarding the efficacy of IVIG in IgGSCD, several open-label studies have reported that IVIG significantly decreases the number of infections, the need for antibiotics, and hospitalization, and improves the quality of life in patients with recurrent infections.712 Therefore, we treated carefully selected groups of adult asthmatic patients with IgGSCD in the present study. All were ethnically Korean. We demonstrated that 400 mg/kg of monthly IVIG resulted in favorable clinical outcomes, such as improvement in infection-related asthma exacerbation, asthma control status, and quality of life.
Apart from its initial use as a supplementary therapy for primary humoral immune deficiencies, IVIG has also benefited patients with various autoimmune or allergic diseases due to its anti-inflammatory and immunomodulatory effects at high doses.28 Several mechanisms by which IVIG exerts its immune modulating effects on asthma have been suggested: it can directly act on T cells by enhancing the activity of CD25FoxP3+ Treg cells, leading to the production of greater amounts of transforming growth factor β and IL-10, and can inhibit IL-4 production in a murine model of allergic airway inflammation.293031 Levels of the Th2 cytokines IL-4 and IL-9 were significantly reduced after IVIG, whereas Th1 cytokine levels were not. The subjects also showed elevated numbers of neutrophils and eosinophils at baseline. Neutrophilia in airways is the consequence of viral or bacterial infections that can lead to acute exacerbations.32 The elevated sputum neutrophils were significantly decreased in patients whose asthma was controlled by treatment with IVIG. These data suggested that lower doses of IVIG might down-regulate IL-4 and IL-9 production, and that clinical improvements of IVIG could result from not only replacement of Ig inhibiting respiratory pathogens, but also anti-inflammatory effects in the airways of asthmatics. However, mechanisms by which IVIG inhibits Th2 cytokine production and airway inflammation are unclear.
This study has several limitations. It used an open-label, single-arm design having inherent weaknesses. There was no control group and the sample size was relatively small. To overcome these weaknesses, we evaluated the patients 6 months before and after treatment. Furthermore, to minimize the effect of potential confounders for primary and secondary endpoints, we enrolled the patients being well-compliant and receiving appropriate asthma treatments during the pre-IVIG treatment period; nonetheless, a monthly scheduled meeting with a physician may detect asthma exacerbation in advance and then prevent severe asthma exacerbation during the post-IVIG treatment period. Furthermore, a 6-month treatment period was relatively short to assess the effect of seasonal variations in asthma exacerbation. However, patient-centered outcomes, such as AQOL scores, were improved by IVIG treatment and no serious adverse reactions leading to discontinuation of IVIG were reported. In contrast to previous trials that focused on reducing systemic steroid use as a primary end point, the efficacy of IVIG as an add-on therapy against asthma exacerbations was demonstrated in our study.1526 These findings could facilitate the development of new therapeutic options for exacerbation-prone asthmatics with IgGSCD.
In conclusion, the addition of IVIG to standard therapy reduces asthma exacerbations and improves asthma control status as well as quality of life, especially for patients whose asthma is not controlled and who have IgGSCD, leading to recurrent respiratory infections. This study will help larger double-blind placebo-controlled trials define the role of IVIG in such patient populations. Further studies are needed to evaluate other outcomes, such as lung function, cost effectiveness, and the optimal dose of IVIG.
Figures and Tables
Fig. 2
Proportion of asthma exacerbations during pre- and post-IVIG treatment periods as well as at each visit.
Fig. 4
Sputum inflammatory cell numbers (A, B) and serum cytokine levels (C, D) before and after treatment with IVIG.

Table 1
Baseline characteristics of the study subjects

Data are presented as mean±SD, n (%), unless otherwise indicated.
*Severity was defined in Expert Panel Report 3; †Control status was defined according to Global Initiative for Asthma (GINA) guidelines.
ITT, intention to treat; PP, per protocol; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; FEV1, forced expiratory volume in 1 s; HTN, hypertension; PID, primary immunodeficiency disease.
Table 2
Reduction rate of asthma exacerbation caused by respiratory infections between pre- and post-treatment of IVIG for 6 months

| Mean±SD | Median (min to max) | P value | |
|---|---|---|---|
| In PP (n=24) | −0.31±1.14 | −0.83 (−1.00 to 3.00) | 0.193 |
| In ITT (n=30) | −0.45±1.05 | −1.00 (−1.00 to 3.00) | 0.026 |
Table 3
Primary and secondary efficacy outcomes

Table 4
Adverse events

ACKNOWLEDGMENTS
This study was supported by Green Cross Corp. and a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea (HI14C1061 and HI16C0992). This study was supported by ARO at Clinical Trial Center of Ajou University Hospital.
References
1. Dougherty RH, Fahy JV. Acute exacerbations of asthma: epidemiology, biology and the exacerbation-prone phenotype. Clin Exp Allergy. 2009; 39:193–202.
2. Fajt ML, Wenzel SE. Development of new therapies for severe asthma. Allergy Asthma Immunol Res. 2017; 9:3–14.
3. Busse WW, Lemanske RF Jr, Gern JE. Role of viral respiratory infections in asthma and asthma exacerbations. Lancet. 2010; 376:826–834.
4. Kurai D, Saraya T, Ishii H, Takizawa H. Virus-induced exacerbations in asthma and COPD. Front Microbiol. 2013; 4:293.
5. Azar AE, Ballas ZK. Evaluation of the adult with suspected immunodeficiency. Am J Med. 2007; 120:764–768.
6. Kim JH, Park HJ, Choi GS, Kim JE, Ye YM, Nahm DH, et al. Immunoglobulin G subclass deficiency is the major phenotype of primary immunodeficiency in a Korean adult cohort. J Korean Med Sci. 2010; 25:824–828.
7. Abdou NI, Greenwell CA, Mehta R, Narra M, Hester JD, Halsey JF. Efficacy of intravenous gammaglobulin for immunoglobulin G subclass and/or antibody deficiency in adults. Int Arch Allergy Immunol. 2009; 149:267–274.
8. Kim JH, Park S, Hwang YI, Jang SH, Jung KS, Sim YS, et al. Immunoglobulin g subclass deficiencies in adult patients with chronic airway diseases. J Korean Med Sci. 2016; 31:1560–1565.
9. Agondi RC, Barros MT, Rizzo LV, Kalil J, Giavina-Bianchi P. Allergic asthma in patients with common variable immunodeficiency. Allergy. 2010; 65:510–515.
10. Touw CM, van de Ven AA, de Jong PA, Terheggen-Lagro S, Beek E, Sanders EA, et al. Detection of pulmonary complications in common variable immunodeficiency. Pediatr Allergy Immunol. 2010; 21:793–805.
11. Ochs HD, Hagin D. Primary immunodeficiency disorders: general classification, new molecular insights, and practical approach to diagnosis and treatment. Ann Allergy Asthma Immunol. 2014; 112:489–495.
12. Abrahamian F, Agrawal S, Gupta S. Immunological and clinical profile of adult patients with selective immunoglobulin subclass deficiency: response to intravenous immunoglobulin therapy. Clin Exp Immunol. 2010; 159:344–350.
13. Jakobsson T, Croner S, Kjellman NI, Pettersson A, Vassella C, Björkstén B. Slight steroid-sparing effect of intravenous immunoglobulin in children and adolescents with moderately severe bronchial asthma. Allergy. 1994; 49:413–420.
14. Haque S, Boyce N, Thien FC, O’Hehir RE, Douglass J. Role of intravenous immunoglobulin in severe steroid-dependent asthma. Intern Med J. 2003; 33:341–344.
15. Kishiyama JL, Valacer D, Cunningham-Rundles C, Sperber K, Richmond GW, Abramson S, et al. A multicenter, randomized, double-blind, placebo-controlled trial of high-dose intravenous immunoglobulin for oral corticosteroid-dependent asthma. Clin Immunol. 1999; 91:126–133.
16. Salmun LM, Barlan I, Wolf HM, Eibl M, Twarog FJ, Geha RS, et al. Effect of intravenous immunoglobulin on steroid consumption in patients with severe asthma: a double-blind, placebo-controlled, randomized trial. J Allergy Clin Immunol. 1999; 103:810–815.
17. Bernatowska-Matuszkiewicz E, Pac M, Skopcynska H, Pum M, Eibl MM. Clinical efficacy of intravenous immunoglobulin in patients with severe inflammatory chest disease and IgG3 subclass deficiency. Clin Exp Immunol. 1991; 85:193–197.
18. Kupczyk M, ten Brinke A, Sterk PJ, Bel EH, Papi A, Chanez P, et al. Frequent exacerbators--a distinct phenotype of severe asthma. Clin Exp Allergy. 2014; 44:212–221.
19. Koga T, Oshita Y, Kamimura T, Koga H, Aizawa H. Characterisation of patients with frequent exacerbation of asthma. Respir Med. 2006; 100:273–278.
20. ten Brinke A, Sterk PJ, Masclee AA, Spinhoven P, Schmidt JT, Zwinderman AH, et al. Risk factors of frequent exacerbations in difficult-to-treat asthma. Eur Respir J. 2005; 26:812–818.
21. Kim HJ, Lee J, Kim JH, Park SY, Kwon HS, Kim TB, et al. Factors affecting recovery time of pulmonary function in hospitalized patients with acute asthma exacerbations. Allergy Asthma Immunol Res. 2016; 8:499–504.
22. Chen Y, Stirling RG, Paul E, Hore-Lacy F, Thompson BR, Douglass JA. Longitudinal decline in lung function in patients with primary immunoglobulin deficiencies. J Allergy Clin Immunol. 2011; 127:1414–1417.
23. Goldstein MF, Hilditch GJ, Dvorin DJ, Belecanech GA. Immunoglobulin replacement for selective IgM immunodeficiency, bronchiectasis, and asthma. Ann Allergy Asthma Immunol. 2016; 116:172–173.
24. Verma N, Grimbacher B, Hurst JR. Lung disease in primary antibody deficiency. Lancet Respir Med. 2015; 3:651–660.
25. Landwehr LP, Jeppson JD, Katlan MG, Esterl B, McCormick D, Hamilos DL, et al. Benefits of high-dose i.v. immunoglobulin in patients with severe steroid-dependent asthma. Chest. 1998; 114:1349–1356.
26. Niggemann B, Leupold W, Schuster A, Schuster R, v Berg A, Grübl A, et al. Prospective, double-blind, placebo-controlled, multicentre study on the effect of high-dose, intravenous immunoglobulin in children and adolescents with severe bronchial asthma. Clin Exp Allergy. 1998; 28:205–210.
27. Yong PL, Boyle J, Ballow M, Boyle M, Berger M, Bleesing J, et al. Use of intravenous immunoglobulin and adjunctive therapies in the treatment of primary immunodeficiencies: a working group report of and study by the Primary Immunodeficiency Committee of the American Academy of Allergy Asthma and Immunology. Clin Immunol. 2010; 135:255–263.
28. Ballow M. The IgG molecule as a biological immune response modifier: mechanisms of action of intravenous immune serum globulin in autoimmune and inflammatory disorders. J Allergy Clin Immunol. 2011; 127:315–323.
29. Yamamoto M, Kobayashi K, Ishikawa Y, Nakata K, Funada Y, Kotani Y, et al. The inhibitory effects of intravenous administration of rabbit immunoglobulin G on airway inflammation are dependent upon Fcγ receptor IIb on CD11c(+) dendritic cells in a murine model. Clin Exp Immunol. 2010; 162:315–324.
30. Massoud AH, Guay J, Shalaby KH, Bjur E, Ablona A, Chan D, et al. Intravenous immunoglobulin attenuates airway inflammation through induction of forkhead box protein 3-positive regulatory T cells. J Allergy Clin Immunol. 2012; 129:1656–1665.e3.
31. Massoud AH, Yona M, Xue D, Chouiali F, Alturaihi H, Ablona A, et al. Dendritic cell immunoreceptor: a novel receptor for intravenous immunoglobulin mediates induction of regulatory T cells. J Allergy Clin Immunol. 2014; 133:853–863.e5.
32. Gern JE. How rhinovirus infections cause exacerbations of asthma. Clin Exp Allergy. 2015; 45:32–42.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download