Abstract
Purpose
Statins are known as cholesterol-lowering agents, but have been suggested for the treatment of asthma because of their anti-inflammatory effects. In this study, the potential therapeutic effects of atorvastatin were investigated in asthmatic patients.
Methods
A total of 62 patients with persistent mild to moderate asthma who presented at asthma clinics of Arak University of Medical Sciences were recruited in a double-blind randomized clinical trial. The asthma clinical control score was assessed based on the standardized Asthma Control Test. Lung volume, i.e., percentage of forced expiratory volume in one second (FEV1%) and percentage of forced vital capacity (FVC%), and peripheral blood eosinophils were also measured. The intervention group was treated with atorvastatin 40 mg per day for 8 weeks, while the control group received a placebo. Asthma controller treatments were not changed. At the beginning and end of the study, serum cholesterol and triglyceride levels were measured to evaluate adherence of the patients to the treatment.
Results
The asthma control score did not significantly differ between the intervention and control groups (P=0.06). Difference in FEV1%, FVC%, and blood eosinophil count between the intervention and control groups were not statistically significant (P>0.05). The differences in post-treatment cholesterol and low-density lipoprotein cholesterol levels were significant (P<0.05).
Asthma is a global health problem that can severely restrict life activities and can even cause death if not controlled. It affects about 300 million people worldwide, with a mortality rate of about 250,000 annually.1 Despite the availability of effective treatments, some patients develop a type of asthma that is difficult to manage and relatively insensitive to corticosteroids. Although the cost of asthma control seems high for both the patient and society, the cost of untreated asthma is even higher.1 As a result of progress in the understanding of asthma pathophysiology, new targets have been identified for its treatment. One of the drug classes that has recently attracted attention is the statins.2,3
Statins are among the most common medications used in lipid disorders and were originally prescribed to treat cardiovascular diseases4,5; however, they have more effects than just lowering blood cholesterol.6,7 The anti-inflammatory and immunomodulatory effects of statins have been widely described.8-10 They have been shown to decrease serum C-reactive protein Iran(CRP) and tumor necrosis factor-α (TNF-α) levels,10 reduce the expression of cyclooxygenase-2 (Cox-2) and other proinflammatory molecules in animal models,11 directly inhibit the major histocompatibility complex-2 (MHC-2) induction in lymphocyte activation,8 and inhibit the activation, proliferation, and differentiation of T lymphocytes to T helper 1 cells (Th1).12-14 Although the exact mechanisms for the various effects of statins are unknown,15 their use in diseases with an inflammatory basis, including asthma, has been proposed.16
The anti-inflammatory effects of simvastatin in a rat experimental model of allergic asthma sensitized with ovalbumin were shown for the first time in 2004,17 and similar results were reported in 2006.18 In another study, simvastatin was used in patients with mild to moderate asthma, but no anti-inflammatory effect was detected.19 The findings regarding the effects of fluvastatin, atorvastatin, and other statins in asthma management have been controversial.20-22 Several studies using mice sensitized with ovalbumin have suggested that statins may be effective in asthmatic patients,23-25 although the results were inconclusive. For this reason, we investigated the effect of atorvastatin for the treatment of asthmatic patients.
The research protocol was approved by the Scientific Ethics Committee of Arak University of Medical Sciences, and written informed consent was obtained from the volunteers. Patients with mild persistent to moderate persistent asthma (grades 2 and 3 of severity criteria for asthma)26 aged 18 to 70 years who presented at asthma clinics of Arak University of Medical Sciences from 24 October 2010 to 10 April 2011 were included in this double-blind clinical trial. Exclusion criteria included taking statin drugs at the time of the study, a history of sensitivity to statins, severe asthma leading to hospitalization within a month prior to the beginning of the study, smoking, hepatitis or active liver disease, myopathy or myositis, and pregnancy. A sample size of 31 patients in the control and intervention groups was deemed to be sufficient to determine a 5-degree increase in the Asthma Control Test (ACT) score, based on an alpha of 0.05 and 90% power. Volunteer patients were alternately allocated to the intervention and control groups based on the time of entering the study. A 24-hour phone line was provided to report any side effects of the drug or asthma exacerbation.
All patients completed a standardized ACT questionnaire27 upon entering the study, followed by a spirometry test (Desktop, Pony FX, Italy). A peripheral blood sample was taken to determine blood eosinophil counts, and serum low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), total cholesterol, and triglyceride (TG) levels. The intervention group was treated using 40-mg atorvastatin tablets (Sobhan Pharmaceutical Company, Iran) once daily for 8 weeks, while the control group received a placebo. The placebo tablets, prepared by the Food and Drug Department of Arak University of Medical Sciences, were composed of starch and were similar to the atorvastatin tablets in size, shape, and color. The drug and placebo tablets were administered to patients by a staff member. Asthma controller treatments were not changed. Improvement in the degree of clinical control of asthma symptoms based on the ACT score was the primary outcome. The improvement in lung volume, which was quantified as the percentage of forced expiratory volume in one second (FEV1%) and the percentage of forced vital capacity (FVC%), and the reduction in the number of peripheral blood eosinophils after atorvastatin treatment compared with placebo were considered secondary outcomes.
Serum cholesterol and TG measurements were used to evaluate adherence to the treatment. Four weeks after treatment initiation, the ACT questionnaire was re-administered, and the patients were visited to assess possible side effects of the drug and evaluate the asthma control level. Finally, after 8 weeks of treatment, the patients completed the ACT questionnaire for the third time, and a spirometry test was performed. A further peripheral blood sample was collected to determine changes in peripheral blood eosinophil counts and serum cholesterol and TG levels. The data were analyzed using the statistical software SPSS version 16.0 (SPSS Inc., Chicago, IL, USA) and charts were plotted using Excel software (Microsoft, USA). Both independent and dependent samples t-tests were used for comparison of the groups and subgroups. Student's t-test and the chi-squared test were used as appropriate to compare the treatment and the control groups. The significance level was set at P<0.05.
A total of 73 patients initially agreed to participate. Eleven voluntarily left the study, and five were excluded due to complications of the disease or asthma exacerbation (Fig. 1). No mortality occurred during the study period. Demographic information of the patients is summarized in Table 1. As shown in Table 2, the improvement in the ACT score was not significantly different between the intervention and control groups (P=0.06). In terms of the secondary outcomes, there were differences in FEV1%, FVC%, and blood eosinophil counts, but the differences between the intervention and control groups was not significant (P>0.05). Although the changes in TG and HDL-C levels between the intervention and control groups were not significant (P>0.05), the post-treatment total cholesterol and LDL-C levels differed significantly between the two groups (P=0.004 and P=0.001, respectively) (Figs. 2 and 3).
In this study, our assumption was that statins have unusual anti-inflammatory properties and may be effective in the treatment of asthma, as inflammation plays a central role in its pathogenesis. The primary variable of the study was the standard ACT questionnaire score, which provided a qualitative evaluation. The spirometric change provided a quantitative assessment of lung function, and the change in peripheral blood eosinophil counts was used as an indirect indicator of airway inflammation severity. Blood lipid measurements were used to assess the reliability of the patient's adherence to the treatment regimen. Our findings showed that atorvastatin was not effective in improving ACT scores or pulmonary volumes, including FEV1 and FVC. Furthermore, the peripheral blood eosinophil count showed no significant change. The significant difference in blood LDL-C and total cholesterol levels between the intervention and control groups indicated that the patients had adhered to the drug regime.
Our results are consistent with those of Menzies and colleagues,19 who used simvastatin for the first time in the treatment of mild to moderate asthma in humans; they were unable to show evidence of an anti-inflammatory effect of simvastatin after 4 weeks of treatment. In another study, the addition of atorvastatin to inhaled corticosteroids resulted in a reduction in the number of macrophages in sputum, but no improvement in the control of asthma symptoms.21
The therapeutic effects of statins were first reported by McKay et al.17 in a mouse sensitized with ovalbumin; simvastatin led to a reduction in inflammatory cells and eosinophilia as well as IL-4 and IL-5 levels in bronchoalveolar lavage (BAL) fluid. A later similar study showed that simvastatin reduced the levels of specific IgE against ovalbumin and the total numbers of inflammatory cells, macrophages, and neutrophils in the BAL fluid of an allergic mouse sensitized with ovalbumin.18 A reduction in allergic inflammation of the airways and inhibition of Th1- and Th2-associated chemokines has also been reported in ovalbumin-sensitized mice.25 Smooth muscle hypersensitivity in response to acetylcholine was significantly reduced in mice treated with lovastatin.24 Pravastatin inhibited systemic sensitivity to allergens, with reduced production of IL-12 and inhibition of antigen presentation in the lungs of mice.23
With regard to human studies, fluvastatin has been shown to inhibit the proliferation of peripheral blood mononuclear cells, decrease the production of IL-5 and IFN-γ, and inhibit Th1 and Th2 cell migration.20 Wachter22 reported a correlation between taking statins and a reduced rate of asthma exacerbation.
One probable reason for the lack of a significant clinical response to statins in our study, as well as in other human studies, is that the therapeutic effects observed in vitro and in mice do not necessarily occur in humans. Alternatively, the effect of statins might have been primarily due to the inhibition of the activity and differentiation of lymphocytes toward the Th1 subtype.12,13,28 However, the effect of statins on Th2 cells, which are the predominant lymphocytes in the pathogenesis of asthma, is inconclusive. Finally, it may be that a favorable clinical response requires a higher drug dosage or a longer period of treatment, or that the variables selected in this study were insufficiently sensitive to show improved asthma control.
As a limitation of this study, we did not have access to more sensitive quantitative methods for evaluating airway hyper-responsiveness and inflammation. Measurements made using a peak flow meter, methacholine challenge test, or quantitative and sensitive CRP assay, or measurements of the number of saliva eosinophils, the fraction of exhaled nitric oxide, or alveolar nitric oxide29 may provide more informative results.
In conclusion, our data indicate that atorvastatin is not effective for the treatment of mild to moderate asthma. More research on the effects of statins on severe asthma is necessary. As statins may exert a therapeutic effect by influencing the remodeling process, we propose the evaluation of patient responses to long-term treatment with statins.
Figures and Tables
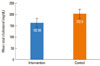 | Fig. 2Mean total cholesterol after 8 weeks of treatment with atorvastatin (intervention) or placebo (control) (P>0.05). |
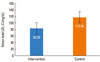 | Fig. 3Mean low-density lipoprotein cholesterol (LDL-C) after 8 weeks of treatment with atorvastatin (intervention) or placebo (control) (P>0.05). |
ACKNOWLEDGMENTS
The authors appreciate the assistance of the Research Council of Arak University of Medical Sciences and the staff of Vali-asr and Amir-al-momenin Hospitals. Our best regards to Dr. Rafii for his kind consultations. We express our sincere gratitude to Farzan Institute for Research & Technology for technical assistance. This paper is the result of a professional dissertation.
References
1. Bateman ED, Bousquet J, FitzGerald M, Haahtela T, O'Byme P, Ohta K, Paggiaro P, Pedersen SE, Soto-Quiroz M, Tan W, Wong G. Global initiative for asthma management and prevention (GINA) [Internet]. 2008. Available from: http://www.ginasthma.org.
2. Kikuchi K, Takizawa H. Lung remodeling in asthma: a new target of anti-asthma therapy. Drug Design Reviews - Online. 2005. 2:129–136. Available from: http://www.citeulike.org/article/115023.
3. Ennis M. New targets for modifying mast cell activation in asthma. Curr Allergy Asthma Rep. 2006. 6:247–251.
4. Maron DJ, Fazio S, Linton MF. Current perspectives on statins. Circulation. 2000. 101:207–213.
5. Shepherd J, Cobbe SM, Ford I, Isles CG, Lorimer AR, MacFarlane PW, McKillop JH, Packard CJ. West of Scotland Coronary Prevention Study Group. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N Engl J Med. 1995. 333:1301–1307.
6. Albert MA, Danielson E, Rifai N, Ridker PM. PRINCE Investigators. Effect of statin therapy on C-reactive protein levels: the pravastatin inflammation/CRP evaluation (PRINCE): a randomized trial and cohort study. JAMA. 2001. 286:64–70.
7. Ridker PM, Rifai N, Pfeffer MA, Sacks F, Braunwald E. The Cholesterol and Recurrent Events (CARE) Investigators. Long-term effects of pravastatin on plasma concentration of C-reactive protein. Circulation. 1999. 100:230–235.
8. Kwak B, Mulhaupt F, Myit S, Mach F. Statins as a newly recognized type of immunomodulator. Nat Med. 2000. 6:1399–1402.
9. Sparrow CP, Burton CA, Hernandez M, Mundt S, Hassing H, Patel S, Rosa R, Hermanowski-Vosatka A, Wang PR, Zhang D, Peterson L, Detmers PA, Chao YS, Wright SD. Simvastatin has anti-inflammatory and antiatherosclerotic activities independent of plasma cholesterol lowering. Arterioscler Thromb Vasc Biol. 2001. 21:115–121.
10. Musial J, Undas A, Gajewski P, Jankowski M, Sydor W, Szczeklik A. Anti-inflammatory effects of simvastatin in subjects with hypercholesterolemia. Int J Cardiol. 2001. 77:247–253.
11. Hernández-Presa MA, Martín-Ventura JL, Ortego M, Gómez-Hernández A, Tuñón J, Hernández-Vargas P, Blanco-Colio LM, Mas S, Aparicio C, Ortega L, Vivanco F, Gerique JG, Díaz C, Hernández G, Egido J. Atorvastatin reduces the expression of cyclooxygenase-2 in a rabbit model of atherosclerosis and in cultured vascular smooth muscle cells. Atherosclerosis. 2002. 160:49–58.
12. Ghittoni R, Lazzerini PE, Pasini FL, Baldari CT. T lymphocytes as targets of statins: molecular mechanisms and therapeutic perspectives. Inflamm Allergy Drug Targets. 2007. 6:3–16.
13. Hakamada-Taguchi R, Uehara Y, Kuribayashi K, Numabe A, Saito K, Negoro H, Fujita T, Toyo-oka T, Kato T. Inhibition of hydroxymethylglutaryl-coenzyme a reductase reduces Th1 development and promotes Th2 development. Circ Res. 2003. 93:948–956.
14. Qi XF, Kim DH, Yoon YS, Li JH, Jin D, Teng YC, Kim SK, Lee KJ. Fluvastatin inhibits expression of the chemokine MDC/CCL22 induced by interferon-gamma in HaCaT cells, a human keratinocyte cell line. Br J Pharmacol. 2009. 157:1441–1450.
15. Greenwood J, Steinman L, Zamvil SS. Statin therapy and autoimmune disease: from protein prenylation to immunomodulation. Nat Rev Immunol. 2006. 6:358–370.
16. Paraskevas KI, Tzovaras AA, Briana DD, Mikhailidis DP. Emerging indications for statins: a pluripotent family of agents with several potential applications. Curr Pharm Des. 2007. 13:3622–3636.
17. McKay A, Leung BP, McInnes IB, Thomson NC, Liew FY. A novel anti-inflammatory role of simvastatin in a murine model of allergic asthma. J Immunol. 2004. 172:2903–2908.
18. Kim DY, Ryu SY, Lim JE, Lee YS, Ro JY. Anti-inflammatory mechanism of simvastatin in mouse allergic asthma model. Eur J Pharmacol. 2007. 557:76–86.
19. Menzies D, Nair A, Meldrum KT, Fleming D, Barnes M, Lipworth BJ. Simvastatin does not exhibit therapeutic anti-inflammatory effects in asthma. J Allergy Clin Immunol. 2007. 119:328–335.
20. Samson KT, Minoguchi K, Tanaka A, Oda N, Yokoe T, Yamamoto Y, Yamamoto M, Ohta S, Adachi M. Inhibitory effects of fluvastatin on cytokine and chemokine production by peripheral blood mononuclear cells in patients with allergic asthma. Clin Exp Allergy. 2006. 36:475–482.
21. Hothersall EJ, Chaudhuri R, McSharry C, Donnelly I, Lafferty J, McMahon AD, Weir CJ, Meiklejohn J, Sattar N, McInnes I, Wood S, Thomson NC. Effects of atorvastatin added to inhaled corticosteroids on lung function and sputum cell counts in atopic asthma. Thorax. 2008. 63:1070–1075.
22. Wachter K. Statins tied to few hospital visits for asthma. Internal Medicine News. 2009. 05. 01. 26.
23. Imamura M, Okunishi K, Ohtsu H, Nakagome K, Harada H, Tanaka R, Yamamoto K, Dohi M. Pravastatin attenuates allergic airway inflammation by suppressing antigen sensitisation, interleukin 17 production and antigen presentation in the lung. Thorax. 2009. 64:44–49.
24. Chiba Y, Sato S, Misawa M. Inhibition of antigen-induced bronchial smooth muscle hyperresponsiveness by lovastatin in mice. J Smooth Muscle Res. 2008. 44:123–128.
25. Zeki AA, Franzi L, Last J, Kenyon NJ. Simvastatin inhibits airway hyperreactivity: implications for the mevalonate pathway and beyond. Am J Respir Crit Care Med. 2009. 180:731–740.
26. National Asthma Education and Prevention Program: Expert panel report III: Guidelines for the diagnosis and management of asthma (NIH publication no. 08-4051). 2007. accessed 2007 Sep 1. Bethesda, MD: National ;Available from: www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm.
27. Nathan RA, Sorkness CA, Kosinski M, Schatz M, Li JT, Marcus P, Murray JJ, Pendergraft TB. Development of the asthma control test: a survey for assessing asthma control. J Allergy Clin Immunol. 2004. 113:59–65.
28. Liu W, Li WM, Gao C, Sun NL. Effects of atorvastatin on the Th1/Th2 polarization of ongoing experimental autoimmune myocarditis in Lewis rats. J Autoimmun. 2005. 25:258–263.
29. Pascual RM, Johnson JJ, Peters SP. Fishman AP, Elias JA, Fishmna JA, Gripp MA, editors. Asthma: clinical presentation and management. Fishman's pulmonary diseases and disorders. 2008. 4th ed. New York: McGraw Hill;817–819.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download