Abstract
BACKGROUND/OBJECTIVES
Previous studies have indicated that when compared to young mice, old mice have lower global DNA methylation and higher p16 promoter methylation in colonic mucosa, which is a common finding in colon cancer. It is also known that a Western-style diet (WSD) high in fat and calories, and low in calcium, vitamin D, fiber, methionine and choline (based on the AIN 76A diet) is tumorigenic in colons of mice. Because DNA methylation is modifiable by diet, we investigate whether a WSD disrupts DNA methylation patterns, creating a tumorigenic environment.
SUBJECTVIES/METHODS
We investigated the effects of a WSD and aging on global and p16 promoter DNA methylation in the colon. Two month old male C57BL/6 mice were fed either a WSD or a control diet (AIN76A) for 6, 12 or 17 months. Global DNA methylation, p16 promoter methylation and p16 expression were determined by LC/MS, methyl-specific PCR and real time RT-PCR, respectively.
RESULTS
The WSD group demonstrated significantly decreased global DNA methylation compared with the control at 17 months (4.05 vs 4.31%, P = 0.019). While both diets did not change global DNA methylation over time, mice fed the WSD had lower global methylation relative to controls when comparing all animals (4.13 vs 4.30%, P = 0.0005). There was an increase in p16 promoter methylation from 6 to 17 months in both diet groups (P < 0.05) but no differences were observed between diet groups. Expression of p16 increased with age in both control and WSD groups.
CONCLUSIONS
In this model a WSD reduces global DNA methylation, whereas aging itself has no affect. Although the epigenetic effect of aging was not strong enough to alter global DNA methylation, changes in promoter-specific methylation and gene expression occurred with aging regardless of diet, demonstrating the complexity of epigenetic patterns.
DNA methylation, the addition of methyl moieties to cytosine residues of CpG dinucleotides in DNA, is a major epigenetic phenomenon. It can be defined as an inheritable but reversible molecular mechanism that affects gene expression without altering the genetic code itself. It is well known that epigenetic phenomena can be significantly influenced by environmental factors during our lifetime. Thus, both the aging process and our diet are associated with significant changes in DNA methylation [123]. In general, aging decreases global DNA methylation with a paradoxical increase in DNA methylation at specific genes, which is quite similar to DNA methylation changes in cancer. We therefore hypothesized that aging may provide an epigenetic environment that promotes the development of cancer [4]. Aging is a major risk factor of many types of cancer including colon cancer. Interestingly the diet can also modify DNA methylation pattern, hence we further hypothesized that the diet may alter the risk of cancer by modifying the age- associated epigenetic environment. Because a Western style diet, rich in fat and calories and low in vitamin D, calcium, fiber, methionine and choline can induce rodent colonic tumors [56], we determined the epigenetic effects of this diet on colonic mucosa by measuring global DNA methylation and p16 gene promoter methylation throughout a 17 month diet intervention. Global DNA methylation is useful because it reflects the gross epigenetic effect and genetic stability during aging and cancer, both of which are associated with reduced global DNA methylation [7]. We chose to also measure p16 gene specific methylation and expression because this gene is a tumor suppressor gene capable of inducing cell cycle arrest in G1 and G2 phases and important for both normal aging and colon cancer development [8910].
Most of the previously published aging and nutrition studies have compared a certain period of dietary intervention in young and old animals that have been aged in another facility. However without the guarantee that all of the animals in the study were raised in identical environments there is a limitation to conclusions that can be made from these studies. The epigenetic difference between these young and old animals may have resulted from environmental factors other than dietary changes the older animals experienced during their longer lifespan. We know that epigenetic traits can be easily altered by many different types of environmental factors [11], therefore it is important to keep the environment of all animals similar to one another. In this study, we ensured that environmental exposures for all mice, except the diet, were comparable during aging. We sequentially measured global DNA methylation, p16 promoter methylation and p16 gene expression in colonic mucosa to determine the epigenetic effects of aging and lifelong dietary exposure.
This Western-style diet (WSD) and aging study was reviewed and approved by the Institutional Animal Care and Use Committee of the USDA Human Nutrition Research Center on Aging at Tufts University (MA7-IACUC-08/12/05). To provide the same environment during aging we purchased 2 month old mice from a single vendor and aged them in our facilities.
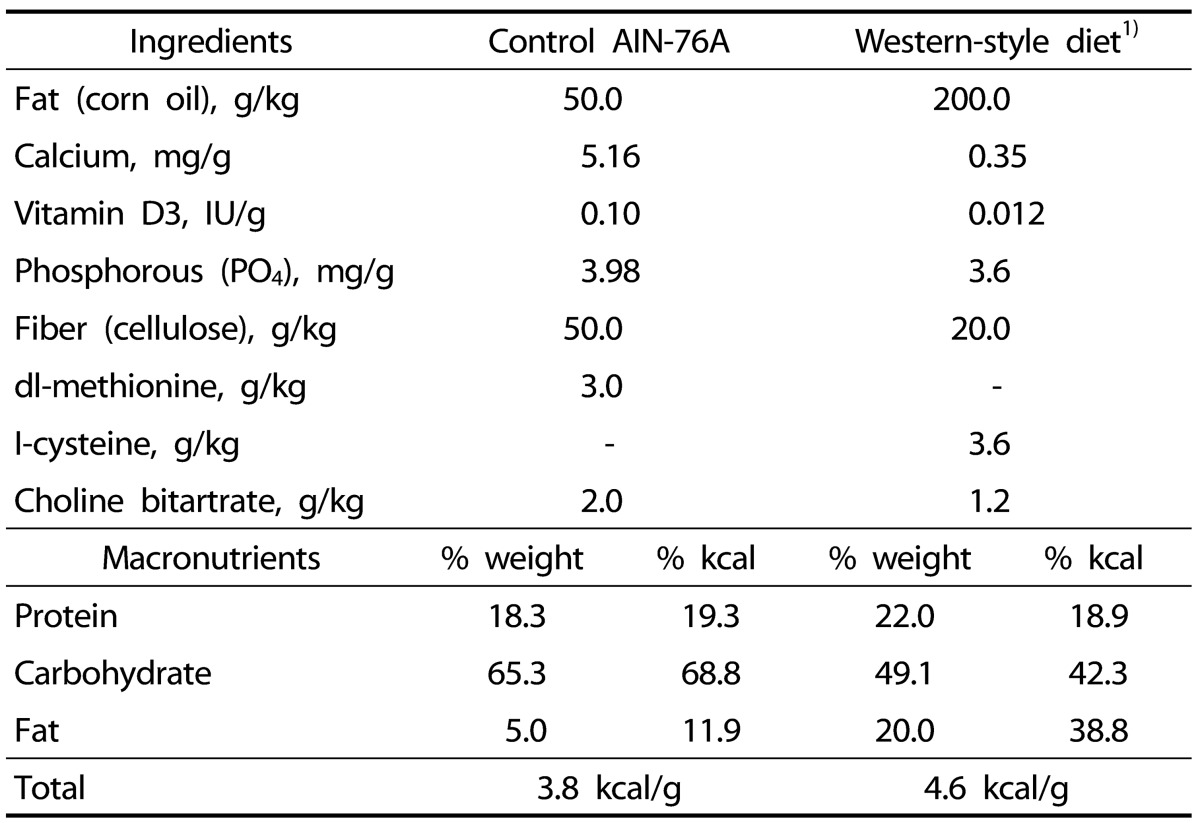
Forty-eight 2-month-old C57BL/6 male mice were aged for 17 months under two different diets: the WSD or the control diet as originally described in the previous studies [56]. The control diet was the AIN 76-A diet and the WSD was modified to be high in calories (4.6 kcal/g) and fat (38.8% kcal) and low in vitamin D, calcium, fiber and methionine (Table 1). For dietary folate we used the rodent daily requirement, 2mg folic acid/kg diet, to avoid the effects that folate has on DNA methylation that are already known in the elder mouse colon [2]. A previous study also demonstrated that this WSD containing 2 mg folic acid/kg diet has the similar tumorigenic effect to the WSD containing low dietary folate (0.23 mg folic acid/kg) [6]. Individually housed mice were group pair-fed to decrease variability in food and nutrient intakes within dietary groups. The mice were killed at either 6, 12 or 17 months after beginning the diet; 1) 6 control and 6 WSD mice at 6 months, 2) 7 control and 7 WSD mice at 12 months, and 3) 9 control and 11 WSD mice at 17 months. Colonic mucosa samples for DNA and RNA were prepared as previously described [2].
Global DNA methylation, p16 promoter methylation and p16 expression were determined by LC/MS, methyl-specific PCR following bisulfite treatment and real time RT-PCR, respectively, as we previously described in detail [2312]. A t-test comparing the weights of both diet groups at each month of the study was used to determine differences. Differences in p16 expression levels were calculated through ΔCt. Methylation and gene expression differences were analyzed through two-way ANOVA with a post hoc analysis using Bonferroni's method for multiple comparisons. Regression analysis was used for determining the aging effect (P-trend), and a Pearson's correlation was performed for the correlation between p16 methylation and expression. All values in text are mean ± SE. A P-value of <0.05 is the criterion for significance in all instances.
Among 48 mice, one mouse fed the control diet died at the 9th month of diet and one mouse fed WSD died at the 5th month. Any specific causes of death were not found at the necropsy.
Compared with the control group the WSD group mice maintained a more than 10% higher body weight (P < 0.001) throughout the study except at the last two months (P = 0.073 and 0.053 at months 16 and 17) (Fig. 1). Because we found that the WSD group mice started to lose body weight at 16 months, we killed mice at 17 month, which was one month ahead for our initial plan. We expected that the epigenetic effect of weight reduction by itself might mask that of the WSD and aging. In the control diet group body weight was stationary from 14 months until the end of the study (Fig. 1).
In contrast to previous studies [56] we could not find any colonic tumors in both diet groups at all ages. At the 17 month time period we found a small liver tumor (diameter 0.5 cm) in one mouse fed WSD and in two mice fed the control diet. This was expected, as aged C57BL/6 mice are known to develop hepatic tumors spontaneously [1314], and our concern is that the tumors were the cause of weight loss near the end of the study. However, it seems that these liver tumors were not the cause of this weight decline in the WSD group because the mouse that harbored a small liver tumor had average levels of body weight.
The WSD significantly reduced global DNA methylation relative to the control group when comparing all animals (4.13 ± 0.04 vs 4.30 ± 0.03%; P = 0.0005). When separating the groups out by age there was a significant reduction in DNA methylation in mice fed the WSD relative to the control in the 17 month age group (4.05 ± 0.04 vs 4.31 ± 0.03%; P = 0.019) but not at earlier time points (Fig. 2). However, as the animals aged neither diet group showed any significant changes in global DNA methylation during the 17 months of observation (Fig. 2).
During the 17 month observation period p16 gene promoter methylation was incrementally increased in both diets with an increase in age (P-trend < 0.0001 for both diet groups). In both diet groups p16 promoter methylation was significantly higher in the 17 month group relative to the 6 month group, but there was no significant difference between two diet groups (Fig. 3).
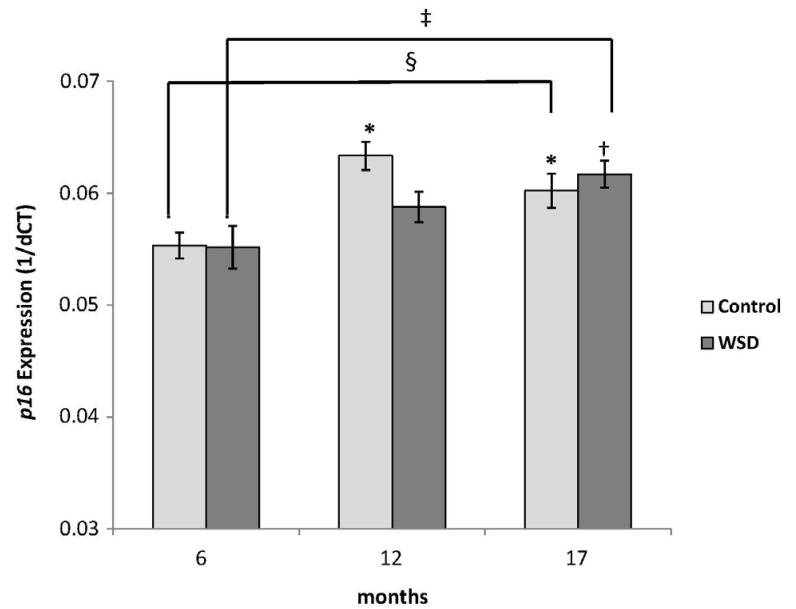
The expression of p16 measured by RT-PCR increased in all mice at both 12 and 17 month time points relative to the 6 month group (Fig. 4A). WSD group mice showed a stepwise increment of p16 expression during the 17 month observation (P-trend = 0.003), while the control group showed a tendency (P-trend = 0.07) (Fig. 4B). However, there were no differences in expression between the WSD and control groups at any time point.
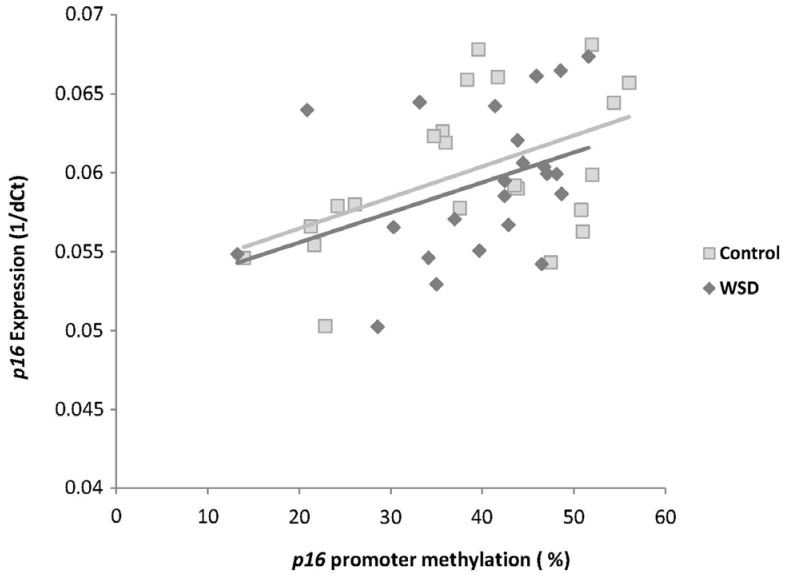
In the control diet group p16 promoter methylation was positively correlated with p16 gene expression (P = 0.015), while in the WSD group this correlation neared significance (P = 0.057) (Fig. 5).
An important strength of this study is that all animals were housed and aged in our own animal facility for the majority of their lives. This ensures that environmental exposures for all mice, except the diet, were comparable during aging.
In the present study a WSD reduces global DNA methylation in the 17 month group only, demonstrating that either the long term feeding of this diet, or this diet coupled with old age has the most effect on DNA methylation. The low amount of dietary methyl donor nutrients methionine and choline in the WSD could be responsible for the reduction of global DNA methylation, even though there is no direct evidence that low dietary methionine or choline reduces global DNA methylation in murine colon. Low dietary methionine or choline is known to alter one-carbon metabolism that regulates DNA methylation through two metabolites, S-adenosylmethionine which is the unique methyl donor to the DNA methylation reaction, and S-adenosylhomocysteine which can inhibit DNA methyltransferases [7]. A disruption in either of these mechanisms may cause a change in DNA methylation.
The high fat and calorie components of the WSD are a possible explanation for altered global DNA methylation through changes in metabolism. Both high fat and high caloric intakes substantially change lipid metabolism and energy metabolism, thereby providing a different metabolic environment. Even though the mechanism has not been clearly elucidated [4], recent studies have demonstrated that both transgenerational and postnatal exposure to the metabolic characteristics of a high fat diet can lead to DNA methylation changes [151617]. Animal studies also suggested that low dietary choline and high dietary fat synergistically can affect DNA methylation through one-carbon metabolism [181920].
The WSD used in this study was low in vitamin D and calcium, both of which are risk factors of colon cancer. A recent human study demonstrated that dietary vitamin D intake is negatively associated with DNA methylation of a Wnt regulatory gene in a Canadian cohort [21]. Furthermore, incubation of MCF-7 breast cancer cells with vitamin D3 reduces the aberrant hypermethylation and restores the expression of genes silenced epigenetically [2223]. Although the mechanism remains to be validated in vivo, this work indicates that vitamin D can modify gene-specific DNA methylation, and therefore should be considered as a possible cause of changes in DNA methylation in this study.
Low dietary fiber is a risk factor of colon cancer. Fiber is known to alter histone acetylation in the colon after converting to short chain fatty acid such as butyrate [24]. Butyrate is a histone deacetylase inhibitor [25]. Since DNA methylation and histone acetylation work together to change gene expression we may also speculate that low dietary fiber indirectly altered DNA methylation through changes in histone acetylation [2627].
Collectively, our observations indicate that each component of the WSD diet, low in dietary methyl donor nutrients, vitamin D and fiber as well as high in fat and calories, may work together to achieve global DNA hypomethylation. This is relevant because DNA hypomethylation is frequently found in colon cancer, even though mice fed the diet in this study did not develop colonic tumors. While testing the effects of feeding a diet as a whole instead of each dietary component individually leads to more complicated results, we feel that it better mimics the complexities of a human diet.
Aging did not change global DNA methylation status within the same diet group. This observation is quite different from what we have previously found in our own research. In such studies comparing young and old mice, old mice had significantly reduced global DNA methylation levels that were modified by the diet [23]. However, there is a possibility that the old mice in the previous studies might have experienced a certain environmental influence other than diet before the study began that the young mice did not. We therefore provided the same environment besides experimental diet throughout the 17 months of this study period. Our observation suggests that under the same environment throughout most of the lifespan, including the diet, the effect of aging on global DNA methylation is minimal in the C57BL/6 male mice colon. On the other hand, p16 gene specific methylation showed stepwise increases in both diet groups during the 17 month study period, which is consistent with a previous young and old mouse study [2] (Fig. 3). This study suggests that aging has a stronger effect on gene specific DNA methylation than global DNA methylation.
Proper expression of p16 is important in both aging and carcinogenesis due to its regulatory roles in the cell cycle [8910]. As we previously reported [23] p16 promoter methylation is positively correlated with p16 gene expression, suggesting that increased p16 promoter methylation during aging might increase p16 gene expression. In contrast to colon cancer, in which a fully methylated p16 promoter silences this gene [28], in the normal aged mouse colon where p16 promoter hypermethylation was increased up to 40-50%, we consistently find that p16 methylation status is positively correlated with this gene's expression. It appears that hypermethylation increases the expression of this gene until the p16 promoter is fully methylated. It is also possible to speculate that changes in p16 methylation caused by aging, a physiologic condition, could be different from those caused by cancer, a pathologic condition, because p16 gene repression by cancer may be combined with other epigenetic changes such as histone modifications and chromatin remodeling. These may in turn affect gene expression differently from DNA methylation itself. In fact a study suggested that altered histone modifications, rather than DNA methylation, is the cause of transcriptional repression of p16 in a breast cancer model [29]. Expression of p16 is also known to be regulated by Polycomb repressive complexes [3031], a chromatin remodeling mechanism that represses gene expression with multiple Polycomb repressive proteins. p16 promoter methylation changes from aging may interfere with the recruitment of such repressive proteins so that the gene cannot be repressed. A previous study has demonstrated that p16 expression was found to be up-regulated in premalignant lesions and was lost in invasive carcinomas [9]. Thus aging may gradually increase p16 gene expression to reduce cell proliferation by increasing promoter methylation, but when a normal cell is converted into a cancer cell, the p16 gene is silenced by a fully methylated promoter along with other repressive epigenetic marks.
In this WSD and aging study we found that lifelong exposure to a certain diet can provide a certain epigenetic environment that may facilitate age-associated diseases such as cancer. The epigenetic effect of aging is not strong enough to alter global DNA methylation but aging can still affect promoter methylation and expression of critical genes regardless of diet. We still do not know the exact meaning of DNA methylation changes by aging and whether these changes are programmatic or randomly acquired by environmental factors including the diet. However, through studies like the type that we have presented in this paper we may clarify the significance of age associated DNA methylation changes, especially in the development of cancer or other age associated diseases.
References
1. Choi SW, Friso S, Keyes MK, Mason JB. Folate supplementation increases genomic DNA methylation in the liver of elder rats. Br J Nutr. 2005; 93:31–35. PMID: 15705222.

2. Keyes MK, Jang H, Mason JB, Liu Z, Crott JW, Smith DE, Friso S, Choi SW. Older age and dietary folate are determinants of genomic and p16-specific DNA methylation in mouse colon. J Nutr. 2007; 137:1713–1717. PMID: 17585020.

3. Sauer J, Jang H, Zimmerly EM, Kim KC, Liu Z, Chanson A, Smith DE, Mason JB, Friso S, Choi SW. Ageing, chronic alcohol consumption and folate are determinants of genomic DNA methylation, p16 promoter methylation and the expression of p16 in the mouse colon. Br J Nutr. 2010; 104:24–30. PMID: 20205967.
4. Park LK, Friso S, Choi SW. Nutritional influences on epigenetics and age-related disease. Proc Nutr Soc. 2012; 71:75–83. PMID: 22051144.

5. Newmark HL, Yang K, Lipkin M, Kopelovich L, Liu Y, Fan K, Shinozaki H. A Western-style diet induces benign and malignant neoplasms in the colon of normal C57Bl/6 mice. Carcinogenesis. 2001; 22:1871–1875. PMID: 11698351.

6. Yang K, Yang W, Mariadason J, Velcich A, Lipkin M, Augenlicht L. Dietary components modify gene expression: implications for carcinogenesis. J Nutr. 2005; 135:2710–2714. PMID: 16251635.

7. Choi SW, Friso S. Epigenetics: a new bridge between nutrition and health. Adv Nutr. 2010; 1:8–16. PMID: 22043447.

8. Rayess H, Wang MB, Srivatsan ES. Cellular senescence and tumor suppressor gene p16. Int J Cancer. 2012; 130:1715–1725. PMID: 22025288.

9. Kriegl L, Neumann J, Vieth M, Greten FR, Reu S, Jung A, Kirchner T. Up and downregulation of p16(Ink4a) expression in BRAF-mutated polyps/adenomas indicates a senescence barrier in the serrated route to colon cancer. Mod Pathol. 2011; 24:1015–1022. PMID: 21423154.

10. Campisi J. Aging, cellular senescence, and cancer. Annu Rev Physiol. 2013; 75:685–705. PMID: 23140366.

11. Tammen SA, Friso S, Choi SW. Epigenetics: the link between nature and nurture. Mol Aspects Med. 2013; 34:753–764. PMID: 22906839.

12. Friso S, Choi SW, Dolnikowski GG, Selhub J. A method to assess genomic DNA methylation using high-performance liquid chromatography/electrospray ionization mass spectrometry. Anal Chem. 2002; 74:4526–4531. PMID: 12236365.

13. Frith CH, Highman B, Burger G, Sheldon WD. Spontaneous lesions in virgin and retired breeder BALB/c and C57BL/6 mice. Lab Anim Sci. 1983; 33:273–286. PMID: 6876733.
14. James SJ, Muskhelishvili L. Rates of apoptosis and proliferation vary with caloric intake and may influence incidence of spontaneous hepatoma in C57BL/6 x C3H F1 mice. Cancer Res. 1994; 54:5508–5510. PMID: 7923185.
15. Vucetic Z, Kimmel J, Totoki K, Hollenbeck E, Reyes TM. Maternal high-fat diet alters methylation and gene expression of dopamine and opioid-related genes. Endocrinology. 2010; 151:4756–4764. PMID: 20685869.

16. Widiker S, Karst S, Wagener A, Brockmann GA. High-fat diet leads to a decreased methylation of the Mc4r gene in the obese BFMI and the lean B6 mouse lines. J Appl Genet. 2010; 51:193–197. PMID: 20453306.
17. Milagro FI, Campión J, García-Díaz DF, Goyenechea E, Paternain L, Martínez JA. High fat diet-induced obesity modifies the methylation pattern of leptin promoter in rats. J Physiol Biochem. 2009; 65:1–9. PMID: 19588726.

18. Zeisel SH. Metabolic crosstalk between choline/1-carbon metabolism and energy homeostasis. Clin Chem Lab Med. 2013; 51:467–475. PMID: 23072856.

19. Wu G, Zhang L, Li T, Lopaschuk G, Vance DE, Jacobs RL. Choline deficiency attenuates body weight gain and improves glucose tolerance in ob/ob mice. J Obes. 2012; 2012:319172. PMID: 22778916.

20. Rubio-Aliaga I, Roos B, Sailer M, McLoughlin GA, Boekschoten MV, van Erk M, Bachmair EM, van Schothorst EM, Keijer J, Coort SL, Evelo C, Gibney MJ, Daniel H, Muller M, Kleemann R, Brennan L. Alterations in hepatic one-carbon metabolism and related pathways following a high-fat dietary intervention. Physiol Genomics. 2011; 43:408–416. PMID: 21303933.

21. Rawson JB, Sun Z, Dicks E, Daftary D, Parfrey PS, Green RC, Gallinger S, McLaughlin JR, Wang PP, Knight JA, Bapat B. Vitamin D intake is negatively associated with promoter methylation of the Wnt antagonist gene DKK1 in a large group of colorectal cancer patients. Nutr Cancer. 2012; 64:919–928. PMID: 22966878.
22. Stefanska B, Salamé P, Bednarek A, Fabianowska-Majewska K. Comparative effects of retinoic acid, vitamin D and resveratrol alone and in combination with adenosine analogues on methylation and expression of phosphatase and tensin homologue tumour suppressor gene in breast cancer cells. Br J Nutr. 2012; 107:781–790. PMID: 21801466.

23. Stefanska B, Rudnicka K, Bednarek A, Fabianowska-Majewska K. Hypomethylation and induction of retinoic acid receptor beta 2 by concurrent action of adenosine analogues and natural compounds in breast cancer cells. Eur J Pharmacol. 2010; 638:47–53. PMID: 20447390.

24. Zimmerman MA, Singh N, Martin PM, Thangaraju M, Ganapathy V, Waller JL, Shi H, Robertson KD, Munn DH, Liu K. Butyrate suppresses colonic inflammation through HDAC1-dependent Fas upregulation and Fas-mediated apoptosis of T cells. Am J Physiol Gastrointest Liver Physiol. 2012; 302:G1405–G1415. PMID: 22517765.

25. Astbury SM, Corfe BM. Uptake and metabolism of the short-chain fatty acid butyrate, a critical review of the literature. Curr Drug Metab. 2012; 13:815–821. PMID: 22571479.
26. Sarkar S, Abujamra AL, Loew JE, Forman LW, Perrine SP, Faller DV. Histone deacetylase inhibitors reverse CpG methylation by regulating DNMT1 through ERK signaling. Anticancer Res. 2011; 31:2723–2732. PMID: 21868513.
27. Spurling CC, Suhl JA, Boucher N, Nelson CE, Rosenberg DW, Giardina C. The short chain fatty acid butyrate induces promoter demethylation and reactivation of RARbeta2 in colon cancer cells. Nutr Cancer. 2008; 60:692–702. PMID: 18791934.
28. Goto T, Mizukami H, Shirahata A, Sakata M, Saito M, Ishibashi K, Kigawa G, Nemoto H, Sanada Y, Hibi K. Aberrant methylation of the p16 gene is frequently detected in advanced colorectal cancer. Anticancer Res. 2009; 29:275–277. PMID: 19331161.
29. Zheng S, Pan YX. Histone modifications, not DNA methylation, cause transcriptional repression of p16 (CDKN2A) in the mammary glands of offspring of protein-restricted rats. J Nutr Biochem. 2011; 22:567–573. PMID: 20934317.

30. Jacobs JJ, Kieboom K, Marino S, DePinho RA, van Lohuizen M. The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature. 1999; 397:164–168. PMID: 9923679.

31. Maertens GN, El Messaoudi-Aubert S, Racek T, Stock JK, Nicholls J, Rodriguez-Niedenführ M, Gil J, Peters G. Several distinct polycomb complexes regulate and co-localize on the INK4a tumor suppressor locus. PLoS One. 2009; 4:e6380. PMID: 19636380.

Fig. 1
Body weight changes during the dietary intervention.
*P < 0.001, **P = 0.073, ***P = 0.053 (t-test) compared to control. WSD: western-style diet.
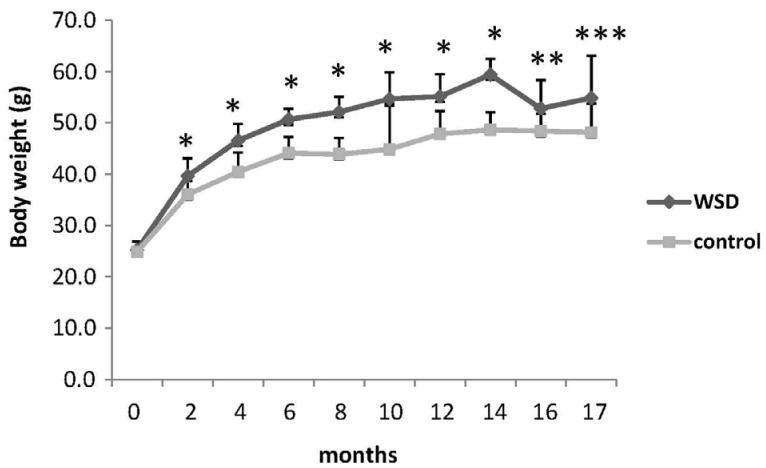
Fig. 2
Global DNA methylation changes in each diet group over the 17 months dietary intervention measured by LC/MS method. DNA methylation status was defined as the percentage of total cytosine (cytosine + 5-methylcytosine) that was in the 5-methylcytosine form.
Values are mean ± SE. *denotes a P-value less than 0.05 (two-way ANOVA with Bonferroni's posthoc test).
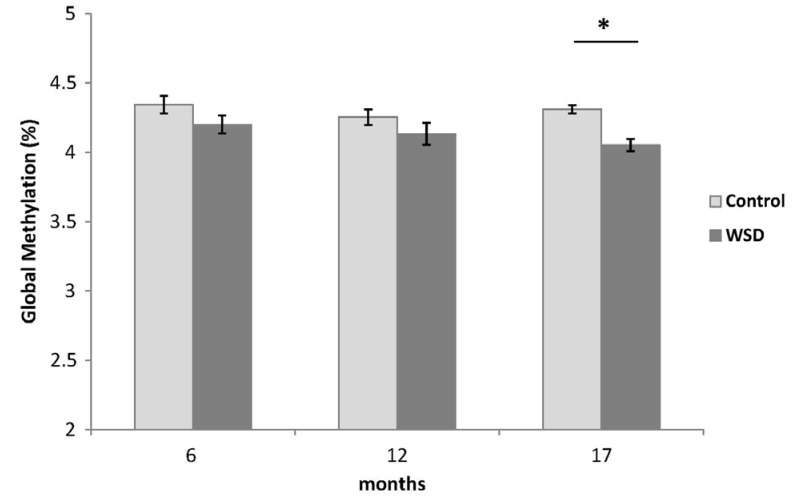
Fig. 3
p16 promoter methylation changes during the dietary intervention measured by methyl specific PCR.
Values are mean % methylation of CpG island ± SE. *Denotes a P-value less than 0.05 relative to the 6 month control group. †Denotes a P-value less than 0.05 relative to the 6 month WSD group. ‡P-trend < 0.0001 (regression analysis for both Control and WSD groups).
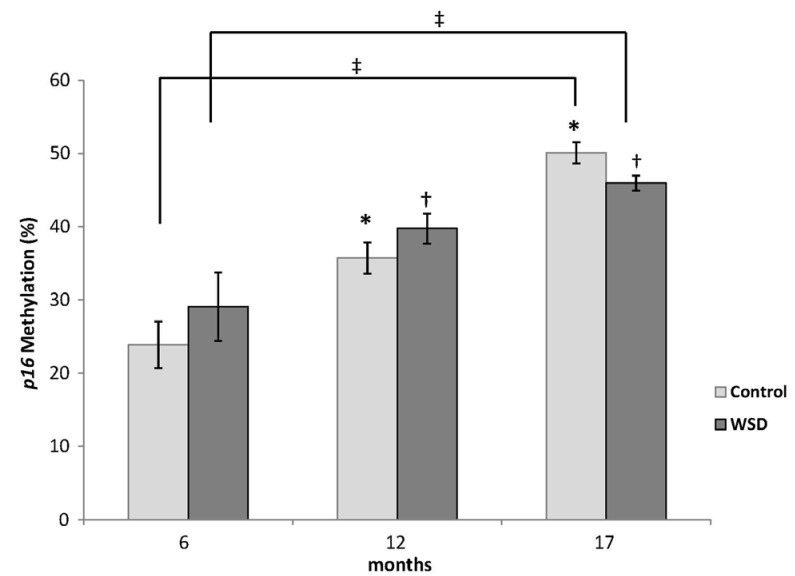
Fig. 4A
Changes in p16 gene expression in all mice measured by RT-PCR.
Values are mean expression (ΔCt) of p16 ± SE. Different letters (ex: a, b) denote a P-value less than 0.05.
Fig. 4B
Changes in p16 gene expression in each diet group.
Values are mean expression (ΔCt) of p16 ± SE. *Denotes a P-value less than 0.05 relative to the 6 month control group. †Denotes a P-value less than 0.05 relative to the 6 month WSD group. ‡P-trend for WSD = 0.003; §P-trend for Control = 0.07.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download