Abstract
Drug-induced hypersensitivity pneumonitis results from interactions between pharmacologic agents and the human immune system. We describe a 54-year-old man with hypersensitivity pneumonitis caused by cephalosporins with identical R1 side chains. The patient, who complained of cough with sputum, was prescribed ceftriaxone and clarithromycin at a local clinic. The following day, he complained of dyspnea, and chest X-ray revealed worsening of inflammation. Upon admission to our hospital, antibiotics were changed to cefepime with levofloxacin, but his pneumonia appeared to progress. Changing antibiotics to meropenem with ciprofloxacin improved his symptoms and radiologic findings. Antibiotics were de-escalated to ceftazidime with levofloxacin, and his condition improved. During later treatment, he was mistakenly prescribed cefotaxime, which led to nausea, vomiting, dyspnea and fever, and indications of pneumonitis on chest X-ray. We performed bronchoalveolar lavage, and the findings included lymphocytosis (23%), eosinophilia (17%), and a low cluster of differentiation (CD) 4 to CD8 ratio (0.1), informing a diagnosis of drug-induced pneumonitis. After a medication change, his symptoms improved and he was discharged. One year later, he was hospitalized for acute respiratory distress syndrome following treatment with ceftriaxone and aminoglycosides for an upper respiratory tract infection. After steroid therapy, he recovered completely. In this patient, hypersensitivity reaction in the lungs was caused by ceftriaxone, cefotaxime, and cefepime, but not by ceftazidime, indicating that the patient's hypersensitivity pneumonitis was to the common R1 side chain of the cephalosporins.
Drug-induced interstitial lung diseases have many clinical patterns, ranging from benign infiltrates to acute respiratory distress syndrome.1 These diseases can involve responses by the oxidant/antioxidant, immunologic, matrix repair, proteolytic or central nervous system.2 Drug-induced hypersensitivity pneumonitis (HP) results from interactions between pharmacologic agents and the immune system. Drug-induced HP can be difficult to diagnose, as it requires ruling out all other possible causes. Although discontinuing the offending drugs can improve symptoms, it is difficult to determine if pulmonary infiltration is related to medication, and failure to appreciate the relationship between the drugs and pulmonary infiltration can lead to death. Several drugs, for instance cyclophosphamide, sulfonamides, or nonsteroidal anti-inflammatory drugs, have been reported to induce HP.345
Cephalosporins have a wide range of chemical effects, and have become more widely used in recent years,6 leading to an increase in reports of adverse reactions. Although adverse reactions to cephalosporins are well-studied, few reports of HP by cephalosporins exist in Japan.78 Herein, we present the first case of HP induced by cephalosporins with identical side chains.
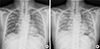
A 54-year-old male was referred to our hospital due to dyspnea on February 16, 2013. He first visited a local medical clinic complaining of cough, rhinorrhea, and fever. He was prescribed antibiotics and anti-inflammatory medications. Several days later, he was admitted to the local clinic due to shortness of breath, coughing, and fever. Although he was prescribed ceftriaxone and clarithromycin, his symptoms and radiologic findings worsened upon admission (Fig. 1A, C), so he was transferred to our hospital.
The patient had a 10-pack years of smoking, and no other history of medical conditions or relevant environmental exposure. At first admission, his blood pressure was 120/70 mmHg with a regular heart rate of 73 beats/min and a body temperature of 36.4℃. He was given 5 L of oxygen, as his peripheral oxygen saturation was 79%. Laboratory tests revealed a white blood cell count of 13.00×103 cells/µL (75.9% neutrophils, 11.2% lymphocytes, 4.9% eosinophils), a C-reactive protein (CRP) level of 8.14 mg/dL, and a procalcitonin level of 1.21 ng/mL. Chest X-ray and computed tomography (CT) revealed diffuse ground glass opacities, consolidation, and a "crazy-paving" pattern in both lungs (Fig. 1B, D). CT also indicated enlargement of the paraaortic, right upper paratracheal, and right hilar lymph nodes. Because these findings were consistent with pneumonia, antibiotics (cefepime and levofloxacin) were administered intravenously. Blood culture, sputum analysis, viral profiling, and assays for Streptococcus pneumoniae urinary antigen, Mycoplasma pneumoniae IgM, Chlamydia pneumoniae antibody, and Pneumocystitis jirovecii DNA failed to reveal any organism. Lack of clinical improvement until 6 days after admission led us to change antibiotics to meropenem and ciprofloxacin. Since then his symptoms and radiologic findings were improving (Fig. 2A). As culture failed to isolate any organisms, antibiotics were de-escalated to ceftazidime with levofloxacin with further radiologic improvement.
During treatment, the doctor mistakenly prescribed cefotaxime instead of ceftazidime, at which time the patients complained of nausea, vomiting, dyspnea, and fever. A chest X-ray showed redeveloped opacities (Fig. 2B), and analysis of bronchoalveolar lavage (BAL) revealed lymphocytosis (23%), a low cluster of differentiation (CD) 4 to CD8 ratio (0.1), and an increase in neutrophils (10%) and eosinophils (17%), indicating HP in response to cefotaxime. Three days after switching medication, his symptoms improved and he was discharged. One year later, the patient was again referred to our hospital due to dyspnea, fever, and cough. One day before admission, he had been prescribed ceftriaxone with aminoglycoside to treat an upper respiratory tract infection at a local medical clinic. Radiologic findings indicated inflammation even at this early point after drug exposure (Fig. 3). Arterial oxygen pressure/fraction of inspired oxygen (PaO2/FiO2) was 83. We diagnosed his pulmonary abnormality as HP in response to ceftriaxone on the basis of rapid progression of radiologic indications and previous history of HP following treatment with cefotaxime. Mechanical ventilation was applied, and the antibiotics of meropenem and ciprofloxacin, as well as immunoglogulin and methylprednisolone, were administered. Three days after steroid therapy, his symptoms and radiologic findings improved. He was discharged 14 days after admission, and has been doing well without other symptoms. In this patient, lung hypersensitivity was caused by ceftriaxone, cefotaxime, and cefepime, but not by ceftazidime.
Here, we report a case of hypersensitivity pneumonia caused by several cephalosporins with identical R1 side chains. Our patient repetitively suffered from pneumonitis after taking cephalosporins; namely, ceftriaxone, cefotaxime, and cefepime, but not ceftazidime. Chest X-ray and CT showed diffuse ground glass opacities and a "crazy-paving" pattern in both lungs. Also, BAL fluid contained increased lymphocytes and eosinophils with a decreased CD4 to CD8 ratio. After steroid therapy, the patient completely recovered.
HP is a lymphocytic allergic response throughout the lungs. In general, it results from exposure to airborne organic antigens, such as mold dust, microorganisms, or occupational dust,9 but the term can also be applied to lung inflammation and fibrosis caused by drugs.10 The disease can present in acute, subacute, or chronic forms, but these frequently overlap.11 In the acute phase, immune complexes initiate the complement cascade and activate alveolar macrophages.10 Cytokines produced by activated macrophages promote maturation of CD8+ cells into cytotoxic cells.
Diagnosing HP is difficult because its symptoms and clinical presentation are nonspecific.10 Laboratory analyses are unhelpful, and radiographic findings vary according to stage. The acute stage is usually characterized by micronodular infiltrates, patchy ground-glass opacities, and mosaic perfusion,12 whereas subacute or chronic HP appears as reticulonodular infiltrates, interstitial fibrosis, or honeycombing. BAL fluid analysis is useful for detecting lymphocytic infiltration of the lung, especially CD8+ T cell lymphocytosis (a low CD4+ to CD8+ ratio).11 However, BAL findings vary according to clinical stage and antigen.1113 As the disease progresses to the chronic stage, the ratio of CD4+ to CD8+ T cells may increase.13 Histologic findings include infiltration of the interstitial space by lymphocytes and plasma cells, and sometimes non-caseating granulomas. The typical findings of an alveolar lymphocytosis in BAL fluid and bilateral ground glass opacities on chest CT would confirm the diagnosis of HP without surgical lung biopsy.14
The drug-induced lymphocyte stimulating test (DLST) and leukocyte migration test (LMT) are capable of detecting drug-sensitized T cells. Especially LMT had a higher positive response rate than the DLST for skin eruptions and hepatic injury.15 However, these tests lack sensitivity and specificity, and thus do not have a definitive role in the diagnosis of drug-induced lung disease.1
Our case presented with respiratory symptoms, including breathlessness, fever, and cough. Chest CT showed diffuse ground glass opacities, consolidation, and a "crazy-paving" pattern. BAL fluid showed increased lymphocytes and eosinophils, and a decreased ratio of CD4+ to CD8+ T cells. Although the lung was not biopsied, clinical and radiologic findings and BAL analyses were compatible with drug-induced pneumonitis.116
Clinical course of the case indicated HP in response to cephalosporins with identical R1 side chains. Cephalosporins consist of a four-member beta-lactam ring; their variety results from substitution in the R1 and R2 side chains.17 Hypersensitivity to beta-lactam antibiotics can be induced by the beta-lactam ring structure (Fig. 4A), but there is also evidence that the R1 side chain contributes to the specificity of immunologic reactions to cephalosporins,18 suggesting that cross-reactions between cephalosporins may occur via identical R1 recognition.6 Cefotaxime, ceftriaxone, cefepime, and cefuroxime have identical R1 side chains, whereas that of ceftazidime differs slightly (Fig. 4B, C).19 Our patient's symptoms were aggravated when ceftriaoxone, cefotaxime, or cefepime, but not ceftazidime, were administered. Therefore, we diagnosed HP due to cephalosporins with identical R1 side chains. Although some studies have reported that cephalosporins, such as ceftazidime and ceftizoxime, can induce pneumonitis,78 and cephalosporins with identical side R1 chains can trigger immediate reactions such as urticaria or anaphylaxis,20 HP in a single patient in response to different cephalosporins with identical R1 side chains has not been reported.
The best treatment of HP is early diagnosis and avoidance of further exposure, and the only accepted pharmacologic therapy is systemic corticosteroid, which can hasten rapid recovery; however, its long-term efficacy has not been demonstrated.11 In our case, at first admission, avoidance of drug was enough to improve clinical course without systemic corticosteroids. In contrast, upon second admission, laboratory findings and chest CT indicated an acute respiratory distress syndrome, so corticosteroids were administered intravenously, which rapidly improved symptoms and radiologic findings.
Although drugs can induce HP, drugs are rarely suspected as a cause of pulmonary abnormality. Nonetheless, this case illustrates the importance of considering HP when a patient presents with compatible respiratory symptoms, radiologic findings, and BAL analysis. Early diagnosis and avoidance of causative agents are essential for avoiding progression to lung fibrosis. Because cephalosporin R1 side chains can induce hypersensitivity reactions, drugs with identical R1 side chains should be avoided for the treatment of patients with suspected cephalosporin-induced HP.
Figures and Tables
ACKNOWLEDGMENTS
This research was supported by a grant from the Ministry of Food and Drug Safety to operate the regional pharmacovigilance center in 2014.
References
1. Matsuno O. Drug-induced interstitial lung disease: mechanisms and best diagnostic approaches. Respir Res. 2012; 13:39.
2. Cooper JA Jr, Matthay RA. Drug-induced pulmonary disease. Dis Mon. 1987; 33:61–120.
3. Mark GJ, Lehimgar-Zadeh A, Ragsdale BD. Cyclophosphamide pneumonitis. Thorax. 1978; 33:89–93.
4. Fujimori K, Yokoyama A, Kurita Y, Uno K, Saijo N. Paclitaxel-induced cell-mediated hypersensitivity pneumonitis. Diagnosis using leukocyte migration test, bronchoalveolar lavage and transbronchial lung biopsy. Oncology. 1998; 55:340–344.
5. Tohyama M, Tamaki Y, Toyama M, Ishimine T, Miyazato A, Nakamoto A, et al. A case of loxoprofen-induced pneumonitis pathologically resembling hypersensitivity pneumonitis. Nihon Kokyuki Gakkai Zasshi. 2002; 40:123–128.
6. Baldo BA. Penicillins and cephalosporins as allergens--structural aspects of recognition and cross-reactions. Clin Exp Allergy. 1999; 29:744–749.
7. Suzuki K, Inagaki T, Adachi S, Matsuura T, Yamamoto T. A case of ceftazidime-induced pneumonitis. Nihon Kyobu Shikkan Gakkai Zasshi. 1993; 31:512–516.
8. Suzuki K, Yamamoto K, Kishimoto A, Hayakawa T, Yamamoto T. A case of ceftizoxime-induced pneumonitis. Nihon Kyobu Shikkan Gakkai Zasshi. 1985; 23:1357–1361.
9. Malmberg P, Rask-Andersen A, Rosenhall L. Exposure to microorganisms associated with allergic alveolitis and febrile reactions to mold dust in farmers. Chest. 1993; 103:1202–1209.
10. Patel AM, Ryu JH, Reed CE. Hypersensitivity pneumonitis: current concepts and future questions. J Allergy Clin Immunol. 2001; 108:661–670.
11. Selman M, Pardo A, King TE Jr. Hypersensitivity pneumonitis: insights in diagnosis and pathobiology. Am J Respir Crit Care Med. 2012; 186:314–324.
12. Cormier Y, Brown M, Worthy S, Racine G, Müller NL. High-resolution computed tomographic characteristics in acute farmer's lung and in its follow-up. Eur Respir J. 2000; 16:56–60.
13. Barrera L, Mendoza F, Zuñiga J, Estrada A, Zamora AC, Melendro EI, et al. Functional diversity of T-cell subpopulations in subacute and chronic hypersensitivity pneumonitis. Am J Respir Crit Care Med. 2008; 177:44–55.
14. Lacasse Y, Selman M, Costabel U, Dalphin JC, Ando M, Morell F, et al. Clinical diagnosis of hypersensitivity pneumonitis. Am J Respir Crit Care Med. 2003; 168:952–958.
15. Saito M, Yagi M, Uno K, Takanaka K. Comparative study of the usefulness of the drug-induced lymphocyte stimulation test and the leukocyte migration test in drug allergies. Biol Pharm Bull. 2008; 31:299–304.
16. Akoun GM, Cadranel JL, Milleron BJ, D'Ortho MP, Mayaud CM. Bronchoalveolar lavage cell data in 19 patients with drug-associated pneumonitis (except amiodarone). Chest. 1991; 99:98–104.
17. Perez-Inestrosa E, Suau R, Montañez MI, Rodriguez R, Mayorga C, Torres MJ, et al. Cephalosporin chemical reactivity and its immunological implications. Curr Opin Allergy Clin Immunol. 2005; 5:323–330.
18. Sánchez-Sancho F, Perez-Inestrosa E, Suau R, Montañez MI, Mayorga C, Torres MJ, et al. Synthesis, characterization and immunochemical evaluation of cephalosporin antigenic determinants. J Mol Recognit. 2003; 16:148–156.
19. Pichichero ME, Zagursky R. Penicillin and cephalosporin allergy. Ann Allergy Asthma Immunol. 2014; 112:404–412.
20. Antunez C, Blanca-Lopez N, Torres MJ, Mayorga C, Perez-Inestrosa E, Montañez MI, et al. Immediate allergic reactions to cephalosporins: evaluation of cross-reactivity with a panel of penicillins and cephalosporins. J Allergy Clin Immunol. 2006; 117:404–410.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download