Abstract
Purpose
Chitin is a potent adjuvant in the development of immune response to inhaled allergens in the airways. According to other studies, chitin is known as multi-faced adjuvants which can induce Th2 responses. Recently, we found that TNF-α is a key mediator in the development of Th2 cell response to inhaled allergens. Here, we evaluated the immunologic mechanisms in the development of airway hypersensitivity to inhaled allergens, enhanced by house dust mite (HDM)-derived chitin.
Methods
The role of TNF-α and TLRs was evaluated in an airway hypersensitivity mouse model induced by a sensitization with an allergen (ovalbumin, OVA) and HDM-derived chitin using mice with the null mutation of target genes.
Results
The present study showed that airway sensitization with HDM-derived chitin plus OVA enhanced OVA-induced airway inflammation v. OVA alone. This phenotype was associated with the increased expression of Th1, Th2, and Th17 cytokines and also with the enhanced production of OVA-specific IgE, IgG1, and IgG2a. As for T cell responses, OVA-specific Th2 cell response, enhanced by chitin, was abolished by the treatment of chitinase, whereas Th1 and Th17 cell responses enhanced by this treatment. Moreover, the null mutation of the TNF-α gene revealed similar effects as the chitinase treatment. In contrast, all the OVA-specific T cell responses, enhanced by chitin, were blocked by the absence of TLR2, but not of TLR1, TLR4, or TLR6.
House dust mites (HDMs), including Dermatophagoides farinae (Der f) and D. pteronyssinus (Der p), are the main source of inhaled allergens, which induce Th2-type airway inflammatory diseases, such as asthma, in sensitized subjects.123 Chitin, a biopolymer of N-acetyl-b-D-glucosamine, is the second most abundant polysaccharide in nature after cellulose, and it is found in the exoskeleton and digestive tract of arthropods, including HDM, the walls of fungi, and the microfilarial sheath of parasitic nematodes.4567 Inhaled allergens derived from HDM are thought to bind to chitin, and a part of HDM-derived allergens are frequently found in the midgut epithelium and fecal pellets.89 A recent report showed that chitin is a potent multifaceted adjuvant that can enhance allergen-specific Th2-, Th1-, and Th17- cell responses.10 In contrast, several reports showed that chitin down-regulated allergen-induced IgE production and airway inflammation, which are associated with the decreased expression of Th2 cytokines, such as IL-4 and IL-5.1112
The innate immune system is the first line of defense against infection by other organisms. Cells and molecules of the innate immune system recognize and respond to pathogens, but unlike the adaptive immune system, it does not confer long-lasting or protective immunity to the host.13 TNF-α, an important mediator of the inflammatory response, is produced by innate immune cells, including macrophages, mast cells, and lung epithelial cells. TNF-α induces the infiltration of inflammatory cells to the inflamed site through the upregulation of adhesion molecules and the production of chemokines.14 In terms of the role of TNF-α in the development of airway hypersensitivity to inhaled allergens, we revealed that TNF-α is a key mediator in the development of Th2-cell response to inhaled allergens, which are augmented by innate immune response induced by lipopolysaccharide (LPS)15 or double-stranded RNA.16
Toll-like receptors (TLRs) are a class of proteins that play a key role in the innate immune system.17 They are a type of pattern recognition receptor (PRR), and single, membrane-spanning, non-catalytic receptors usually expressed in sentinel cells, such as macrophages, dendritic cells, and epithelial cells, that recognize structurally conserved molecules.18 Chitin, a kind of conserved molecules, is known to bind to TLR2.10 Though most TLRs appear to function as homodimers, TLR2 forms heterodimers with TLR1 or TLR6.18
In the present study, we investigated the effect of HDM-derived chitin in the development of airway hypersensitivity to inhaled allergens using chitinase. We also evaluated immunologic mechanisms of airway hypersensitivity enhanced by HDM-derived chitin using TNF-α and TLR-deficient mice.
TNF-α-deficient and WT mice (C57BL/6 background) were purchased from Jackson Laboratories (Bar Harbor, ME, USA). TLR1-, TLR2-, and TLR6-deficient mice were purchased from Oriental BioService (Kyoto, Japan). Mice were kept and mated in a specific pathogen-free room at POSTECH, and all of the mice experiments were permitted by POSTECH Ethics Committee (permit No: 2014-01-0008).
OVA, HDM (D. pteronyssinus), and Brugua malayi chitinase were obtained from Sigma-Aldrich (St. Louis, MO, USA), Korean National Arthropods of Medical Importance Resource Bank (Seoul, Korea), and New England Biolabs (Ipswich, MA, USA), respectively.
For the purification of chitin from HDM, 100 mg of HDM was washed in phosphate buffered saline (PBS) for 24 hours and then dried at 55℃ for 10 hours. Next, 2 M NaOH was added and incubated at room temperature for 15 hours. Then, NaOH-added HDM was washed with distilled water 3 times and then incubated with 0.5 M H2SO4 at 50℃ for 16 hours. After incubation, it was washed again and incubated with 2 M NaOH for 30 minutes. Finally, chitin was washed with distilled water 3 times and lyophilized to remove the moisture. Fourier transform infrared (FTIR) spectroscopy was performed at a spectral resolution of 4 cm-1 with a Bomem DA8 FTIR spectrometer equipped with a liquid nitrogen-cooled mercury cadmium telluride (MCT) detector which is in the range 800-4,000 cm-1 under vacuum. The diffuse reflectance attachment (Harrick Scientific Corporation) was used in this paper. Samples were prepared using the mortar to make properly fine powder to get FTIR spectra. All the diffuse-reflectance FTIR spectra were measured by co-adding 256 scans. Acquired chitin amount was 700 µg per 1 mg of mite, and the endotoxin level was less than 0.6 endotoxin unit per 100 µg of mite. Protein was not detected when we evaluated using BCA methods.
The concept of study protocols was mentioned in our previous study.19 Briefly, 6-week-old mice were sensitized intranasally with 75 µg of OVA plus 100 µg of HDM-derived chitin with or without chitinase on days 0, 1, 2, and 7. After stimulation, 50 µg of OVA alone was administrated intranasally on days 14, 15, 21, and 22, and then evaluated 24 hours after final OVA challenge. In the case of the chitinase-treated group, 1 unit of chitinase was co-incubated with 100 µg of HDM-derived chitin (reaction volume-in 30 µL PBS) at 37℃ for 1 hour, and after enzyme digestion, heat inactivation was done for stopping the reaction. Thereafter, it was administrated intranasally with OVA. In the present study, we used the same amount or concentration of chitinase in all the experiments. All of the protocols are mentioned in supplementary Fig. 1.
Immune cells in BAL fluids were analyzed as previously described.20 Briefly, through centrifugation, cell pellets were isolated and diluted in 50 µL of PBS, and more than 300 inflammatory cells were counted and classified as macrophages, lymphocytes, neutrophils, or eosinophils.
To isolate single-cell from the lung tissue, we chopped tissue and incubated at 37℃ with 0.05% trypsin (GIBCO, Grand Island, NY, USA) and 200 unit/mL of collagenase (GIBCO). Ten minutes after incubation, we ground lung tissue with the cell strainer (BD Falcon, Bedford, MA, USA) and incubated at 4℃ with RBC lysis buffer (StemCell Technologies, Vancouver, Canada). For cell isolation from lymphnodes, we ground lymphnode using the cell strainer and incubated with RBC lysis buffer as in the above method.
Sectioned lung tissues (4-µm thickness) were stained with hematoxylin and eosin (H&E) after pressure fixation with Streck solution (Streck Laboratories, La Vista, NE, USA). Under the same magnification (Magnification: 100×, scale bar: 200 µm), slides were observed.19
After single preparation, cells were cultured (2.0×106/mL) with anti-CD28 antibody in anti-CD3 antibody (1 µg/mL each; eBioscience, San Diego, CA, USA)- coated 24-well plates at 37℃ in RPMI 1640 (Hyclone, UT, USA). Cell culture supernatant was harvested 12 hours after restimulation.
Levels of cytokines, including IL-1β, IL-4, IL-5, IL-6, IL-10, IL-12p70, IL-17, IL-23, TNF-α, eotaxin, TGF-β, TSLP, IFN-γ, and IP-10 in BAL fluid or harvested supernatants, were evaluated using the enzyme-linked immunosorbent assays (ELISA) in accordance with the manufacturer's instructions (R&D Systems, Minneapolis, MN, USA). The detection limit for cytokine ELISA kits were 7.8125 pg/mL.
The levels of OVA-specific antibody sutbypes, such as IgG1, IgG2a, and IgE, in serum were measured by using the ELISA in accordance with the manufacturer's instructions (Bethyl laboratories INC, Montgomery, TX, USA).
For evaluating the T-cell proliferation, CFSE (Invitrogen, Carlsbad, CA, USA) was labeled to isolated cells from lung-draining lymphnodes (2.0×106 cells/mL) as the manufacturer's instructions. After labeling, with 100 µg/mL of OVA, cells were stimulated for 72 hours, stained with anti-CD3 (APC labeled) antibody for 30 minutes at 4℃, and then fixed with 4% paraformaldehyde for 10 minutes at room temperature. Then, cells were acquisited on a fluorescence-activated cell sorting (FACS) Calibur flow cytometer (BD Biosciences, San Jose, CA, USA) analyzed using CellQuest Pro software.
Data are presented as mean with SEM. The statistical significance was evaluated by analysis of variance (ANOVA) or Student's t test. To determine differences between cells of individual groups, we used ANOVA, and Student's t test was used for the comparison of cytokines and antibodies. Statistical significance was accepted in case of P<0.05.
HDMs are usually found in house dust, and their body crusts are likely to exist there ere as well. Recent evidence indicates that chitin particles are one of the candidates that may induce immune responses. First, we evaluated the effect of HDM-derived whole extract on the development of airway hypersensitivity to inhaled allergens. To test this, wild type (WT) mice were sensitized intranasally with 75 µg of OVA+100 µg of HDM extract, with or without chitinase 3 times, and then phenotypes were evaluated 24 hours after the last sensitization. Cellularity in bronchoalveolar lavage (BAL) fluid showed that the infiltration of inflammatory cells, such as macrophages, neutrophils, and eosinophils, was increased in mice sensitized with OVA and HDM extract compared to OVA alone. Furthermore, this phenotype was reversed by treatment with chitinase (Fig. S2A). The production of pro-inflammatory cytokines, such as IL-1β, TNF-α, IL-4, and thymic stromal lymphopoietin (TSLP), were elevated in mice sensitized with OVA and HDM extract; this result was reversed by chitinase treatment (Fig. S1B). The production of IL-6, IL-10, IL-12p70, and IL-23 was also enhanced in mice sensitized with OVA and HDM extract, but chitinase treatment further enhanced the production of these cytokines (Fig. S2B). These findings suggest that HDM-derived chitin is an important adjuvant in the development of airway hypersensitivity to inhaled HDM-derived allergens.
Next, we evaluated the effect of chitinase treatment on the development of adaptive immune responses to inhaled HDM allergens after airway exposure to HDM-derived whole extract. To test this, WT mice were sensitized intranasally with 75 µg of OVA and 100 µg of HDM extract, with or without chitinase, on days 0, 1, 2, and 7, and then challenged intranasally with 50 µg of OVA on days 14, 15, 21, and 22. The evaluation was performed 24 hours after the last OVA challenge. BAL cellularity and lung histology showed that the airway application of OVA plus HDM extract induced airway inflammation, which was inhibited by chitinase treatment (Fig. 1A and B). As for the production of allergen-specific antibody subtypes, OVA-specific IgE production, enhanced by HDM extract, was reversed by chitinase treatment, but OVA-specific IgG1 and IgG2a productions, enhanced by HDM extract, were rather enhanced by this treatment (Fig. 1C). With regard to cytokine production, the production of Th2-type cytokines, such as IL-4, IL-5, eotaxin, and TGF-β, were enhanced in mice sensitized with OVA plus HDM extract vs OVA alone, and this phenotype was reversed by chitinase treatment (Fig. 1D, left panel). However, the production of IL-17, a Th17 cytokine, and interferon gamma-induced protein 10 (IP-10) and IFN-γ, Th1 cytokines was enhanced by HDM extract, but not inhibited by chitinase treatment (Fig. 1D, right). When T- cell response was evaluated by using T cells isolated from the lung tissues and lung-draining lymph nodes, IL-4 production in lung and LN T cells was increased in mice sensitized with OVA and HDM extract vs OVA alone, and this phenotype was reversed by chitinase treatment; however, IL-17 and IFN-γ productions in lung and lymph node T cells, enhanced by HDM extract, were further enhanced by chitinase treatment (Fig. 1E). These findings suggest that HDM-derived chitin enhances Th2-cell response to inhaled HDM allergens, but is not involved in Th1- or Th-17 cell responses to HDM allergens.
In the present study, chitin was extracted from HDM to evaluate the direct adjuvant effect of HDM-derived chitin on the development of airway hypersensitivity to inhaled allergens. When the isolated HDM-derived chitin was compared to a commercially available chitin using FTIR spectroscopy, the 2 chitins showed similar profiles (Fig. 2A). To evaluate the direct effect of HDM-derived chitin on the production of pro-inflammatory cytokines, HDM-derived chitin was applied intranasally into WT mice once (each dose of HDM chitin), and then the effect was evaluated 12 hours after application. The production of pro-inflammatory cytokines, such as IL-6 and TNF-α, was not enhanced upon treatment with HDM-derived chitin less than 100 µg, but significantly enhanced by treatment with 100 µg of HDM-derived chitin (Fig. 2B). We also evaluated the direct adjuvant effect of HDM-derived chitin on the development of adaptive immune response. To test this, WT mice were sensitized intranasally with OVA, with or without different doses of HDM-derived chitin 4 times, and then challenged with OVA alone 4 times (OVA challenge model). The evaluation was performed 24 hours after the last OVA challenge. Inflammatory cell infiltration into the lung was observed in the groups sensitized with 10- or 100-µg of HDM-derived chitin plus OVA, and the type of infiltrated cells was similar between the 2 groups (Fig. 2C). However, in the case of OVA-specific antibody production, the production of OVA-specific IgG1, IgG2a, and IgE was enhanced only in mice sensitized with 100 µg of chitin plus OVA, whereas it was not enhanced in the other groups (Fig. 2D). Similarly, the production of cytokines, related to Th1-, Th2-, and Th17- cell responses, was increased in mice sensitized with 100 µg of chitin plus OVA v. the other groups (Fig. 2E). These data suggest that HDM-derived chitin may be a sufficient adjuvant to induce allergen-specific Th2-, Th1-, and Th17- cell responses.
To evaluate innate immune response induced by HDM-derived chitin, 100 µg of chitin was applied intranasally to WT mice, and the response was evaluated at each time point. Inflammatory cell infiltration was significantly enhanced 12 hours after airway exposure to HDM-derived chitin (Fig. 3A). As for the production of T cell-polarizing cytokines, the production of Th2-polarizing cytokines, such as TNF-α, IL-4, and TSLP, was significantly enhanced from 6 or 12 hours after the application (Fig. 3B, left); that of Th1-/Th17-polarizing cytokines, such as VEGF, IL-6, and IL-12, were significantly enhanced from 3 or 12 hours after the application (Fig. 3B, right).
We also applied HDM-derived chitin into the mouse airways, with or without chitinase, and then evaluated the innate immune response 12 hours after the last application. BAL cellularity showed that HDM-derived chitin enhanced the infiltration of inflammatory cells, such as macrophages, neutrophils, and eosinophils, in the lung; among the infiltrated cells, eosinophil infiltration was completely inhibited by chitinase treatment, whereas macrophage and neutrophil infiltration partially inhibited (Fig. 3C). In addition, the production of Th2 polarizing cytokines, such as TNF-α, IL-4, and TSLP, enhanced by HDM-derived chitin was significantly inhibited by this treatment, but that of Th1-/Th17-polarizing cytokines, such as IL-6, IL-12p70, and IL-23, were rather enhanced (Fig. 3D). Taken together, these findings suggest that HDM-derived chitin induces innate immune response, such as the up-regulation of Th2-polarizing cytokines, for the generation of Th2- cell response to inhaled allergens.
We evaluated the effect of chitinase treatment on the HDM-derived chitin for the development of OVA-specific adaptive immune response. To do this, the OVA challenge model was applied to WT mice, with or without chitinase treatment. The evaluation was performed 24 hours after the last OVA challenge. BAL cellularity and lung histology revealed that the lung infiltration of inflammatory cells, enhanced by HDM-derived chitin, was significantly inhibited by chitinae treatment on the HDM-derived chitin (Fig. 4A and B). As for the production of OVA-specific antibody subtypes, OVA-specific IgE production, enhanced by the chitin was completely inhibited by the chitinase treatment, but OVA-specific IgG1 and IgG2a enhanced by the chitin were enhanced by this treatment (Fig. 4C). Additionally, the production of Th2-type cytokines, such as IL-4, IL-5, eotaxin, and TGF-β, enhanced by the chitin was found to be significantly inhibited by the chitinase treatment (Fig. 4D, left). However, the production of IL-10 and of Th1-/Th17-type cytokines, such as IL-17, IL-12p70, and IFN-γ, enhanced by the chitin, was enhanced by this treatment (Fig. 4D, right). We also evaluated the production of cytokines from T cells isolated from lung tissues and regional lymph nodes. Th2 (IL-4), Th1 (IFN-γ), and Th17 (IL-17) cytokine productions from both lung and lymph node T cells were enhanced after stimulation with anti-CD3/-CD28 antibodies in the OVA+HDM-derived chitin group vs the OVA group; the production of the Th2 cytokine IL-4 was reversed by the chitinase treatment, whereas that of Th1 (IFN-γ) and Th17 (IL-17) cytokines enhanced by this treatment (Fig. 4E). To sum up, these findings indicate that the chitinase may inhibits Th2-cell response to inhaled allergens, enhanced by HDM-derived chitin, but enhances Th1-/Th17-cell responses.
Chitin is known to be recognized by TLR2.10 We evaluated the role of TLR2 in the development of allergen-specific airway hypersensitivity enhanced by HDM-derived chitin using TLR2-deficient mice. To evaluate the role of TLR2 in innate immune response induced by HDM-derived chitin, HDM-derived chitin with OVA was applied intranasally to WT and TLR2-deficient mice 3 times, and then evaluated 12 hours after the last sensitization. BAL cellularity showed that the lung infiltration of macrophages and eosinophils, enhanced by HDM-derived chitin, was completely abolished by the absence of TLR2, although neutrophilic infiltration was incompletely reduced (Fig. 5A). As for the production of immune modulating cytokines, such as IL-1β, TNF-α, IL-4, TSLP, IL-6, IL-12p70, and IL-23, enhanced by HDM-derived chitin was significantly inhibited by the absence of TLR2 (Fig. 5B). We also evaluated the role of TLR2 in the development of allergen-induced adaptive immune response and airway inflammation. To do this, the OVA-induced airway hypersensitivity mouse model was applied to WT and TLR2-deficient mice, and the evaluation was performed 24 hours after the last OVA challenge. BAL cellularity and lung histology showed that the lung infiltration of inflammatory cells, such as macrophages, neutrophils, and eosinophils, induced by OVA challenge in WT mice was almost completely inhibited by the absence of TLR2 (Fig. 5C and D). Lung histological findings were similar to these observations (Fig. 5D). As for the production of allergen-specific antibody subtypes, OVA-specific IgE, IgG1, and IgG2a productions were significantly inhibited by the absence of TLR2 (Fig. 5E). Similarly, the production of Th2-type cytokines, such as IL-4, IL-5, eotaxin, and TGF-β, and Th1/Th17-type cytokines, such as IL-17, IFN-γ, and IP-10, induced by allergen challenge was significantly inhibited by the absence of TLR2 (Fig. 5F). In terms of T cell responses, the production of Th2, Th17, and Th1 cytokines, such as IL-4, IL-17, and IFN-γ, respectively, in T cells from both lung tissues and lung-draining LNs, induced by allergen challenge in WT mice were abolished by the absence of TLR2 (Fig. 5G). Taken together, these data indicate that TLR2 is an essential PRR in the recognition of HDM-derived chitin.
It is possible that TLR2 could exist in homodimer, or heterodimer with TLR1 or TLR6. Thus, we aimed to evaluate the role of TLR1 and TLR6 in the recognition of HDM-derived chitin. To test this, HDM-derived chitin was applied intranasally to TLR1- and TLR6-deficient mice. In addition, to exclude the effects of LPS contamination in HDM-derived chitin, HDM-derived chitin was also applied intranasally to TLR4-deficient mice. This study showed that innate immune response, including airway inflammation, induced by HDM-derived chitin was not affected by the absence of TLR1, TLR4, or TLR6. These data suggest that the TLR2 homodimer form, rather than heterodimer with TLR1 or TLR6, plays a key role in the recognition of HDM-derived chitin (Fig. S3).
Our previous data showed that TNF-α plays a key role in the development of Th2-cell response to inhaled allergens enhanced by dsRNA.16 Finally, we evaluated the role of TNF-α in the development of airway hypersensitivity to inhaled allergens enhanced by HDM-derived chitin. First, to test innate immune response, OVA with or without HDM-derived chitin was applied intranasally to WT and TNF-α-deficient mice 4 times and then the evaluation performed 24 hours after the last sensitization. BAL cellularity showed that the infiltration of inflammatory cells enhanced by HDM-derived chitin was significantly inhibited by the absence of TNF-α (Fig. 6A). Additionally, the production of Th2-polarizing cytokines, such as IL-4 and TSLP, enhanced by HDM-derived chitin was significantly inhibited by the absence of TNF-α, but that of Th1-/Th17-polarizing cytokines, such as IL-12p70, IL-6, and IL-23, was rather enhanced (Fig. 6B). Together, these findings suggest that TNF-α produced by HDM-derived chitin may be a key innate immune mediator in the development of Th2-cell response to inhaled allergens.
Next, we evaluated the role of TNF-α in the development of allergen-induced airway inflammation enhanced by HDM-derived chitin. To test this, the OVA challenge model was applied to WT and TNF-α-deficient mice. BAL cellularity and lung histology showed that airway inflammation induced by the OVA challenge in WT mice was significantly decreased in TNF-α-deficient mice (Fig. 6C and D). In addition, the production of OVA-specific IgE enhanced by HDM-derived chitin was abolished by the absence of TNF-α, but that of OVA-specific IgG1 and IgG2a was enhanced (Fig. 6E). As for the production of cytokines, BAL levels of Th2-type cytokines, such as IL-4, IL-5, eotaxin, and TGF-β, were significantly decreased TNF-α-deficient mice sensitized with OVA plus chitin vs WT mice sensitized in the same manner, although those of Th1/17-type cytokines, such as IL-17, IP-10, and IFN-γ, were not decreased by the absence of TNF-α (Fig. 6F). With regard to T-cell response, the production of a Th2 cytokine, IL-4, from both lung and LN T cells was abolished by the absence of TNF-α, but that of Th1, IFN-γ, and Th17, IL-17, cytokines was enhanced (Fig. 6G). Taken together, TNF-α induced by HDM-derived chitin exposure may be an important mediator in the development of Th2 cell response to inhaled allergens.
Various mouse models of allergic airway diseases have been used and have improved our understanding of the pathogenesis of these diseases. Most of these models have evaluated responses to a single antigen or allergen, most commonly OVA, a prototypic innocuous antigen.2122 In several studies, a variety of adjuvants, including aluminum hydroxide, lipopolysaccharide (LPS), and double-stranded RNA, were added to OVA to induce distinct types of airway inflammation.16192123 However, humans are frequently exposed simultaneously to various allergens, each with distinct antigenic potential. In a previous study, HDM, the major source of inhaled allergens in asthma pathogenesis was reported to enhance airway sensitization to other antigens, such as OVA, through the "bystander" effect, in addition to its own allergenic properties.24 However, the mechanism of how HDM induces the bystander effect remains to be investigated. Chitin is frequently found in the exoskeleton, mid-gut epithelium, and fecal pellets of HDM.9 In the present study, we demonstrated that HDM-derived chitin enhances airway hypersensitivity to inhaled allergens, which is dependent on the TLR2 signaling pathway. In addition, we found that TNF-α induced by HDM-derived chitin is a key mediator in the development of Th2-cell response to inhaled allergens, but not of Th1- and Th17-cell responses.
In the present study, we used the HDM-derived chitin to investigate the bystander effect of HDM on the development of airway hypersensitivity to inhaled allergens. This study showed that chitins, extracted from HDM, elicited Th2- and Th1-/Th17-cell responses to inhaled allergens, although the Th2-cell response play a major role in the development of allergic airway inflammation. Chitinase is an enzyme that degrades the β (1-4) glycosidic bond in chitin by hydrolysis.25 A previous study showed that allergen-induced Th2-type airway inflammation enhanced by a commercially available chitin was inhibited by chitin digestion with acid mammalian chitinase (AMCase).26 In the present study, we also found that the OVA-induced Th2-type airway inflammation was abolished by chitinase treatment. However, the present study showed that OVA-specific Th1- or Th17-cell responses enhanced by HDM-derived chitin were rather enhanced by chitinase treatment. Previous studies suggest that the size of chitins is an important factor for determining the character of immune responses. Shibata et al.27 reported that phagocytosable chitin (1-10 µm) induces IFN-γ production in alveolar macrophages. Reese et al.26 showed that chitin (of unknown, varying sizes) induces Th2-type immune response, and Da Silva et al.28 reported that intermediate-size chitin (40-70 µm) induces mixed T cell responses (Th1, Th2, and Th17). In addition, it has been reported that small-size chitin (<40 µm) induces TNF-α and IL-10 production in alveolar macrophages.1028 We evaluated the size change of HDM-derived chitin following chitinase digestion and found a modest increase in small-sized chitin, as shown in Fig. S4. Briefly, small- (<40 µm) and intermediate-size of chitins (40-70 µm) were increased after enzyme digestion. In contrast, large-size chitin showed opposite tendency. In this context, our result is consistent with those of previous reports. Taken together, these findings suggest that the change of HDM-derived chitins to smaller ones by chitinase treatment induces the down-regulation of allergen-specific Th2-cell response enhanced by chitin.
Adaptive immune responses enhanced by chitins have been reported to be mainly dependent on the TLR2 signaling pathway.1029 The TLR2 signaling pathway induces the innate immune response via TLR2 homodimer or TLR2 heterodimer, with TLR1 or TLR6.30 The present study showed that innate and adaptive immune responses induced by HDM-derived chitin were abolished by the absence of TLR2. However, this study also showed that innate immune response induced by chitin was not affected by the absence of TLR1 or TLR6. These findings indicate that HDM-derived chitin may enhance the airway hypersensitivity to inhaled allergens, mainly via the TLR2 homodimer form.
Our previous studies indicate that TNF-α is a key mediator in the development of Th2-cell response enhanced by LPS and dsRNA.1516 In the present study, we postulated that TNF-α induced by HDM-derived chitin would induce OVA-specific Th2 cell response. Indeed, this study showed that OVA-specific Th-2, but Th1-/Th17-cell responses, enhanced by HDM-derived chitin were blocked by the absence of TNF-α. This study also showed that the production of TNF-α was enhanced in alveolar macrophages and airway epithelial cells after the in vitro stimulation of HDM-derived chitin, as shown in Fig. S5. Previous evidence indicates that intermediate-and small-sized chitins recognized by macrophages can enhance the production of TNF-α.28 Note that chitinase treatment in the present study induces HDM-derived chitins into the smaller ones and abolishes allergen-specific Th2-cell response enhanced by chitin. To sum up, these findings suggest that TNF-α produced by small-sized chitins derived from HDM may enhance Th2-type airway hypersensitivity to inhaled allergens.
Previous reports showed that chitin induces the production of the Th2 cytokines IL-4 and 13 by eosinophils and basophils, and alternative macrophage activation.26 Recently, we reported that macrophage-derived TNF-α induces the Th2-cell response to inhaled allergen through NKT cells.31 In this study, we showed that induction of Th2 cytokines from NKT cells by treatment of recombinant TNF-α or co-culture of alveolar macrophages. In the present study, we showed that stimulation of macrophages by chitin treatment induced TNF-α production which is able to induce Th2 cytokines from NKT cells. In this context, we can suggest that HDM-derived chitin may induce Th2 immune response through the TLR2-macrophage-NKT axis.
In the case of enhanced Th1-and Th17- cell responses under the absence of TNF-α, it is possible that the down-regulation of IL-4 by the absence of TNF-α could induce the enhancement of both responses. According to a previous study, IL-4 can induce the down-regulation of IL-6 and the induction of M2 macrophages.32 In our previous and this studies, the absence of TNF-α showed the down-regulation of IL-4 and the elevation of IL-6/IL-12.16 In this context, it is possible that enhanced Th1 and Th17 could be caused by absence of suppression and M2 macrophages by TNF-α-induced IL-4.
In summary, this study indicates that HDM-derived chitins may induce innate immune response and enhance adaptive immune response to inhaled allergens, mainly via the TLR2 homodimer-signaling pathway. In addition, this study suggest that TNF-α produced by small-sized HDM-derived chitins after chitinase treatment can be a key mediator in the development of Th2 sensitization to inhaled allergens. These findings imply that HDM-derived chitin is an important target for the prevention of respiratory allergic diseases induced by HDM and that TNF-α is a good therapeutic target for the treatment of Th2 hypersensitivity to inhaled allergens.
Figures and Tables
Fig. 1
Th2-dominant airway inflammation induced by HDM extracts plus OVA is down-regulated by chitinase treatment. For all panels, wild-type mice (C57BL/6 background) were sensitized with 75 µg of OVA+100 µg of HDM with or without chitinase, then challenged with OVA (50 µg) alone. The evaluations were performed 24 hours after last OVA challenge. *P<0.05; **P<0.01; ***P<0.001 relative to individual control groups, OVA or OVA+Chitinase, respectively; #P<0.05; ##P<0.01; ###P<0.001; n.s, not significant. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least 3 independent experiments. (A) Bronchoalveolar lavage (BAL) cellularity after acute allergen challenge. (B) Lung histologic findings (a, OVA; b, OVA+chitinase; c, OVA+HDM; d, OVA+HDM+chitinase; magnification: 100×, scale bar: 200 µm). (C) The production of OVA-specific IgE, IgG1, and IgG2a in serum after acute OVA challenge. (D) Cytokine productions of IL-4, IL-5, eotaxin, TGF-β, IL-10, IL-17, IP-10, and IFN-γ in BAL fluid after last allergen challenge. For (E) panel, cells were isolated from lung-draining lymph nodes (LN) and lung tissues, and incubated with PBS or anti-CD3 and CD28 antibodies for 12 hours. Levels of each cytokine were evaluated in supernatant fraction. (E) Levels of IL-4, IL-10, IL-17, and IFN-γ from lung cells, and lung-draining LN cells.
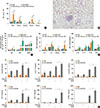
Fig. 2
Chitins extracted from HDM have an adjuvant effect to enhance adaptive immune response to inhaled allergens. (A) Fourier transform infrared (FTIR) spectroscopy of commercial crab chitin (commercial chitin) and HDM derived chitin (sample1). For (B) panel, HDM-derived chitin was sensitized to wild type (WT) mouse airways and then evaluated after 12 hours. For (C-E) panels, wild-type mice (C57BL/6 background) were sensitized with 75 µg of OVA, or 75 µg of OVA+1, 10, or 100 µg of HDM-derived chitin. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least 2 independent experiments. *P<0.05; **P<0.01; ***P<0.001 relative to the OVA group; (B) Levels of IL-6, and TNF-α in BAL fluid after sensitization of each dose of HDM-derived chitin. For (C-E), mice were challenged with OVA (50 µg) alone and evaluated 24 hours after the last OVA challenge. (C) Bronchoalveolar lavage (BAL) cellularity after 24 hours of last OVA challenge. (D) Levels of OVA-specific IgE, IgG1, and IgG2a in serum after acute challenge. (E) Levels of IL-4, IL-5, eotaxin, TGF-β, IL-10, IL-17, IFN-γ, and IP-10 in BAL fluid after last acute OVA challenge.
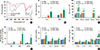
Fig. 3
Th2-type innate response, induced by HDM-derived chitin is abolished by chitinase treatment. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least 3 independent experiments. *P<0.05; **P<0.01; ***P<0.001; n.s, not significant. relative to basal or the OVA group; For panel (A) and (B), 100 µg of HDM-derived chitin was sensitized once to wild type (WT) mouse airways and then evaluated at various time points. (A) Inflammatory cell infiltration of airway after HDM-derived chitin sensitization. (B) Levels of TNF-α, IL-4, TSLP, VEGF, IL-6, IL-12p70, and IL-23 in bronchoalveolar lavage (BAL) fluids. For (C-D), the evaluation was performed at 12 hours after HDM-derived chitin sensitization. (C) BAL cellularity after sensitization. (D) Levels of IL-1β, TNF-α, IL-4, TSLP, IL-6, IL-10, IL-12p70, and IL-23 in BAL fluid after sensitization.

Fig. 4
OVA-induced Th2-cell response enhanced by HDM-derived chitin is abolished by chitinase treatment. For all panels, wild-type (WT) mice (C57BL/6 background) were sensitized with 75 µg of OVA, 75 µg of OVA+chitinase, 75 µg of OVA+100 µg of HDM-derived chitin, or 75 µg of OVA+100 µg of HDM-derived chitin+chitinase, then challenged with OVA (50 µg) alone, and evaluated 24 hours after last OVA challenge. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least 3 independent experiments. *P<0.05; **P<0.01; ***P<0.001 relative to the OVA group; #P<0.05; ##P<0.01; ###P<0.001. (A) Bronchoalveolar lavage (BAL) cellularity after acute OVA challenge. (B) Lung histologic findings (a, OVA; b, OVA+chitinase; c, OVA+HDM-derived chitin; d, OVA+HDM-derived chitin+chitinase; magnification: 100×, scale bar: 200 µm). (C) Levels of OVA-specific IgE, IgG1, IgG2a in serum after acute challenge. (D) Levels of IL-4, IL-5, eotaxin, TGF-β, IL-10, IL-17, IL-12p70, and IFN-γ in BAL fluid after acute challenge. For panels (E), cells were isolated from lung-draining lymph nodes (LNs) and lung tissues, and incubated with PBS or CD3 and CD28 antibodies for 12 hours. Levels of each cytokine were evaluated in supernatant fraction. (E) Levels of IL-4, IL-10, IL-17, and IFN-γ from lung and lung-draining LN cells.
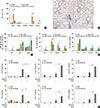
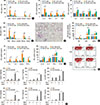
Fig. 5
OVA-induced adaptive immune responses enhanced by HDM-derived chitin is abolished by the absence of TLR2. For all panels, TLR2-deficient mice and wild-type (WT) mice (C57BL/6 background) were sensitized with 75 µg of OVA, or 75 µg of OVA+100 µg of HDM derived chitin. *P<0.05; **P<0.01; ***P<0.001 relative to the wild-type OVA group; #P<0.05; ##P<0.01; ###P<0.001. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least 2 independent experiments. For (A-C) panels, evaluations were performed after 12-hour sensitization. (A) Bronchoalveolar lavage (BAL) cellularity after sensitization. (B) Levels of IL-1β, TNF-α, IL-4, TSLP, IL-6, IL-10, IL-12p70, and IL-23 in BAL fluid after sensitization. For panels (C-H), mice were challenged with OVA (50 µg) alone, and evaluated 24 hours after last OVA challenge. (C) BAL cellularity after acute challenge. (D) Lung histologic findings (a, WT_OVA; b, TLR2 KO_OVA; c, WT_Chitin+OVA; d, TLR2 KO_OVA+chitin; magnification: 100×, scale bar: 200 µm). (E) Levels of OVA-specific IgE, IgG1, IgG2a in serum after acute challenge. (F) Levels of IL-4, IL-5, eotaxin, TGF-β, IL-10, IL-17, IFN-γ, and IP-10 in BAL fluid after acute challenge. For panels (G, H), cells were isolated from lung-draining lymph nodes (LNs) and lung tissues. (G) The levels of memory T-cell proliferation by stimulation of OVA in LN cells from each mouse. For (H), isolated cells were incubated with PBS or CD3 and CD28 antibodies for 12 hours. Levels of individual cytokines were evaluated in supernatant fraction. (H) Levels of IL-4, IL-10, IL-17, and IFN-γ from lung and lung-draining LN cells.
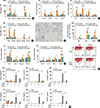
Fig. 6
OVA-induced Th2-cell response, enhanced by HDM-derived chitin is abolished by the absence of TNF-α. Th2 responses induced by airway sensitization with ovalbumin (OVA) plus HDM-derived chitin is diminished in the absence of TNF-α, whereas the Th1- and Th17-cell responses are enhanced. For all panels, TNF-α-deficient (TNF KO) mice and wild-type (WT) mice (C57BL/6 background) were sensitized with 75 µg of OVA, or 75 µg of OVA+100 µg of HDM-derived chitin. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least two independent experiments. *P<0.05; **P<0.01; ***P<0.001 relative to the wild-type OVA group; #P<0.05; ##P<0.01; ###P<0.001; n.s, not significant. For panel (A) and (B), the evaluation was performed 12 hours after sensitization. (A) Bronchoalveolar lavage (BAL) cellularity after sensitization. (B) Levels of IL-1β, TNF-α, IL-4, TSLP, IL-6, IL-10, IL-12p70, and IL-23 in BAL fluid after sensitization. For (C-H), mice were challenged with OVA (50 µg) alone and evaluated 24 hours after last OVA challenge. (C) BAL cellularity after acute challenge. (D) Lung histologic findings (a, WT_OVA; b, TNF KO_OVA; c, WT_OVA+chitin; d, TNF KO_OVA+chitin; magnification: 100×, scale bar: 200 µm). (E) Levels of OVA-specific IgE, IgG1, and IgG2a in serum after acute challenge. (F) Levels of IL-4, IL-5, eotaxin, TGF-β, IL-10, IL-17, IP-10, and IFN-γ in BAL fluid after acute challenge. For panels (G, H), cells were isolated from lung-draining lymph nodes (LNs) and lung tissues. (G) The levels of memory T-cell proliferation by stimulation of OVA in LN cells from each mouse. For (H), isolated cells were incubated with PBS or CD3 and CD28 antibodies for 12 hours. Levels of individual cytokine were evaluated in supernatant fraction. (H) Levels of IL-4, IL-10, IL-17, and IFN-γ from lung and lung-draining LN cells.
ACKNOWLEDGMENTS
We thank Soo-Jeong Lee and Seo-Yoon Lim for their providing secretarial assistance, and the members of the POSTECH animal facility for their experimental expertise. This study was supported by grants from the Korea Ministry of Health & Welfare, Republic of Korea (HI 13C 0040-010013 & HI14C2628), RP-Grant 2014 of Ewha Womans University and Ewha Womans University Research Grant of 2014.
The authors made the following contributions to the study. Study concept and design: Choi JP, Lee SM, Kim YK; acquisition of data: Choi JP, Lee SM, Jeon SG; analysis and interpretation of data: Choi JP, Lee SM, Kim MH, Jee YK, Cho YJ, Kim YK; drafting of the manuscript: Choi JP, Lee SM, Kim MH, Jee YK, Yang S, Kim YK; statistical analysis: Choi JP, Lee SM; obtained funding: Kim YK.
References
1. Dowse GK, Turner KJ, Stewart GA, Alpers MP, Woolcock AJ. The association between Dermatophagoides mites and the increasing prevalence of asthma in village communities within the Papua New Guinea highlands. J Allergy Clin Immunol. 1985; 75:75–83.
2. Charpin D, Birnbaum J, Haddi E, Genard G, Lanteaume A, Toumi M, et al. Altitude and allergy to house-dust mites. A paradigm of the influence of environmental exposure on allergic sensitization. Am Rev Respir Dis. 1991; 143:983–986.
3. Platts-Mills TA, Vervloet D, Thomas WR, Aalberse RC, Chapman MD. Indoor allergens and asthma: report of the Third International Workshop. J Allergy Clin Immunol. 1997; 100:S2–S24.
4. Neville AC, Parry DA, Woodhead-Galloway J. The chitin crystallite in arthropod cuticle. J Cell Sci. 1976; 21:73–82.
5. Fuhrman JA, Piessens WF. Chitin synthesis and sheath morphogenesis in Brugia malayi microfilariae. Mol Biochem Parasitol. 1985; 17:93–104.
6. Araujo AC, Souto-Padrón T, de Souza W. Cytochemical localization of carbohydrate residues in microfilariae of Wuchereria bancrofti and Brugia malayi. J Histochem Cytochem. 1993; 41:571–578.
7. Debono M, Gordee RS. Antibiotics that inhibit fungal cell wall development. Annu Rev Microbiol. 1994; 48:471–497.
8. Jeong KY, Park JW, Hong CS. House dust mite allergy in Korea: the most important inhalant allergen in current and future. Allergy Asthma Immunol Res. 2012; 4:313–325.
9. Gao YF, Wang de Y, Ong TC, Tay SL, Yap KH, Chew FT. Identification and characterization of a novel allergen from Blomia tropicalis: Blo t 21. J Allergy Clin Immunol. 2007; 120:105–112.
10. Da Silva CA, Pochard P, Lee CG, Elias JA. Chitin particles are multifaceted immune adjuvants. Am J Respir Crit Care Med. 2010; 182:1482–1491.
11. Shibata Y, Foster LA, Bradfield JF, Myrvik QN. Oral administration of chitin down-regulates serum IgE levels and lung eosinophilia in the allergic mouse. J Immunol. 2000; 164:1314–1321.
12. Strong P, Clark H, Reid K. Intranasal application of chitin microparticles down-regulates symptoms of allergic hypersensitivity to Dermatophagoides pteronyssinus and Aspergillus fumigatus in murine models of allergy. Clin Exp Allergy. 2002; 32:1794–1800.
13. Medzhitov R, Janeway C Jr. Innate immunity. N Engl J Med. 2000; 343:338–344.
14. Duez C, Gosset P, Tonnel AB. Dendritic cells and toll-like receptors in allergy and asthma. Eur J Dermatol. 2006; 16:12–16.
15. Kim YK, Oh SY, Jeon SG, Park HW, Lee SY, Chun EY, et al. Airway exposure levels of lipopolysaccharide determine type 1 versus type 2 experimental asthma. J Immunol. 2007; 178:5375–5382.
16. Choi JP, Kim YS, Kim OY, Kim YM, Jeon SG, Roh TY, et al. TNF-alpha is a key mediator in the development of Th2 cell response to inhaled allergens induced by a viral PAMP double-stranded RNA. Allergy. 2012; 67:1138–1148.
17. Kawai T, Akira S. The role of pattern-recognition receptors in innate immunity: update on Toll-like receptors. Nat Immunol. 2010; 11:373–384.
18. Akira S, Uematsu S, Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006; 124:783–801.
19. Choi JP, Kim YS, Tae YM, Choi EJ, Hong BS, Jeon SG, et al. A viral PAMP double-stranded RNA induces allergen-specific Th17 cell response in the airways which is dependent on VEGF and IL-6. Allergy. 2010; 65:1322–1330.
20. Jeon SG, Oh SY, Park HK, Kim YS, Shim EJ, Lee HS, et al. TH2 and TH1 lung inflammation induced by airway allergen sensitization with low and high doses of double-stranded RNA. J Allergy Clin Immunol. 2007; 120:803–812.
21. Fuchs B, Braun A. Improved mouse models of allergy and allergic asthma--chances beyond ovalbumin. Curr Drug Targets. 2008; 9:495–502.
22. Kumar RK, Herbert C, Foster PS. The "classical" ovalbumin challenge model of asthma in mice. Curr Drug Targets. 2008; 9:485–494.
23. Yamashita M, Nakayama T. Progress in allergy signal research on mast cells: regulation of allergic airway inflammation through toll-like receptor 4-mediated modification of mast cell function. J Pharmacol Sci. 2008; 106:332–335.
24. Fattouh R, Pouladi MA, Alvarez D, Johnson JR, Walker TD, Goncharova S, et al. House dust mite facilitates ovalbumin-specific allergic sensitization and airway inflammation. Am J Respir Crit Care Med. 2005; 172:314–321.
25. Shuhui L, Mok YK, Wong WS. Role of mammalian chitinases in asthma. Int Arch Allergy Immunol. 2009; 149:369–377.
26. Reese TA, Liang HE, Tager AM, Luster AD, Van Rooijen N, Voehringer D, et al. Chitin induces accumulation in tissue of innate immune cells associated with allergy. Nature. 2007; 447:92–96.
27. Shibata Y, Foster LA, Metzger WJ, Myrvik QN. Alveolar macrophage priming by intravenous administration of chitin particles, polymers of N-acetyl-D-glucosamine, in mice. Infect Immun. 1997; 65:1734–1741.
28. Da Silva CA, Chalouni C, Williams A, Hartl D, Lee CG, Elias JA. Chitin is a size-dependent regulator of macrophage TNF and IL-10 production. J Immunol. 2009; 182:3573–3582.
29. Da Silva CA, Hartl D, Liu W, Lee CG, Elias JA. TLR-2 and IL-17A in chitin-induced macrophage activation and acute inflammation. J Immunol. 2008; 181:4279–4286.
30. Chen Q, Davidson TS, Huter EN, Shevach EM. Engagement of TLR2 does not reverse the suppressor function of mouse regulatory T cells, but promotes their survival. J Immunol. 2009; 183:4458–4466.
31. Choi JP, Kim YM, Choi HI, Choi SJ, Park HT, Lee WH, et al. An important role of tumor necrosis factor receptor-2 on natural killer T cells on the development of dsRNA-enhanced Th2 cell response to inhaled allergens. Allergy. 2014; 69:186–198.
32. Biswas SK, Mantovani A. Macrophage plasticity and interaction with lymphocyte subsets: cancer as a paradigm. Nat Immunol. 2010; 11:889–896.
SUPPLEMENTARY MATERIAL
Supplementary Fig. 2
Effect of chitinase on the development of airway inflammation and cytokine production induced by HDM-derived chitin. For all panels, wild-type mice (C57BL/6 background) were sensitized with 75 µg of OVA+100 µg of HDM with or without chitinase, and evaluated 24 hours after the last OVA challenge. Data are presented as the mean±SEM (n=5 mice per group) are from a single experiment representative of at least two independent experiments. *P<0.05; **P<0.01; ***P<0.001 relative to each control groups, OVA or OVA+Chitinase, respectively; #P<0.05; ##P<0.01; ###P<0.001. Bars in cytokine graphs mean sensitivity limitation. (A) Bronchoalveolar lavage (BAL) cellularity level after sensitization. (B) Production of IL-1β, TNF-α, IL-4, TSLP, IL-6, IL-10, IL-12p70, and IL-23 in BAL fluid after sensitization.
Supplementary Fig. 3
Immune responses of Th2, Th17, and Th1 induced by airway sensitization with ovalbumin (OVA) plus house dust mite (HDM) derived chitin are not mediated by toll-like receptor (TLR) 1, TLR 4, or TLR 6. For all panels, TLR-deficient mice and wild-type mice (C57BL/6 background) were sensitized with 75 µg of OVA, or 75 µg of OVA+100 µg of HDM derived chitin, and then evaluated 12 hours after the sensitization. *P<0.05 relative to wild-type OVA group; n.s, not significant. Data are presented as the mean±SEM (n=5 mice per group). Bars in cytokine graphs mean sensitivity limitation. (A) Bronchoalveolar lavage (BAL) cellularity after sensitization. (B) Levels of IL-1β, IL-4, IL-5, IL-6, IL-10, IL-12p70, IL-13, IL-23, TNF-α, and TSLP in BAL fluid after sensitization. +, sensitized; -, not sensitized.
Supplementary Fig. 4
Size alteration after chitinase treatment. Chitinase was added to HDM derived chitin and incubated for 1 hour in 37℃. The reaction was done in 1 mL of filtered PBS, and stopped by heating step (100℃, 10 minutes). After enzyme digestion, 2 L of distilled water was added for size analysis. Particle size was evaluated by Mastersizer 2000 (Malvern, Worcestershire, UK).
Supplementary Fig. 5
HDM derived chitin induce inflammatory cytokines in lung epithelial cell and macrophages. MLE and MH-S cells were stimulated with isolated chitin (100 µg/mL) and levels of IL-6 and TNF-α were measured in culture supernatants. 5×105 cells were incubated in DMEM or RPMI 1640 media on 24 well plate. Cells were stimulated during 24 hours and then supernatants were harvested and evaluated. ***P<0.001 relative to wild-type group. Bars in cytokine graphs mean sensitivity limitation.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download