Abstract
Despite a range of efficacious therapies for asthma, including inhaled corticosteroids (ICS) and long-acting β2-agonists (LABA), a significant proportion of patients have poor asthma control and retain a risk of future worsening of their symptoms. Long-acting muscarinic antagonist (LAMA) bronchodilators offer a well-tolerated, efficacious, and cost-effective add-on to a patient's treatment. Of the LAMAs currently under investigation or available for the treatment of asthma, evidence from a comprehensive clinical trial program in adults and children shows that once-daily treatment with tiotropium provides benefits for patients with uncontrolled asthma despite the use of ICS and LABAs. Tiotropium is included in the Global Initiative for Asthma (GINA) strategy document as an add-on therapy option for patients at Step 4 or 5 with a history of asthma exacerbations. Tiotropium Respimat® has demonstrated safety and efficacy in patients with a range of disease severities, ages, and phenotypes. This review describes the evidence for the use of LAMA as add-on therapy for patients with asthma who remain uncontrolled despite the use of ICS and LABA treatments.
An estimated 235-334 million people worldwide suffer from asthma,12 with an estimated 14% of the world's children experiencing asthma symptoms. Poor asthma control is associated with decreased quality of life, more frequent use of rescue medications, increased healthcare utilization, and ultimately death.3 The reasons for poor control include misdiagnosis, non-adherence and non-compliance to treatment, individual variations in response to treatment, poor inhaler techniques, and patients' treatment expectations.4 The majority of patients state that they are dissatisfied with their current treatment and believe that there is a need for new therapies.4 It has been estimated that in the United States approximately 15,000 people die each year from asthma exacerbations,5 with approximately 500,000 hospitalizations from “acute asthma” each year.5 Hospitalizations and associated costs are also high in Asia; in the Asia-Pacific Asthma Insight Management (AP-AIM) survey, 54% of patients with asthma reported having been hospitalized overnight 2 or more times. Substantial variations were observed between countries, where unscheduled urgent and emergency care ranged from 46% in Taiwan, 42% in India, and 40% in Malaysia to 15% in both Hong Kong and Australia, and 16% in South Korea.6 This highlights the discrepancy between accepted goals for asthma management and the actual degree of asthma control in the Asia-Pacific region.7 The burden of asthma (per-patient costs) as a percentage of healthcare costs or per capita gross domestic product (GDP) was also high for the Asthma Insights and Reality in Asia-Pacific (AIRIAP) countries (300% of per capita healthcare costs and 13% of per capita GDP overall).8
Asthma management is a continuous cycle of assessing symptoms, adjusting treatment, and reviewing the response. The Global Initiative for Asthma (GINA) definition of asthma control has 2 domains:9 1) symptom control—current impairment or day-to-day asthma control (absence of symptoms, minimal reliever use, normal activity levels, and lung function); and 2) control of future risk (absence of exacerbations, prevention of decline in lung function, and absence of side effects from drugs).10 This can be achieved by assessing symptom control over the last 4 weeks and assessing risk factors for poor outcomes, including low lung function.9 The proportion of patients who achieve control remains low, with control rates of around 50% in real-life settings and up to 70% in randomized controlled trials.1112 Factors underlying the poor levels of control include the heterogeneity in the clinical spectrum and pathophysiological features of asthma, in addition to the presence of comorbidities and lack of treatment adherence.413
The Asthma Control Test (ACT) questionnaire is widely used in asthma clinics to assess patients' subjective perception of their symptoms 4 weeks prior to the clinic visit. This test involves patients' self-assessment of their symptoms for each item (frequency of dyspnea, use of rescue medications, effect of asthma on daily functioning, frequency of night-time symptoms, and overall assessment of asthma control), using a 5-point scale for each item, giving a total score ranging from 0 to 25, with a higher score indicating well-controlled status. The scores resulting from these tests do not always correlate with lung function parameters such as forced expiratory volume in 1 second (FEV1), leading to confusion in assessing asthma control status. A study by Park et al.14 retrospectively reviewed ACT questionnaire results, baseline lung function, and changes in FEV1; it was concluded that physicians should consider other factors, such as patient age, gender, blood eosinophil levels, and comorbidities along with ACT score and lung function in the assessment of asthma control.
Uncontrolled asthma has a negative impact on health status and work productivity,15 and is associated with increased healthcare costs.16 In Europe, the mean total cost per patient has been estimated at €2,281 in patients with uncontrolled persistent asthma, compared with €509 in patients with controlled persistent asthma (based on 2010 values).16 Despite this, many patients overestimate their level of control: in a survey of asthma control and management in 8,000 European patients (REcognise Asthma and LInk to Symptoms and Experience [REALISE]),11 levels of asthma control were low, with 45% of respondents having uncontrolled asthma (as defined by GINA guidelines9). However, many patients (>80%) regarded their asthma as controlled and not serious, despite experiencing symptoms and exacerbations.11 The survey was extended to include 2,467 patients17 and 375 physicians18 from 8 countries in Asia (REALISE Asia). Similarly, while approximately 90% of these patients considered their asthma to be under control, less than 20% actually achieved controlled asthma, with nearly 50% having uncontrolled asthma by the GINA-defined criteria.9 In the AP-AIM study, only 2% were considered controlled across the entire region when compared with the GINA guidelines. Overall, 59.4% of all respondents reported daytime symptoms; of these, 23% reported having daytime symptoms either every day or most days. Daytime shortness of breath and chest tightness was considered the most bothersome symptom by over 60% of respondents. 45.2% of respondents reported nighttime symptoms; of these, 44% reported symptoms at least once or twice per week.6
Despite this increasing need for effective therapeutic intervention in asthma, there have been no new treatments for asthma in the non-severe population until recently. Tiotropium is an effective long-acting muscarinic antagonist (LAMA) bronchodilator that has been used in the management of chronic obstructive pulmonary disease (COPD) for over 10 years. Tiotropium is now also indicated for use in asthma in several regions. This review discusses the position of LAMAs in the treatment of asthma, and provides an overview of the recent clinical trial data of tiotropium in both adult and pediatric patient populations.
Low-dose inhaled corticosteroids (ICS) and ICS/long-acting β2-agonist (LABA) are the mainstays of asthma treatment for patients in steps 1-3 of the GINA guidelines, with as-needed short-acting β2-agonist (SABA) or low-dose ICS/formoterol employed as reliever medications. The LAMA tiotropium is included in the guidelines as an add-on therapy option at step 4 or 5 in patients with a history of asthma exacerbations.9 Tiotropium Respimat® is approved in adult patients in Singapore (aged 18 years or over), Japan (aged 15 years and over), and the EU (aged 18 years or over), and in patients aged ≥6 years in the US. Other options at step 4 include the combination of high-dose ICS with LABA, or medium- or high-dose ICS with the addition of a leukotriene modifier receptor antagonist (LTRA) or low-dose sustained-release theophylline. At step 5, options include anti-immunoglobulin E (IgE) (omalizumab) and anti-interleukin-5 (IL-5) (mepolizumab) in patients with severe asthma, or the addition of low-dose oral corticosteroids, among others.9
Cholinergic parasympathetic nerves provide dominant innervation to the lungs.19 Release of acetylcholine from these nerves regulates airway tone, vasodilation, and cholinergic activity, and is the predominant driver of bronchial smooth muscle contraction.2021 Patients with asthma may have increased release of acetylcholine from cholinergic nerve endings,22 leading to bronchoconstriction. In addition, it has been hypothesized that local airway inflammatory mediators, along with the mechanisms listed above, may be driving an increase in cholinergic tone. Other reasons suggesting that LAMAs may be beneficial for the control of asthma include: 1) cholinergic antagonists may have non-neuronal anti-inflammatory actions;23 2) patients with asthma may have abnormal muscarinic (M) receptor expression; and 3) increased cholinergic and smooth muscle tone may contribute to airway hyper-responsiveness (Figure).21242526
Tiotropium has been approved for use in COPD for over 10 years, and its mechanism of action as a bronchodilator has been studied extensively in this disease. In the lungs, release of acetylcholine from parasympathetic nerves regulates airway tone, airway smooth muscle contraction, mucus secretion, and vasodilation via interactions with M-receptors on the airway smooth muscle, glands, and the pulmonary vasculature.27 Blockade of M1 and M3 anticholinergic receptors reduce smooth muscle tone and cause bronchodilation, but inhibition of the M2-receptor has the opposite effect.28 Ipratropium nonselectively blocks all three receptors whereas tiotropium dissociates much more slowly from the M1 and M3 receptors, making it a more potent bronchodilator.29 Importantly, single-dose studies showed that tiotropium has a long-lasting effect,29 and a wealth of clinical data in COPD support its 24-hour profile.30 There have been observations of anti-inflammatory effects of anticholinergics in in vitro and in vivo studies using experimental models.3132 Combination therapy with anticholinergics and corticosteroids might have additive protective effects on airway inflammation. This effect has been investigated in guinea pig models. It was found that combined treatment with tiotropium and ciclesonide did not inhibit acute allergen-induced inflammation, but did inhibit chronic allergen-induced airway inflammation and remodeling.33
As acetylcholine has a key role in the pathophysiology of asthma, there is a clear rationale for the development of anticholinergic bronchodilators as therapies in this disease as well as in COPD. In an early investigation of the duration of protection from a single dose of inhaled tiotropium against methacholine-induced bronchoconstriction in male volunteers with atopic asthma, tiotropium produced a dose-dependent mild bronchodilation as measured by an increase in FEV1 that was sustained for 24 hours. This prolonged bronchodilator response and protection against methacholine challenge suggested that tiotropium would be useful in the treatment of asthma, and that once-daily dosing may be sufficient.34
Aclidinium bromide is currently being investigated in COPD and experimental asthma.35 However, there are currently no clinical trials investigating its effect in asthma.
There are several studies completed or currently ongoing with the LAMAs umeclidinium and glycopyrronium (Table 1). Umeclidinium is a quinuclidine derivative and potent anticholinergic with slow functional reversibility at the human M3 receptor.36 The dose response, efficacy, and safety of once-daily umeclidinium bromide combined with fluticasone furoate were investigated in adults with asthma who were symptomatic despite maintenance ICS treatment. Clinically meaningful and statistically significant increases in trough FEV1 were found with the combination treatment, but not with fluticasone furoate alone.36 In addition, monotherapy with umeclidinium resulted in modest improvements in trough FEV1 that were not dose-related or consistent in magnitude, and therefore did not conclusively support a therapeutic benefit of monotherapy with umeclidinium in patients with asthma not requiring ICS treatment.37
Glycopyrronium has been investigated in a study comparing the effects of single doses of tiotropium and glycopyrronium against methacholine-induced bronchoconstriction. Tiotropium provided at least 7 days of bronchoprotection against methacholine, whilst glycopyrronium provided up to 7 days of bronchoprotection against methacholine. Tiotropium provided statistically superior bronchoprotection at 24 and 72 hours compared to glycopyrronium.38
The efficacy and safety of tiotropium have been investigated in a number of phase II and III clinical trials in patients with symptomatic asthma and in a large-scale clinical trial program comprising 18 randomized, double-blind, placebo-controlled studies in >6,000 patients with asthma who continued to be symptomatic despite receiving ICS maintenance therapy with or without other maintenance therapies. The phase II clinical trial program included 5 trials investigating tiotropium at various doses (10, 5, 2.5, and 1.25 µg) vs placebo as add-on to ICS with or without LABA in adult patients with moderate to severe symptomatic asthma.3139404142 These studies showed that tiotropium consistently improved lung function as assessed by improvements in peak FEV1 measured within 3 hours of dosing (FEV1(0-3h)). The safety profile of tiotropium across the phase II studies was established and balanced across treatment groups except for increased systemic anticholinergic effects (such as dry mouth) in the 10 µg dosage group. Phase II, randomized, dose-ranging studies of tiotropium at 5 µg, 2.5 µg, and 1.25 µg in children (aged 6-11 years)43 and adolescents (aged 12-17 years)44 with symptomatic asthma despite maintenance treatment with ICS have demonstrated the efficacy and safety of tiotropium in younger patients, suggesting the potential indication of tiotropium across all age groups. In terms of phase III trials, 6 trials have been conducted to assess the efficacy and safety of once-daily tiotropium 5 µg vs placebo in adult patients with mild, moderate, or severe symptomatic asthma as an addon therapy to ICS (low to high dose) with or without LABA (Table 2). Four of the studies also included the 2.5 µg dose. Both doses of tiotropium had a significantly greater improvement in FEV1(0-3h) at 24 weeks compared with placebo. Five phase III studies have been conducted to assess the efficacy and safety of once-daily tiotropium at 2.5 and 5 µg vs placebo in children with moderate or severe symptomatic asthma as an add-on therapy to ICS (low to high dose) with or without another controller (Table 3). Both doses of tiotropium had a significantly greater improvement in FEV1(0-3h) at 24 weeks compared with placebo (data not shown).
One phase III study examined tiotropium as an add-on to low-dose ICS, in line with step 2 of the GINA guidelines where low-dose ICS is the preferred controller choice.45 Another study examined the effect of tiotropium when added on to at least ICS, complementing step 3 of the guidelines, where low-dose ICS/LABA is the preferred choice of controller therapy.46 In a further study, tiotropium was added on to at least ICS/LABA, which corresponds with step 4, where medium-dose ICS and high-dose LABA are recommended as the preferred controller therapy.47
Asthma is a heterogeneous disease, and different asthma phenotypes and endotypes exist, which may respond differently to targeted therapies.48 It has long been known that patients with asthma with higher levels of eosinophils in their sputum respond to systemic corticosteroids and that patients with lower levels of eosinophils do not. Severe asthma is defined by the European Respiratory Society and American Thoracic Society (ERS/ATS) as “asthma that requires treatment with high-dose ICS plus a second controller and/or systemic corticosteroids to prevent it from becoming uncontrolled or that remains uncontrolled despite this therapy”49 As 5%-10% of patients with asthma can be classified as severe, many new approaches to the treatment of severe asthma have been attempted, including targeting components of the immune response pathway.48 Tiotropium has been shown to be efficacious across asthma severities, but it is nevertheless important to determine whether there is a clinical phenotype or characteristic that would predict whether certain patients would respond favorably with the addition of this treatment. A recent study investigated the effectiveness of once-daily tiotropium Respimat® as an add-on to ICS/LABA in patients with severe symptomatic asthma. This study revealed that tiotropium 5 µg improved lung function, reduced the risk of asthma exacerbations (time to first severe exacerbation), and improved asthma symptom control irrespective of IgE levels, eosinophils counts, age, gender, or baseline demographics compared with placebo.50 Comparisons of adjusted mean difference (mL) made in patients stratified into 2 groups with serum IgE levels of ≤430 µg/L and >430 µg/L, and blood eosinophils of ≤0.6×109/L and >0.6×109/L showed no interaction between risk of exacerbations or asthma worsening. These results suggest that an important clinical advantage of tiotropium is that patient phenotyping is not necessary. However, further studies are needed.
Although the prevalence of asthma in the elderly population is similar to that in the adult population (approximately 5%-10%) the severity and mortality of asthma are reported to increase with age.1 The prevalence of asthma in South Korea is higher in elderly than in non-elderly patients.51 Many questions remain unanswered in elderly patients, as this population is partially excluded from randomized clinical trials due to age criteria and comorbidities. A recent study examined the predicators of asthma control in elderly patients with asthma in South Korea.5253 This study showed that the number of medications for comorbidities, as well as functional status, determined by the higher physical functioning scale, are important parameters in assessing and predicting asthma control.53 A large number of medications were associated with unfavorable outcomes, and it is recommended that treatment regimens for the elderly should be as simple as possible for elderly asthmatic patients. As tiotropium has been shown to be efficacious and have a favorable safety profile in COPD patients who are generally older, it offers a good option for the treatment of elderly asthma patients, especially for those with comorbid conditions such as cardiovascular diseases.
Using data from the replicate phase III trial in patients with moderate asthma,47 Willson and colleagues54 created a Markov model to estimate the cost-effectiveness of tiotropium from the perspective of the UK National Health Service. The analysis concluded that in adult patients with severe asthma, tiotropium provided a cost-effective treatment option if used in combination with high-dose ICS/LABA therapy. Add-on tiotropium generated an incremental 0.24 in quality-adjusted life years and £5,238 costs over a lifetime horizon, resulting in an incremental cost-effectiveness ratio of £21,906 per QALY gained (based on 2012 pricing). The cost-effectiveness of treatment of uncontrolled severe asthma with tiotropium added to standard of care has been confirmed by regional studies from Spain,55 Poland,56 and Portugal.57 Additional studies to further clarify the cost-effectiveness of tiotropium in asthma are needed, particularly in relation to novel biologic treatments such as omalizumab and mepolizumab.
The use of LAMA in large-scale trials has demonstrated clinical benefits and improvements in lung function where tiotropium is used as add-on therapy to ICS alone, or plus an LABA in adult and pediatric patients with poorly controlled asthma by ICS with or without other controllers. Tiotropium is also a useful add-on therapy for patients at risk of future asthma worsening or exacerbations.
Figures and Tables
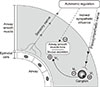 | FigureThe role of cholinergic activity in the pathophysiology of asthma. Autonomic regulation of airway smooth muscle tone. M1, M2, M3 represent muscarinic receptors 1, 2, and 3. + and − symbols represent signals increasing and decreasing airway smooth muscle tone, respectively. Reproduced with permission from Price et al.21 M, muscarinic. |
Table 1
Clinical trials with LAMAs other than tiotropium

| Patients | Treatment duration | Baseline therapy | No. (enrolled) | Study drugs |
|---|---|---|---|---|
| Symptomatic inadequately controlled58 | Variable 24–52 wk | ICS/LABA | 2,250 | Umeclidinium bromide 31.25 and 62.5 µg |
| Symptomatic59 | 14 wk | Non-ICS controller | 350 | Umeclidinium bromide 15.6, 31.25, 62.5, 125, and 250 μg |
| Symptomatic36,59 | 14 wk | At least ICS/LABA | 421 | Umeclidinium bromide 15.6, 31.25, 62.5, 125, and 250 µg |
| Symptomatic60 | 14 days | ICS or non-ICS maintenance | 249 | Glycopyrronium MDI 1.9, 3.6, 7.2, 14.4, and 28.8 µg |
| Uncontrolled61 | 6 wk | ICS | 98 | Glycopyrrolate bromide 12.5 µg |
| Symptomatic38 | 96 hr | NS | 13 | Glycopyrronium 50 µg |
| Symptomatic mild-to-moderate62 | 24–48 hr | NS | 30 | Glycopyrronium 50 µg |
Table 2
Phase III studies with tiotropium in adults

| Patients | Treatment duration (wk) | Baseline therapy | No. (treatment group) | Study drugs | Primary and key secondary endpoints | Difference from placebo | |
|---|---|---|---|---|---|---|---|
| Trial 1 | Trial 2 | ||||||
| Poorly controlled47 | 48 | At least ICS/LABA | 456 |
Tiotropium Respimat 5 µg |
Peak FEV1 (Week 24) | 86 (20–152) mL (P=0.010) | 154 (91–217) mL (P<0.001) |
| Trough FEV1 (Week 24) | 88 (27–149) mL (P=0.010) | 111 (53–169) mL (P<0.001) | |||||
| Time to first severe exacerbation | 21% reduction in risk (HR=0.79; 95% CI=0.62–1.00; P=0.030) | ||||||
| ACQ-7 (adjusted mean score) | NS | −0.2 (P=0.003) | |||||
| Symptomatic63 | 52 | At least ICS±LABA | 114 |
Tiotropium Respimat 5 µg* |
Trough FEV1 (Week 52) | 112 mL (95% CI=18-207; P=0.020) | |
| Trough FVC (Week 52) | NS | ||||||
| ACQ-7 responder rate (Week 52) | 76.3% tiotropium vs 73.2% placebo | ||||||
| Moderate symptomatic46 | 24 | At least ICS |
519 tiotropium 5 µg |
Tiotropium Respimat 5 µg* |
Peak FEV1 | 198 (142–253) mL (P<0.0001) | 169 (116–222) mL (P<0.0001) |
| Peak FVC | 102 (42–162) mL (P<0.0008) | 89 (30–147) mL (P=0.0031) | |||||
| ACQ-7 (adjusted mean score) | −0.12 (SD=0.04; P=0.0084) | ||||||
| Symptomatic45 | 12 | Low-dose ICS | 155 |
Tiotropium Respimat 5 µg* |
Peak FEV1 (Week 12) | 128 mL (95% CI=57-199; P<0.001) | |
| Trough FEV1 (Week 12) | 122 mL (95% CI=49-194; P<0.001) | ||||||
| ACQ-7 total score (Week 12) | 0.014 (95% CI=−118–0.146; P=0.830) | ||||||
ACQ-7, Asthma Control Questionnaire-7; CI, confidence intervals; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; HR, hazard ratio; ICS, inhaled corticosteroids; LABA, long-acting β2-agonist; NS, not significant; SD, standard deviation; wk, week.
*2.5 µg tiotropium Respimat also included in study but not reported here.
Table 3
Phase III studies with tiotropium in children

| Patients | Treatment duration (wk) | Baseline therapy | No. (treatment group) | Study drugs |
|---|---|---|---|---|
| Moderate persistent 12- to 17-year-olds64 | 48 | At least ICS | 259 | Tiotropium respimat 2.5 and 5 µg |
| Severe persistent 12- to 17-year-olds65 | 12 | ICS+≥1 controller | 257 | Tiotropium respimat 2.5 and 5 µg |
| Moderate persistent 6- to 11-year-olds66 | 48 | At least ICS | 270 | Tiotropium respimat 2.5 and 5 µg |
| Severe persistent 6- to 11-year-olds67 | 12 | ICS+≥1 controller | 262 | Tiotropium respimat 2.5 and 5 µg |
| Persistent 1- to 5-year-olds68 | 12 | At least ICS | 67 | Tiotropium respimat 2.5 and 5 µg |
ACKNOWLEDGMENTS
We thank Dr Manda Gent from MediTech Media for medical writing assistance, funded by Boehringer Ingelheim. This study is supported by a grant of the Korea Health Technology R&D Project, through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, ROK (grant number: HI16C0992).
References
1. Global Asthma Network (NZ). The global asthma report: 2014. Auckland: Global Asthma Network;2014.
2. World Health Organization. Asthma: fact sheet no.307. Geneva: World Health Organization;2013.
3. Braman SS. The global burden of asthma. Chest. 2006; 130:4S–12S.
4. Haughney J, Price D, Kaplan A, Chrystyn H, Horne R, May N, et al. Achieving asthma control in practice: understanding the reasons for poor control. Respir Med. 2008; 102:1681–1693.
5. Dougherty RH, Fahy JV. Acute exacerbations of asthma: epidemiology, biology and the exacerbation-prone phenotype. Clin Exp Allergy. 2009; 39:193–202.
6. Thompson PJ, Salvi S, Lin J, Cho YJ, Eng P, Abdul Manap R, et al. Insights, attitudes and perceptions about asthma and its treatment: findings from a multinational survey of patients from 8 Asia-Pacific countries and Hong Kong. Respirology. 2013; 18:957–967.
7. Lai CK, De Guia TS, Kim YY, Kuo SH, Mukhopadhyay A, Soriano JB, et al. Asthma control in the Asia-Pacific region: the Asthma Insights and Reality in Asia-Pacific Study. J Allergy Clin Immunol. 2003; 111:263–268.
8. Lai CK, Kim YY, Kuo SH, Spencer M, Williams AE. Cost of asthma in the Asia-Pacific region. Eur Respir Rev. 2006; 15:10–16.
9. Global Initiative for Asthma. 2016 GINA report, global strategy for asthma management and prevention. [place unknown]: Global Initiative for Asthma;2016.
10. Bateman ED, Harrison TW, Quirce S, Reddel HK, Buhl R, Humbert M, et al. Overall asthma control achieved with budesonide/formoterol maintenance and reliever therapy for patients on different treatment steps. Respir Res. 2011; 12:38.
11. Price D, Fletcher M, van der Molen T. Asthma control and management in 8,000 European patients: the REcognise Asthma and LInk to Symptoms and Experience (REALISE) survey. NPJ Prim Care Respir Med. 2014; 24:14009.
12. Bateman ED, Boushey HA, Bousquet J, Busse WW, Clark TJ, Pauwels RA, et al. Can guideline-defined asthma control be achieved? The Gaining Optimal Asthma ControL study. Am J Respir Crit Care Med. 2004; 170:836–844.
13. Peters SP, Ferguson G, Deniz Y, Reisner C. Uncontrolled asthma: a review of the prevalence, disease burden and options for treatment. Respir Med. 2006; 100:1139–1151.
14. Park SY, Yoon SY, Shin B, Kwon HS, Kim TB, Moon HB, et al. Clinical factors affecting discrepant correlation between asthma control test score and pulmonary function. Allergy Asthma Immunol Res. 2015; 7:83–87.
15. Demoly P, Annunziata K, Gubba E, Adamek L. Repeated cross-sectional survey of patient-reported asthma control in Europe in the past 5 years. Eur Respir Rev. 2012; 21:66–74.
16. Accordini S, Corsico AG, Braggion M, Gerbase MW, Gislason D, Gulsvik A, et al. The cost of persistent asthma in Europe: an international population-based study in adults. Int Arch Allergy Immunol. 2013; 160:93–101.
17. Price D, David-Wang A, Cho SH, Ho JC, Jeong JW, Liam CK, et al. Time for a new language for asthma control: results from REALISE Asia. J Asthma Allergy. 2015; 8:93–103.
18. Price D, David-Wang A, Cho SH, Ho JC, Jeong JW, Liam CK, et al. Asthma in Asia: physician perspectives on control, inhaler use and patient communications. J Asthma. 2016; 53:761–769.
19. Kolahian S, Gosens R. Cholinergic regulation of airway inflammation and remodelling. J Allergy (Cairo). 2012; 2012:681258.
20. Barnes PJ. Neural mechanisms in asthma. Br Med Bull. 1992; 48:149–168.
21. Price D, Fromer L, Kaplan A, van der Molen T, Román-Rodríguez M. Is there a rationale and role for long-acting anticholinergic bronchodilators in asthma? NPJ Prim Care Respir Med. 2014; 24:14023.
22. Hashimoto A, Maeda H, Yokoyama M. Augmentation of parasympathetic nerve function in patients with extrinsic bronchial asthma--evaluation by coefficiency of variance of R-R interval with modified long-term ECG monitoring system. Kobe J Med Sci. 1996; 42:347–359.
23. Gosens R, Bos IS, Zaagsma J, Meurs H. Protective effects of tiotropium bromide in the progression of airway smooth muscle remodeling. Am J Respir Crit Care Med. 2005; 171:1096–1102.
24. Bos IS, Gosens R, Zuidhof AB, Schaafsma D, Halayko AJ, Meurs H, et al. Inhibition of allergen-induced airway remodelling by tiotropium and budesonide: a comparison. Eur Respir J. 2007; 30:653–661.
25. Bateman ED, Rennard S, Barnes PJ, Dicpinigaitis PV, Gosens R, Gross NJ, et al. Alternative mechanisms for tiotropium. Pulm Pharmacol Ther. 2009; 22:533–542.
26. Goyal M, Jaseja H, Verma N. Increased parasympathetic tone as the underlying cause of asthma: a hypothesis. Med Hypotheses. 2010; 74:661–664.
27. Novelli F, Malagrinò L, Dente FL, Paggiaro P. Efficacy of anticholinergic drugs in asthma. Expert Rev Respir Med. 2012; 6:309–319.
28. Belmonte KE. Cholinergic pathways in the lungs and anticholinergic therapy for chronic obstructive pulmonary disease. Proc Am Thorac Soc. 2005; 2:297–304.
29. Barnes PJ. The pharmacological properties of tiotropium. Chest. 2000; 117:63S–66S.
30. Koumis T, Samuel S. Tiotropium bromide: a new long-acting bronchodilator for the treatment of chronic obstructive pulmonary disease. Clin Ther. 2005; 27:377–392.
31. Beeh KM, Moroni-Zentgraf P, Ablinger O, Hollaenderova Z, Unseld A, Engel M, et al. Tiotropium Respimat® in asthma: a double-blind, randomised, dose-ranging study in adult patients with moderate asthma. Respir Res. 2014; 15:61.
32. Kistemaker LE, Gosens R. Acetylcholine beyond bronchoconstriction: roles in inflammation and remodeling. Trends Pharmacol Sci. 2015; 36:164–171.
33. Kistemaker LE, Bos IS, Menzen MH, Maarsingh H, Meurs H, Gosens R. Combination therapy of tiotropium and ciclesonide attenuates airway inflammation and remodeling in a guinea pig model of chronic asthma. Respir Res. 2016; 17:13.
34. O'Connor BJ, Towse LJ, Barnes PJ. Prolonged effect of tiotropium bromide on methacholine-induced bronchoconstriction in asthma. Am J Respir Crit Care Med. 1996; 154:876–880.
35. Antoniu SA. Aclidinium bromide in experimental asthma. Expert Opin Investig Drugs. 2011; 20:871–873.
36. Lee LA, Yang S, Kerwin E, Trivedi R, Edwards LD, Pascoe S. The effect of fluticasone furoate/umeclidinium in adult patients with asthma: a randomized, dose-ranging study. Respir Med. 2015; 109:54–62.
37. Lee LA, Briggs A, Edwards LD, Yang S, Pascoe S. A randomized, three-period crossover study of umeclidinium as monotherapy in adult patients with asthma. Respir Med. 2015; 109:63–73.
38. Blais CM, Davis BE, Cockcroft DW. Duration of bronchoprotection of the long-acting muscarinic antagonists tiotropium & glycopyrronium against methacholine-induced bronchoconstriction in mild asthmatics. Respir Med. 2016; 118:96–101.
39. Kerstjens HA, Disse B, Schröder-Babo W, Bantje TA, Gahlemann M, Sigmund R, et al. Tiotropium improves lung function in patients with severe uncontrolled asthma: a randomized controlled trial. J Allergy Clin Immunol. 2011; 128:308–314.
40. Bateman ED, Tashkin D, Siafakas N, Dahl R, Towse L, Massey D, et al. A one-year trial of tiotropium Respimat plus usual therapy in COPD patients. Respir Med. 2010; 104:1460–1472.
41. Beeh KM, Kirsten AM, Dusser D, Sharma A, Cornelissen P, Sigmund R, et al. Pharmacodynamics and pharmacokinetics following once-daily and twice-daily dosing of tiotropium Respimat® in asthma using standardized sample-contamination avoidance. J Aerosol Med Pulm Drug Deliv. 2016; 29:406–415.
42. Timmer W, Moroni-Zentgraf P, Cornelissen P, Unseld A, Pizzichini E, Buhl R. Once-daily tiotropium Respimat® 5 µg is an efficacious 24-h bronchodilator in adults with symptomatic asthma. Respir Med. 2015; 109:329–338.
43. Vogelberg C, Moroni-Zentgraf P, Leonaviciute-Klimantaviciene M, Sigmund R, Hamelmann E, Engel M, et al. A randomised dose-ranging study of tiotropium Respimat® in children with symptomatic asthma despite inhaled corticosteroids. Respir Res. 2015; 16:20.
44. Vogelberg C, Engel M, Moroni-Zentgraf P, Leonaviciute-Klimantaviciene M, Sigmund R, Downie J, et al. Tiotropium in asthmatic adolescents symptomatic despite inhaled corticosteroids: a randomised dose-ranging study. Respir Med. 2014; 108:1268–1276.
45. Paggiaro P, Halpin DM, Buhl R, Engel M, Zubek VB, Blahova Z, et al. The effect of tiotropium in symptomatic asthma despite low- to medium-dose inhaled corticosteroids: a randomized controlled trial. J Allergy Clin Immunol Pract. 2016; 4:104–113.e2.
46. Kerstjens HA, Casale TB, Bleecker ER, Meltzer EO, Pizzichini E, Schmidt O, et al. Tiotropium or salmeterol as add-on therapy to inhaled corticosteroids for patients with moderate symptomatic asthma: two replicate, double-blind, placebo-controlled, parallel-group, active-comparator, randomised trials. Lancet Respir Med. 2015; 3:367–376.
47. Kerstjens HA, Engel M, Dahl R, Paggiaro P, Beck E, Vandewalker M, et al. Tiotropium in asthma poorly controlled with standard combination therapy. N Engl J Med. 2012; 367:1198–1207.
48. Fajt ML, Wenzel SE. Development of new therapies for severe asthma. Allergy Asthma Immunol Res. 2017; 9:3–14.
49. Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014; 43:343–373.
50. Kerstjens HA, Moroni-Zentgraf P, Tashkin DP, Dahl R, Paggiaro P, Vandewalker M, et al. Tiotropium improves lung function, exacerbation rate, and asthma control, independent of baseline characteristics including age, degree of airway obstruction, and allergic status. Respir Med. 2016; 117:198–206.
51. Song WJ, Cho SH. Challenges in the management of asthma in the elderly. Allergy Asthma Immunol Res. 2015; 7:431–439.
52. Trinh HK, Ban GY, Lee JH, Park HS. Leukotriene receptor antagonists for the treatment of asthma in elderly patients. Drugs Aging. 2016; 33:699–710.
53. Ban GY, Ye YM, Lee Y, Kim JE, Nam YH, Lee SK, et al. Predictors of asthma control by stepwise treatment in elderly asthmatic patients. J Korean Med Sci. 2015; 30:1042–1047.
54. Willson J, Bateman ED, Pavord I, Lloyd A, Krivasi T, Esser D. Cost effectiveness of tiotropium in patients with asthma poorly controlled on inhaled glucocorticosteroids and long-acting beta-agonists. Appl Health Econ Health Policy. 2014; 12:447–459.
55. Echave M, Ojanguren ME, Elías I, de Andrés-Nogales F, Oyagüez I, Casado M, et al. Cost-effectiveness of tiotropium in the treatment of patients with asthma. Value Health. 2015; 18:A501–A502.
56. Pawlik M, Walczak J, Pieniazek I. Economic evaluation of tiotropium administrated through the Respimat inhaler as add-on therapy in patients with uncontrolled severe asthma in Poland. Value Health. 2015; 18:A502.
57. Silva Miguel L, Manaças M, Pinheiro B. Economic evaluation of tiotropium for severe persistent asthma in Portugal. Value Health. 2015; 18:A502.
58. ClinicalTrials.gov (US). A Phase III parallel group study, comparing the efficacy, safety and tolerability of the fixed dose combination (FDC) of fluticasone furoate+umeclidinium bromide+vilanterol (FF/UMEC/VI) with the FDC of FF/VI in subjects with inadequately controlled asthma (NCT02924688). Bethesda (MD): ClinicalTrials.gov;2017.
59. Yang S, Goyal N, Beerahee M, Trivedi R, Lee L, Pascoe S. Dose-response modelling of umeclidinium and fluticasone furoate/umeclidinium in asthma. Eur J Clin Pharmacol. 2015; 71:1051–1058.
60. ClinicalTrials.gov (US). Chronic dosing cross-over study to assess the efficacy and safety of glycopyrronium (PT001) in adult subjects with intermittent asthma or mild to moderate persistent asthma (NCT02433834). Bethesda (MD): ClinicalTrials.gov;2016.
61. ClinicalTrials.gov (US). A multicentre, randomised, double-blind, placebo-controlled, 2-way cross-over study to evaluate the efficacy and safety of CHF 5259 (glycopyrrolate bromide) pMDI on top of QVAR® pMDI for the treatment of patients with uncontrolled asthma on low-medium dose of inhaled corticosteroids (NCT02296411). Bethesda (MD): ClinicalTrials.gov;2016.
62. ClinicalTrials.gov (US). The effect of glycopyrronium and indacaterol, as monotherapy and in combination, on the methacholine dose-response curve (NCT02953041). Bethesda (MD): ClinicalTrials.gov;2017.
63. Ohta K, Ichinose M, Tohda Y, Engel M, Moroni-Zentgraf P, Kunimitsu S, et al. Long-term once-daily tiotropium Respimat® is well tolerated and maintains efficacy over 52 weeks in patients with symptomatic asthma in Japan: a randomised, placebo-controlled study. PLoS One. 2015; 10:e0124109.
64. Hamelmann E, Bateman ED, Vogelberg C, Szefler SJ, Vandewalker M, Moroni-Zentgraf P, et al. Tiotropium add-on therapy in adolescents with moderate asthma: a 1-year randomized controlled trial. J Allergy Clin Immunol. 2016; 138:441–450.e8.
65. Hamelmann E, Bernstein JA, Vandewalker M, Moroni-Zentgraf P, Verri D, Unseld A, et al. A randomised controlled trial of tiotropium in adolescents with severe symptomatic asthma. Eur Respir J. 2017; 49:1601100.
66. ClinicalTrials.gov (US). A randomised, double-blind, placebo-controlled, parallel-group trial to evaluate efficacy and safety of tiotropium inhalation solution (2.5 mcg and 5 mcg) delivered via Respimat® inhaler once daily in the evening over 48 weeks in children (6 to 11 years old) with moderate persistent asthma (NCT01634139). Bethesda (MD): ClinicalTrials.gov;2016.
67. Szefler SJ, Murphy K, Harper T 3rd, Boner A, Laki I, Engel M, et al. A phase III randomized controlled trial of tiotropium add-on therapy in children with severe symptomatic asthma. J Allergy Clin Immunol. 2017; Forthcoming.
68. ClinicalTrials.gov (US). A phase II/III, randomised, double-blind, placebo-controlled, parallel group trial to evaluate safety and efficacy of tiotropium inhalation solution (2.5 µg and 5 µg) administered once daily in the afternoon via Respimat® inhaler for 12 weeks in patients 1 to 5 years old with persistent asthma (NCT01634113). Bethesda (MD): ClinicalTrials.gov;2015.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download