Abstract
Purpose
Mycoplasma pneumoniae (M. pneumoniae) is one of the most common causes of community-acquired pneumonia in children. The clinical course is typically self-limited and benign; however, rare cases of severe pneumonia can develop despite appropriate antibiotic therapy. We studied the effects of methylprednisolone pulse therapy on severe refractory M. pneumoniae pneumonia in children.
Methods
The clinical effects of methylprednisolone therapy were evaluated retrospectively in 12 children with severe refractory M. pneumoniae pneumonia, which was diagnosed serologically. All patients developed respiratory distress, high fever, and initial lobar pneumonic consolidation based on radiological findings. All clinical symptoms deteriorated despite appropriate antibiotic therapy. Thus, children were treated with intravenous methylprednisolone pulse therapy in addition to antibiotics.
Results
The average febrile period before admission was 4.9±1.7 days, and fever persisted in all children until steroid administration. Methylprednisolone pulse therapy (30 mg/kg) was given 5.4±2.5 days after admission. After methylprednisolone pulse therapy, clinical symptoms improved in all patients without adverse events. The fever subsided 0-2 h after initiation of corticosteroid therapy. The abnormal radiological findings resolved within 2.6±1.3 days, and the high C-reactive protein levels (6.7±5.9 mg/dL) on admission decreased to 1.3±1.7 mg/dL within 3.0±1.1 days after starting corticosteroid therapy.
M. pneumoniae accounts for 10%-40% of community-acquired pneumonia in school-aged children and young adults.1-4 Although M. pneumoniae pneumonia (MP) typically follows a benign course, it may develop into severe fulminant pneumonia and pulmonary complications such as acute respiratory distress syndrome, necrotizing pneumonia, or bronchiolitis obliterans in rare cases.5 In Korea, the epidemic incidence of M. pneumoniae infections, which formerly peaked at 4-7-year intervals in school-aged children, now occurs every 3-4 years at a high prevalence. In addition, the age of children with MP has decreased and more severe symptoms have presented in older children during the past decade.6,7 In 2011, MP was more prevalent throughout the country and caused more severe clinical features than at any other time, which may be associated with macrolide-resistant M. pneumoniae (unpublished observation).
Macrolides are generally recognized as first-choice agents with excellent effectiveness against M. pneumoniae infection3. However, severe pneumonia can develop despite appropriate antibiotic therapy. Although the exact mechanism of progression remains unclear, several have been proposed. For example, patients may develop antibiotic resistance8 or show an excessive immune response against pathogens, such as vigorous expression of cytokines and highly activated cell-mediated immune responses.9 Clinical studies have shown that systemic corticosteroid treatment temporarily improved the clinical symptoms of MP in children,10-12 which is suggestive of an overreaction of host immune systems. However, effects, optimal dose and timing of systemic corticosteroid treatment remain unclear. In the present study, we examined clinical signs and symptoms, as well as laboratory and radiological findings, to evaluate the impact of high-dose systemic corticosteroid administration concomitant with antibiotics on refractory MP in children.
We retrospectively reviewed the medical records of 12 previously healthy children with refractory MP who were treated with high doses of methylprednisolone therapy at Chungnam National University Hospital (Daejeon, Korea) between January 2011 and December 2011. Vital signs of all patients were checked every 2-4 h daily. Laboratory tests including white blood cell counts, serum aminotransferase and CRP, as well as radiological examinations were determined upon admission and followed up over time. M. pneumoniae infection was confirmed based on detection of M. pneumonia IgM using an indirect enzyme-linked immunosorbent assay (ELISA, DIESSE Diagnostica Senese, Siena, Italy). The test was performed upon admission, and the positive cutoff value of IgM was established as >1.1 according to the manufacturer's instructions. The cold agglutinin titers of paired serum samples were measured at specific intervals after admission and a positive result was defined as an initial cold agglutinin titer of 1:64 or a fourfold rising titer in double-checked samples. To exclude other respiratory infections, we performed microbiological testing of blood and sputum cultures and reverse transcription-polymerase chain reaction (RT-PCR) analysis of several respiratory viruses-influenza viruses, parainfluenza viruses, respiratory syncytial viruses, coronaviruses, human rhinoviruses, adenoviruses and metapneumoviruses-before antibiotic therapy. None of the tests resulted in detection of any other pathogens. All children with severe MP showed clinically high fever, respiratory distress, or initial lobar pneumonic consolidation with or without pleural effusion. All patients were treated initially with macrolides (clarithromycin or roxithromycin) or beta-lactam antibiotics (ampicillin with sulbactam or ceftriaxone). Since symptoms were aggravated despite antibiotic therapy, methylprednisolone was administered intravenously at a dose of 30 mg/kg once daily for 3 consecutive days. Refractory pneumonia was defined as follows: 1) Prolonged fever for 7 days or more or 2) persistent consolidation of more than one lobe of the lung despite appropriate antibiotic treatment, including macrolides.
Data were analyzed using the SPSS software, version 19 (SPSS Inc., Chicago, IL, USA). The body temperature and CRP concentration during the follow-up were subjected to repeated-measures analysis of variance (ANOVA). Significance was defined as P<0.05.
The study was approved by the Ethical Committee of the Chunganm National University Hospital (No. 2012-11-003-001).
The mean age of the 12 patients (nine females and three males) was 7±2.6 years (range, 3-13 years). The average febrile duration before admission was 4.9±1.7 days (range, 3-8 days), and 7 of the 12 children were treated with oral antibiotics before admission. Upon admission, the mean body temperature was 39.0±0.9℃, pulse rate was 141.3±15.6 beats per minute, and respiratory rate was 35.3±7.9 breaths per minute. All patients showed lobar consolidation in the initial chest radiography, and 10 patients had pleural effusion. Thoracentesis was performed to relieve the respiratory distress symptoms in one patient with moderate pleural effusion, and the pleural fluid was transudate. Based on serological diagnostic tests, three patients had a 1:128 cold agglutinin titer upon admission, while the others had a fourfold rising cold agglutinin titer with a mean interval of 4.3±2.3 days (range, 3-10 days) (Table 1).
All patients responded well to the adjuvant methylprednisolone therapy. The total duration of hospitalization was 11.8±4.3 days (range, 7-20 days), and the duration of hospitalization after methylprednisolone pulse therapy was 6.4±3.3 days (range, 3-12 days) (Table 1). The mean duration of the high fever was 10.3±3.1 days (range, 7-15 days), which subsided within 2 h after methylprednisolone pulse therapy in all patients (Fig 1A, P<0.001). Defervescence was accompanied by improvement in clinical symptoms such as cough, sputum and respiratory distress. Radiological abnormal findings of consolidation and/or pleural effusion also decreased at 2.6±1.3 days (range, 0-7 days) after initiation of methylprednisolone pulse therapy (Fig. 2).
WBC and lymphocyte counts were 7.7±3.0×103/µL and 1.6±0.7×103/µL, respectively, upon admission. After methylprednisolone pulse therapy, these values increased to 9.7±3.1×103/µL and 2.0±0.8×103/µL, respectively, showing clinical improvement. However, these differences were not statistically significant.
The initial serum aminotransferase levels were elevated in four patients, and recovered to normal values after methylprednisolone pulse therapy. The initial mean serum CRP concentrations (6.7±5.9 mg/dL) increased to 10.1±8.5 mg/dL before methylprednisolone therapy (4.3±2.3 days after admission). They also decreased to 1.3±1.6 mg/dL after 3.0±1.1 days of methylprednisolone pulse therapy (Fig 1B, P<0.001).
M. pneumoniae infection is common in children. The clinical courses of MP infection are typically benign and symptoms resolve quickly with antibiotic therapy.13 In rare cases, MP may develop into severe pneumonia and progress to fatal pneumonia with serious complications, despite appropriate antibiotic treatment.5,13 These conditions are known as severe refractory MP.11,14 The pathogenesis of severe refractory M. pneumoniae infections is attributed partly to excessive cell-mediated immunity and cytokine responses against the pathogens.9
For pathogenesis, M. pneumoniae shows strong cytoadherence to the host respiratory epithelium. The cytoadherence of M. pneumoniae initiates immune responses and progression toward excessive inflammation in the late serological phase rather than through an early direct invasion phase, which is often implicated in pulmonary or various extrapulmonary manifestations. However, M. pneumoniae may induce pneumonia through local cellular damage by producing superoxide radicals and initiating the innate immune response, including the secretion of proinflammatory cytokines such as interleukin-1β.9,15 Although cytokines and recruitment of lymphocytes play a role in minimizing lung tissue damage by activating host defenses, the immunological reactions ultimately exacerbate the immunological hypersensitivity and contribute to the development of vigorous cell-mediated immune responses, which can lead to pulmonary injury and induce severe clinical illness.15,16
In our study, all children were healthy prior to M. pneumoniae infection, and infected children were older than 6 years. Moreover, the markedly elevated CRP concentration (6.7±5.9 mg/dL upon admission and 10.1±8.5 mg/dL before initiation of steroid therapy) reflected the acute severe systemic inflammatory reactions to M. pneumoniae infection, and was suggestive of a well-developed immune system. Previous reports associating the host immune status with M. pneumoniae infection showed that the clinical signs and symptoms of MP were more severe in previously healthy young adults, while the clinical manifestations of a relatively immunocompromised host were not as severe.5 These results agreed with animal study models.17 In animal studies, pulmonary lesions were not as severe in cell-mediated immune deficient animals compared to controls.17
Since pathogenesis of severe M. pneumoniae infection is closely related to an excessive, activated cell-mediated immune response to pathogens, immune-suppressive therapy may be valuable. Although the effect of steroids on severe MP is controversial, some reports have shown that temporary steroid therapy results in clinical improvement in children with severe MP.10-12 Animal experiments showed that combined treatment of clarithromycin and dexamethasone reduced the lung histopathological score and cytokine levels more than either treatment alone.18 Steroids can exert immune regulatory and anti-inflammatory effects through several molecular mechanisms, such as inhibition of nuclear factor κB, and also influence natural lung immunity by regulating alveolar macrophages and neutrophils.19,20
In our study, all children showed serious clinical features upon admission, possibly caused by excessive inflammatory responses. The symptoms of respiratory distress including high fever and lobar pneumonic consolidation with pleural effusion deteriorated and were refractory despite appropriate antibiotic treatment. The deteriorated clinical and radiological findings of severe refractory MP improved rapidly after intravenous methylprednisolone pulse therapy at a dose of 30 mg/kg once daily for 3 days to suppress immune reactions without adverse events. All patients achieved defervescence within 2 h, resolution of radiological findings 2.6±1.3 days after initiation of corticosteroid therapy, and a reduction in CRP levels 3.0±1.1 days after initiation of methylprednisolone pulse therapy.
Although the clinical effects of oral prednisolone and intravenous methylprednisolone therapy on severe MP have been evaluated,10,11,20 disease severity and treatment protocols remain unclear, and treatment guidelines for corticosteroid therapy have not been established. Thus, the dosage and duration of corticosteroid therapy should be modified according to severity to effectively treat refractory MP.
Since macrolide such as erythromycin resistance of MP was first reported in Japan in 1970,21 the prevalence of macrolide-resistant M. pneumoniae has increased to 10%-30% worldwide.8,22,23 The macrolide-resistant strains are known to cause severe refractory MP.8 The M144V mutation of the L4 protein and mutation at position 2064 of domain V in the 23S rRNA gene of M. pneumoniae was reported in 2010 in Korea, and the appearance of macrolide-resistant strains has also increased recently.24 Although we did not perform an antimicrobial susceptibility test in this study, the unresponsiveness to antibiotics in these patients was likely associated with macrolide resistance.
In conclusion, we report on the management of methylprednisolone pulse therapy, which was associated with a good outcome in children with severe refractory MP. This study suggests that excessive host immune responses to M. pneumonia infection can lead to severe pulmonary injury and high dose corticosteroid therapy can effectively treat severe refractory MP in children. A larger prospective study is required to evaluate the mechanisms and define the benefits of corticosteroids for the treatment of severe refractory MP.
Figures and Tables
Fig. 1
Defervescence was observed in all children after methylprednisolone pulse therapy. (A) Body temperature data were analyzed using repeated-measures ANOVA (P<0.001). (B) Decreasing serum CRP levels were observed in all children after methylprednisolone pulse therapy. CRP concentrations were analyzed using repeated-measures ANOVA (P<0.001). *The one day before admission. †The day on admission. ‡The first day of admission. §The second day of admission. ∥The third day of admission. ¶The 4.3±2.3 day of admission (before steroid pulse therapy). **The 8.3±2.0 day of admission (after steroid pulse therapy). CRP, C-reactive protein; HD, hospital day.
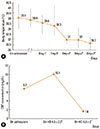
Fig. 2
Chest radiographs of patient 4. (A) Chest radiography upon admission showed consolidation of the left lower lobe with pleural effusion. (B) Before methylprednisolone pulse therapy, the radiographic findings remained unchanged with deteriorated clinical signs at hospital day 3. (C) Chest radiography showed resolution of consolidation of the left lower lobe and decreased pleural effusion on the day after initiation of methylprednisolone pulse therapy at hospital day 4. (D) hospital day 7.
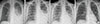
Table 1
Clinical characteristics of patients with refractory severe Mycoplasma pneumoniae pneumonia.

ACKNOWLEDGMENTS
This study was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (2012-0008388).
References
1. Waites KB, Talkington DF. Mycoplasma pneumoniae and its role as a human pathogen. Clin Microbiol Rev. 2004; 17:697–728.
2. Atkinson TP, Balish MF, Waites KB. Epidemiology, clinical manifestations, pathogenesis and laboratory detection of Mycoplasma pneumoniae infections. FEMS Microbiol Rev. 2008; 32:956–973.
3. Heiskanen-Kosma T, Korppi M, Jokinen C, Kurki S, Heiskanen L, Juvonen H, Kallinen S, Stén M, Tarkiainen A, Rönnberg PR, Kleemola M, Mäkelä PH, Leinonen M. Etiology of childhood pneumonia: serologic results of a prospective, population-based study. Pediatr Infect Dis J. 1998; 17:986–991.
4. Sinaniotis CA, Sinaniotis AC. Community-acquired pneumonia in children. Curr Opin Pulm Med. 2005; 11:218–225.
5. Chan ED, Welsh CH. Fulminant Mycoplasma pneumoniae pneumonia. West J Med. 1995; 162:133–142.
6. Lee KY. Pediatric respiratory infections by Mycoplasma pneumoniae. Expert Rev Anti Infect Ther. 2008; 6:509–521.
7. Lee SH, Noh SM, Lee KY, Lee HS, Hong JH, Lee MH, Lee JS, Lee BC. Clinico-epidemiologic study of Mycoplasma pneumoniae pneumonia (1993 through 2003). Korean J Pediatr. 2005; 48:154–157.
8. Morozumi M, Takahashi T, Ubukata K. Macrolide-resistant Mycoplasma pneumoniae: characteristics of isolates and clinical aspects of community-acquired pneumonia. J Infect Chemother. 2010; 16:78–86.
9. Shimizu T, Kida Y, Kuwano K. Cytoadherence-dependent induction of inflammatory responses by Mycoplasma pneumoniae. Immunology. 2011; 133:51–61.
10. Lu A, Wang L, Zhang X, Zhang M. Combined treatment for child refractory Mycoplasma pneumoniae pneumonia with ciprofloxacin and glucocorticoid. Pediatr Pulmonol. 2011; 46:1093–1097.
11. Tamura A, Matsubara K, Tanaka T, Nigami H, Yura K, Fukaya T. Methylprednisolone pulse therapy for refractory Mycoplasma pneumoniae pneumonia in children. J Infect. 2008; 57:223–228.
12. Lee KY, Lee HS, Hong JH, Lee MH, Lee JS, Burgner D, Lee BC. Role of prednisolone treatment in severe Mycoplasma pneumoniae pneumonia in children. Pediatr Pulmonol. 2006; 41:263–268.
13. Ferwerda A, Moll HA, de Groot R. Respiratory tract infections by Mycoplasma pneumoniae in children: a review of diagnostic and therapeutic measures. Eur J Pediatr. 2001; 160:483–491.
14. Radisic M, Torn A, Gutierrez P, Defranchi HA, Pardo P. Severe acute lung injury caused by Mycoplasma pneumoniae: potential role for steroid pulses in treatment. Clin Infect Dis. 2000; 31:1507–1511.
15. Waites KB, Balish MF, Atkinson TP. New insights into the pathogenesis and detection of Mycoplasma pneumoniae infections. Future Microbiol. 2008; 3:635–648.
16. Yang J, Hooper WC, Phillips DJ, Talkington DF. Cytokines in Mycoplasma pneumoniae infections. Cytokine Growth Factor Rev. 2004; 15:157–168.
17. Denny FW, Taylor-Robinson D, Allison AC. The role of thymus-dependent immunity in Mycoplasma pulmonis infections of mice. J Med Microbiol. 1972; 5:327–336.
18. Tagliabue C, Salvatore CM, Techasaensiri C, Mejias A, Torres JP, Katz K, Gomez AM, Esposito S, Principi N, Hardy RD. The impact of steroids given with macrolide therapy on experimental Mycoplasma pneumoniae respiratory infection. J Infect Dis. 2008; 198:1180–1188.
19. De Pascale G, Bello G, Antonelli M. Steroids in severe pneumonia: a literature review. Minerva Anestesiol. 2011; 77:902–910.
20. Rhen T, Cidlowski JA. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N Engl J Med. 2005; 353:1711–1723.
21. Niitu Y, Hasegawa S, Suetake T, Kubota H, Komatsu S, Horikawa M. Resistance of Mycoplasma pneumoniae to erythromycin and other antibiotics. J Pediatr. 1970; 76:438–443.
22. Okazaki N, Narita M, Yamada S, Izumikawa K, Umetsu M, Kenri T, Sasaki Y, Arakawa Y, Sasaki T. Characteristics of macrolide-resistant Mycoplasma pneumoniae strains isolated from patients and induced with erythromycin in vitro. Microbiol Immunol. 2001; 45:617–620.
23. Bébéar C, Pereyre S, Peuchant O. Mycoplasma pneumoniae: susceptibility and resistance to antibiotics. Future Microbiol. 2011; 6:423–431.
24. Oh CE, Choi EH, Lee HJ. Detection of genetic mutations associated with macrolide resistance of Mycoplasma pneumoniae. Korean J Pediatr. 2010; 53:178–183.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download