This article has been corrected. See "Erratum: Funding Acknowledgment" in Volume 7 on page 242.
Abstract
Obesity, an intractable metabolic disease, currently has no medical treatment without side effects, so studies have been actively carried out to find natural compounds that have anti-obesity activity with minimum side effects. In this study, the anti-obesity effects of water extracts of seven Capsicum annuum L. varieties being Putgochu (Pca), Oyee gochu (Oca), Kwari putgochu (Kca), Green pepper (Gca), Yellow paprika (Yca), Red paprika (Rca) and Cheongyang gochu (Cca), were examined through the evaluation of lipoprotein lipase (LPL) mRNA expression level in 3T3-L1 cells (mouse pre-adipocytes). After capsaicin elimination by chloroform defatting, freeze-dried powder of Cca was treated to 3T3-L1 cells and anti-obesity effects were examined by determining the LPL mRNA level using the RT-PCR method. Of the primary fractions, only proven fractions underwent secondary and tertiary refractionating to determine anti-obesity effects. From seven different Capsicum annuum L., there was a significant decrease of the LPL mRNA expression level of 50.9% in Cca treatment compared to the control group. A significant decrease of the LPL mRNA expression level was shown in primary fractions (Fr) 5 (36.2% decrease) and 6 (30.5% decrease) of the Cca water extracts. Due to the impurities checked by UPLC chromatography, Fr 5 and 6 were refractionated to determine the LPL mRNA expression level. Treatment of Fr 6-6 (35.8% decrease) and Fr 5-6 (35.3% decrease) showed a significant decrease in the LPL mRNA expression level. When analyzed using UPLC, major compounds of Fr 6-6 and Fr 5-6 were very similar. Subsequently, we refractionated Fr 6-6 and Fr 5-6 to isolate the major peak for structure elucidation. Treatment of Fr 5-6-1 (26.6% decrease) and Fr 6-6-1 (29.7% decrease) showed a significant decrease in the LPL mRNA expression level. Consequently, the fractions may have a possibility to ameliorate obesity through the decrease of the LPL mRNA expression level.
Obesity comes from the storage of surplus energy in fat cells through the abnormal metabolism of energy in the body, and is considered a major cause of several metabolic diseases [1]. When excess food energy is taken in and fat is accumulated in the body, obesity is the result. The intake of high fatty and/or high carbohydrate foods not only increases the number of fat cells in the body but also increases the weight of cells, causing obesity [2]. So, obesity involves an excess of fatty tissues rather than excessive weight. Obesity has a close relationship with diverse metabolic diseases such as diabetes, high cholesterol, high blood pressure and other cardiovascular diseases. It is well known that the prevalence and death rate of such diseases are relatively high among the obese [3-5]. One of the most general methods for treating obesity is to restrict the intake of energy sources such as fats and carbohydrates, but the restriction of energy intake can cause other problems, such as a reduction of the basal metabolic rate [6]. Though many anti-obesity medicines have been developed, they are also known to have numerous side effects. Up to now there has been no perfect obesity medicine developed without side effects [7-9]. In order to overcome such problems in obesity treatment, much research has been carried out on the use of natural food materials to prevent and cure obesity, as it is known that food not only supplies the calories and the nutrients but also helps to prevent and treat diseases with the natural functional materials it contains [10,11].
As a part of this area of research, this study deals with how the green peppers of the eggplant family (Solanaceae) work against obesity. Green pepper is a spice widely used around the world, particularly as a main material in kimchi and kochujang, typical Korean foods. It contains high amounts of phenolic acid, flavonoids, and vitamins C and E [12]. In particular, the capsaicin in green pepper has been studied a great deal because of its influence on both fat metabolism and energy metabolism [13-15]. Operating on the central nervous system, capsaicin is well known to have anti-obesity effects by controlling the appetite, reducing the fat concentration in the blood and restricting the generation of white fat cells [16-20].
However, according to our previous experiments, in addition to the fat-soluble capsaicin, water extracts of green pepper have considerable anti-obesity effects [21,22]. To further verify our previous studies, this study fractionates the water extracts of green pepper and investigates the fractions which have anti-obesity function through the manifestation of a restriction of lipoprotein lipase (LPL).
Dimethyl sulfoxide (DMSO), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT), chloroform, isopropanol and diethylpyrocarbonate (DEPC) manufactured by Sigma Co. (St. Louis, MO, USA); dNTP, Nuclease free water and GoTaq® Green Master mix by Promega Co. (S1, USA); SuperScript III reverse transcriptase, 5 × first-stand and DTT by Invifogen Co. (Carlsbat. Ca 92008, USA); Dulbecco's minimum essential medium (DMEM) for the culture of cells, fetal bovine serum (FBS), penicillin-streptomycin and trypsin-EDTA by Welgene (Daegu, Korea) were used. In addition, Sephadex LH20 was used as the resin for fraction.
Equipments used for the experiment included UV-visible spectrophotometer (2120 UV plus, Optigen), microplate reader (EL 808 series, BioTek), inverted microscope (Nikon), Freeze dryer (VD-16, Taitec Co.), shaking incubator (KMC8480SF, Vision), CO2 incubator (KMC-8409C, Vision), Freeze dryer (FD 8508, Ilshin), Evaporator (Eyela, Japan), centrifuge (MF600, Hanil), micro centrifuge (Micro 17R, Hanil) and gel reader (UVP, BioDoc-it).
Materials for this experiment included 7 edible green peppers belonging to Capsicum annuum L. of Solanacese: Chungyang gochu (Cca), Putgochu (Pca), Oyee gochu (Oca), Kwari putgochu (Kca), Green pepper (Gca), Yellow paprika (Yca) and Red paprika (Rca). Organic products with the names of actual farmers were purchased at Lotte Mart in Chuncheon City.
Each material was washed and the internal seeds were removed. After it was dried and crushed, distilled water (as much as 10 times its weight) was added to it, and the mix was stirred for 24 hours at 60℃ to extract the active elements. The extracts were centrifuged and filtered with a 0.45 µm syringe filter. After an amount of chloroform equal to the amount of the filtered liquid was added to it to remove the fat, reduced pressure concentration and lyophilization were carried out to make it a specimen.
Each natural material was used in concentrations of 100 ppm, 250 ppm, 500 ppm, 1,000 ppm, 2,500 ppm, 5,000 ppm and 10,000 ppm in the experiment.
Primary fractionation for pepper Cca dissolved in 750 g/3 L H2O was performed using sephadex LH20 gel CHP-20 (Φ4.5 × 20 cm), and 11 primary fractions were eluted with H2O. Secondary fractionations for fraction No. 5 3.3654 g dissolved in 20 ml H2O and No. 6 2.0198 g dissolved in 15 ml H2O were performed with MCI gel CHP-20 (Φ4.0 × 15 cm), and 11 (No. 5) and 12 (No. 6) fractions were eluted with H2O, ethanol, and acetone in each step. Tertiary fractions for fraction No. 5-6 dissolved in 247.9 mg/5 ml H2O and No. 6-6 dissolved in 138.7 mg/5 ml H2O were performed with Wakosil 40C18 (Φ4.0 × 15 cm), and fractions were eluted with 10% methanol.
The cell lines for this experiment were 3T3-L1 fat cells, which were dispensed from the Korean cell line bank (KCLB) and cultured in the lab. The 3T3-L1 cell lines were cultured on Dulbecco's minimum essential medium (DMEM) with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin (PEST) added in the incubator of 37℃ and 5% CO2.
MTT assay was carried out to check whether the water extracts from the natural materials at various concentrations showed toxicity to 3T3-L1 preadipocytes. The survival rate of cells was measured using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) reduction method. The 3T3-L1 cells were plated at an initial density of 1 × 105 cell/ml in a 96-well plate and treated with various concentrations of green pepper extracts for 24 hours. MTT solution was added to each well to a final concentration of 0.5 mg/ml. After 4 h of exposure at 37℃, the formazan crystals were dissolved in dimethylsulfoxide (DMSO), and the absorbance was estimated with an ELISA reader at 570 nm.
To estimate LPL and β-actin mRNA levels, total RNA was extracted by the QIAzol lysis reagent according to the manual. Complementary DNA was synthesized by SuperScript III reverse transcriptase from total RNA, and polymerase chain reactions for LPL and β-actin were administered with GoTaq®Green Master mix.
The primer sequences used for RT-PCR are as follows (Table 1).
UPLC analysis was performed using the Waters UPLC-XXX system. The analysis column was Waters BEH C18 (2.1 × 100 mm, 1.7 µm). The mobile phase (with flow rate 0.2 ml/min) consisted of A: H2O solution containing 0.1 % (v/v) formic acid, B: CH3CN solution containing 0.1 % (v/v) formic acid (gradient). The mobile solution was filtered through a 0.45 µm membrane filter and degassed prior to use. The injection volume was 1 µl. Compounds were detected by a UV detector (280 nm).
All experiments were carried out in triplicate, and statistical analyses were conducted by using the GraphPad InStat statistical package (GraphPad InStat Version 3.00, 2003). All expressed values are the mean ± standard deviation (SD). Analysis of variance (ANOVA) was conducted, and the Tukey-Kramer test was used to determine the significance of differences among groups more than three. Student's t-test was used to decide the significance between two groups. The level of statistical significance was set as P < 0.05.
To determine the influence of water extracts from natural materials on the manifestation of lipoprotein lipase (LPL) in 3T3-L1 cells, MTT assay was carried out first to set the material treatment concentration (Fig. 1). Each extract from 7 kinds of green pepper (Cca, Pca, Kca, Oca, Gca, Yca, Rca) was diluted to seven concentrations of 100 ppm, 250 ppm, 500 ppm, 1,000 ppm, 2,500 ppm, 5,000 ppm and 10,000 ppm, and added to cells in order to check their survival rate. Most materials demonstrated toxicity at the concentration of 5,000 ppm or higher. Notably, Cca killed the cells up 73.7% at 5,000 ppm and 89.3% at 10,000 ppm. Next, Kca killed 40.1% at 5,000 ppm and 70.9% at 10,000 ppm. At 2,500 ppm, Pca (18.0%) and Kca (22.6%) showed significant toxicity. But at 1,000 ppm or lower, the cell toxicities of Cca (10.5%), Pca (8.4%), Kca (11.8%), Oca (7.3%), Gca (2.7%), Yca (5.3%) and Rca (3.8%) were relatively small compared to other concentrations. Therefore, the treatment concentration for 7 kinds of pepper was determined as 1,000 ppm for this experiment.
The manifestation of LPL mRNA was measured by using 7 varieties of capsicum (Fig. 2). While Oca, Gca, Yca and Rca tended to increase the manifestation of LPL mRNA, Kca showed a decrease of 19.7% and Pca 9.9%, both of which were rather insignificant. However, Cca demonstrated a significant decrease of 50.9%. Therefore, Cca was selected as the final material to identify the substance restricting the manifestation of LPL mRNA.
Of the 7 kinds of pepper extracts, Chungyang gochu (Cca), which restricted the manifestation of LPL mRNA the most, was divided into 11 fractions, and the LPL manifestation of each fraction was measured (Fig. 3). Of these, the most active fractions were Fr. 5 and 6. Fr. 6 showed a 36.2% decrease, and Fr. 5 showed 30.5%. The next active fractions in descending order were Fr. 4 (20.3%) > Fr. 7 (18.0%) > Fr. 8 (17.2%) > Fr. 9 (16.5%). Thus, fractions other than Fr. 4 exhibited insignificant effects. Therefore, two fractions Fr. 5 and 6 with the greatest effects were selected as the best fractions to restrict the manifestation of LPL mRNA.
To identify the components of effective fractions, UPLC analysis was carried out. As the present fractions were the primary ones fractionated with natural extracts of diverse substances, both turned out to be mixtures of many elements (Fig. 4). Based on the UPLC result, the best solvent for the specimens was selected and the secondary fraction method was developed.
Based on UPLC data of Fr. 5, it was separated into 11 sub-fractions, and the LPL manifestation of each sub-fraction was measured (Fig. 5). Of the sub-fractions, Fr. 5-6 demonstrated the most significant reduction, followed in order of reduction amount by Fr. 5-7 (25.1%) > Fr. 5-8 (21.7%) > Fr. 5-9 (20.9%) > Fr. 5-4 (20.6%).
Based on UPLC data of Fr. 6, it was separated into 11 sub-fractions and the LPL manifestation of each sub-fraction was measured (Fig. 6). Of the sub-fractions, Fr. 6-6 demonstrated the highest significant reduction, followed in order of reduction amount by Fr. 6-7 (26.7%) > Fr. 6-1 (25.1%) > Fr. 6-2 (25.9%) > Fr. 6-8 (20.9%).
Through a comprehensive comparison of Fr. 5 and 6 subfractions, it was found that the most effective fractions for reducing the manifestation of LPL mRNA in the cell were Fr. 6-6 (35.8%) and Fr. 5-6 (35.3%). The next most effective ones were Fr. 6-7 (26.7%) > Fr. 5-7 (25.1%) > Fr. 6-1 (24.7%), which showed reductions that were significant but were lower than the foregoing most effective ones by 10% or more. In this respect, Fr. 5-6 and Fr. 6-6 were selected as the effective fractions.
According to the UPLC result for two effective fractions that showed the restriction of LPL mRNA of 35% or more (Fig. 7), the same peak appeared at the range of 6 min in both fractions. It is highly probable that the peak appearing at the same time range in the data of UPLC executed under the same conditions can represent the same element. However, as there are still many other peaks, it may not be a single element. Therefore, the solvent was selected to separate them as suitably as possible, and the method for additional fractioning was provided.
The LPL manifestation amount of cells was measured with Fr. 5-6-1, which was expected to be the final fraction of Fr. 5-6 (Fig. 8). While it was water-soluble before the fraction, the substance after fraction turned out to be dissolved in water of 70℃ or higher and congealed again at 4℃ or lower. Out of concern that there might be some problems if the medium was heated up to 70℃, the tertiary sterilized distilled water was heated to 70℃ and the fraction was dissolved with the water. It was then added to the medium. As a result of the experiment, manifestation of LPL mRNA was significantly restricted by approximately 26.6% in comparison with the control group.
The LPL manifestation amount of cells was measured with Fr. 5-6-1, which was expected as the final fraction of Fr. 6-6 (Fig. 9). The result of the experiment showed that the manifestation of LPL mRNA was significantly restricted by about 29.7% in comparison with the control group.
Green peppers used in this study are the vegetables having vitamins C and E of high contents. Capsaicin, a major component of Capsicum, is the ingredient that gives pepper its 'chili' taste. It is a phenolic compound of the alkaloid line, which operates on the adrenal medulla to promote energy metabolism [14], to create antioxidant [23,24], pain-relieving [25], anticancer [26] and anti-inflammation effects [27]. There have been many studies on the effects of capsaicin on lipid metabolism. In animal experiments, it has been shown to restrict the generation of white fat cells and the activation of lipoprotein lipase (LPL) to reduce the amount of body fat, showing anti-obesity effects [18-20].
In one study, methanol extract from hot pepper (Capsicum annuum L.) seeds decreased lipid accumulation in the adipocyte and significantly reduced the activity of glycerol-3-phosphate dehydrogenase. In addition, the expression of C/EBPβ, C/EBPα, and PPARγ, important adipogenic transcription factors, was significantly reduced when hot pepper seed extract was added to cells [28].
First of all, this research was administered that how the water extracts from several kinds of green pepper influence the expression of mRNA of lipoprotein lipase (LPL), a main lipoprotein enzyme that is composed in the fat cells and secreted into capillary vessels and cell envelopes to decompose the lipoproteins with rich triglycerides such as chylomicron (CM), very low density lipoprotein (VLDL) and low density lipoprotein (LD) into monoacylglycerols and free fatty acids [29].
Of the 7 pepper fruits, Cca demonstrated a significant effect in reducing the manifestation of LPL mRNA up to 50.9% when compared with the control group; Kca reduced 19.7% and Pca 9.9%, which were insignificant. The others increased the manifestation of LPL mRNA.
The LPL that is increased after eating a meal participates in lipoprotein metabolism, introducing the triglycerides into fat tissues and accumulating them [30]. Even if it does not directly participate in the decomposition metabolism of accumulated fats, it has been reported that the activation of LPL and the gene expression of LPL are radically decreased when an ultra-low calorie diet is adopted by someone suffering from obesity, leading to a reduction in weight [29]. In this respect, the water extract of Cca seems to have the function whereby it restricts the expression of LPL mRNA and lowers the accumulation of fats into the fat cells.
The extract of Chungyang gochu, which was previously selected as the most favorable material, was fractionated with the open column. Primary fraction was made only with the water. Of the 11 fractions, Fr. 5 and Fr. 6 showed the best effects in restricting the manifestation of LPL mRNA, 30.5% and 36.2%, respectively.
Therefore, the profiles of Fr. 5 and 6 with the best effects were checked through UPLC, and it was found that they contained a lot of components. Based on each UPLC profile, the composition ratio of solvents for the most effective separation was determined, and the secondary fractions were obtained through the separation. As the result of an analysis regarding the LPL mRNA reduction of secondary fractions, Fr. 6-6 (35.8%) demonstrated the best effect and Fr. 5-6 (35.3%) also showed a significant effect to restrict the manifestation of LPL.
To determine the compositions of selected fractions, their profiles were analyzed again through UPLC. The result showed that it is highly probable that the materials with the highest ratio in two fractions are the same material. On this basis, the composition ratio of solvents was determined for the 3rd fraction.
From the two previously-selected secondary fractions, tertiary fractions were obtained. Fr. 5-6-1, the tertiary fraction of 5-6, presented a 26.6% LPL restriction; Fr. 6-6-1, the tertiary fraction of 6-6, presented a 29.7% LPL restriction. As these were expected to be similar or the same as the two materials previously mentioned, the two tertiary fractions also showed similar effects. Although the effect of functional constituent on specific gene expression during fractionation processes is generally increased, there was no significant difference among fractions from fractionations in this study. It is supposed that because of the very low yield of functional constituent in a fraction or other unknown factors in fractionations, the inhibitory activity for LPL was not elevated through fractionation, nevertheless more exact cause should be elucidated through additional experiments in the future.
One of the well-known components of pepper known to have an anti-obesity effect is capsaicin [14,19-22]. However, according to current research, the material in this study is not capsaicin. Consequently, it seems necessary to investigate which material in green peppers restricts LPL, and which role it plays in doing so.
Figures and Tables
 | Fig. 1
Effect of plant water extracts on the viability of 3T3-L1 cells. The results are from three experiments and are expressed as mean ± SD. Cca, Chungyang gochu; Pca, Putgochu; Oca, Oiegochu; Kca, Kkwari putgochu; Gca, Green pepper; Yca, Yellow paprika; Rca, Red paprika; data values are expressed as mean ± SD of triplicate determinations. *P < 0.05 vs control, +P < 0.01 vs control, #P < 0.01 vs control. |
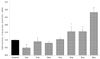 | Fig. 2
Effect of Capsicum Fructus extracts on the lipoprotein lipase (LPL) mRNA expression in 3T3-L1 cells. The results are from three experiments and are expressed as mean ± SD. The doses of all pepper extracts are 1,000 ppm. Cca, Chungyang gochu; Pca, Putgochu; Oca, Oiegochu; Kca, Kkwari putgochu; Gca, Green pepper; Yca, Yellow paprika; Rca, Red paprika; data values are expressed as mean ± SD of triplicate determinations. *P < 0.05 vs control, +P < 0.01 vs control, #P < 0.01 vs control. |
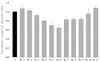 | Fig. 3
Effect of Cca first fractions on lipoprotein lipase (LPL) mRNA expression after non-differentiation in 3T3-L1 cells. The results are from three experiments and are expressed as mean ± SD. The doses of all fractions are 1,000 ppm. Cca, Chungyang gochu; data values are expressed as mean ± SD of triplicate determinations. *P < 0.05 vs control, +P< 0.01 vs control, #P < 0.01 vs control. |
 | Fig. 4
UPLC-UV chromatogram of fraction isolated from Cca. [A]: Cca Fr. 5, [B]: Cca Fr. 6. Cca, Chungyang gochu. |
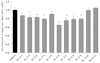 | Fig. 5
The effect of lipoprotein lipase (LPL) mRNA expression on sub-fractions of Cca Fr. 5 in 3T3-L1 cells. Results are from three experiments and are expressed as mean ± SD. The doses of all fractions are 1,000 ppm. Cca, Chungyang gochu; data values are expressed as mean ± SD of triplicate determinations. *P < 0.05 vs control, +P < 0.01 vs control, #P < 0.01 vs control. |
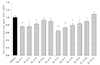 | Fig. 6
The effect of lipoprotein lipase (LPL) mRNA expression on sub-fractions of Cca Fr. 6 in 3T3-L1 cells. The results are from three experiments and are expressed as mean ± SD. The doses of all fractions are 1,000 ppm. Cca, Chungyang gochu; data values are expressed as mean ± SD of triplicate determinations. *P < 0.05 vs control, +P < 0.01 vs control, #P < 0.01 vs control. |
 | Fig. 7
UPLC-UV chromatogram of fraction isolated from Cca. [A]: Cca Fr. 5-6, [B]: Cca Fr. 6-6. Cca, Chungyang gochu. |
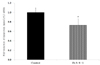 | Fig. 8
The effect of third fractions of CHE Fr. 5-6 on lipoprotein lipase (LPL) mRNA expression after non-differentiation in 3T3-L1 cells. Results are from three experiments and are expressed as mean ± SD. The dose of fraction is 1,000 ppm. Cca, Chungyang gochu; data values are expressed as mean ± SD of triplicate determinations. +P < 0.01 vs control. |
 | Fig. 9
The effect of third fractions of CHE Fr. 6-6 on lipoprotein lipase (LPL) mRNA expression after non-differentiation in 3T3-L1 cells. The results are from three experiments and are expressed as mean ± SD. The dose of fraction is 1,000 ppm. Cca, Chungyang gochu; data values are expressed as mean ± SD of triplicate determinations. +P < 0.01 vs control. |
References
1. Bray MS. Genomics, genes, and environmental interaction: the role of exercise. J Appl Physiol. 2000. 88:788–792.
2. Nam HK, Cho JH, Kim SH. Relations between self-selected intake of nutrients and body fat accumulation in rats fed ad libitum or for 8-hours a day. Korean J Nutr. 1992. 25:12–21.
3. Huh KB. Symposium: recent progress in obesity research: pathogenesis of obesity. Korean J Nutr. 1990. 23:333–336.
4. Sjöström LV, Lönn L, Chowdhury B, Grangård U, Lissner L, Sjöström D, Sullivan L. Angel A, Anderson H, Lau BD, Leiter L, Mendelson R, editors. The sagittal diameter is a valid maker of the visceral adipose tissue volume. Progress in Obesity Research: 7. 1996. London: John Libbey & Co. Ltd.;309–319.
6. Kim SG, An GH, Yoon SW, Lee YC, Ha SD. A study on dietary supplement to reduce obesity by the mechanism of decreasing lipid and carbohydrate absorption. Korean J Food Sci Technol. 2003. 35:519–526.
7. Vivero LE, Anderson PO, Clark RF. A close look at fenfluramine and dexfenfluramine. J Emerg Med. 1998. 16:197–205.

8. Park HS. Pharmacological therapy of obesity. J Korean Soc Study Obes. 2001. 10:118–127.
9. Kim YS. Drug treatment of obesity. J Korean Soc Endocrinol. 2001. 16:9–15.
10. Chang SH, Jun MH, Kang TB, Mun SH, Lee JH, Seong NS, Lee ST, Kim JB, Her E. The effect of Korean mistletoe extract M11C (non-lectin components) on IL-1β release and expression from macrophages. Immune Netw. 2001. 1:170–178.

11. Kim HP, Son KH, Chang HW, Kang SS. Anti-inflammatory plant flavonoids and cellular action mechanisms. J Pharmacol Sci. 2004. 96:229–245.

12. Chang CH. The future prospect of traditional Korean fermented foods. Korean J Diet Cult. 1988. 3:341–345.
13. Sambaiah K, Satyanarayana MN. Influence of red pepper and capsaicin on body composition and lipogenesis in rats. J Biosci. 1982. 4:425–430.

14. Watanabe T, Kawada T, Iwai K. Enhancement by capsaicin of energy metabolism in rats through secretion of catecholamine from adrenal medulla. Agric Biol Chem. 1987. 51:75–79.

15. Chae KY, Hong JS. The quality characteristics of Jeolpyon with different amounts of job's tears flour. Korean J Food Cookery Sci. 2007. 23:770–776.
16. Buck SH, Burks TF. The neuropharmacology of capsaicin: review of some recent observations. Pharmacol Rev. 1986. 38:179–226.
17. Hsu CL, Yen GC. Effects of capsaicin on induction of apoptosis and inhibition of adipogenesis in 3T3-L1 cells. J Agric Food Chem. 2007. 55:1730–1736.

18. Watanabe T, Kawada T, Yamamoto M, Iwai K. Capsaicin, a pungent principle of hot red pepper, evokes catecholamine secretion from the adrenal medulla of anesthetized rats. Biochem Biophys Res Commun. 1987. 142:259–264.

19. Kawada T, Hagihara K, Iwai K. Effects of capsaicin on lipid metabolism in rats fed a high fat diet. J Nutr. 1986. 116:1272–1278.

20. Srinivasan MR, Satyanarayana MN. Effect of capsaicin on skeletal muscle lipoprotein lipase in rats fed high fat diet. Indian J Exp Biol. 1989. 27:910–912.
21. Lee HJ. Studies on effects of natural plants water extracts on activities of enzymes related with fat metabolism [master's thesis]. 2009. Chuncheon: Kangwon National University.
22. Seo DJ. Anti-obesity effects of natural plants mixture in high fat diet-induced C57BL/6J mice [master's thesis]. 2010. Chuncheon: Kangwon National University.
23. Henderson DE, Slickman AM, Henderson SK. Quantitative HPLC determination of the antioxidant activity of capsaicin on the formation of lipid hydroperoxides of linoleic acid: a comparative study against BHT and melatonin. J Agric Food Chem. 1999. 47:2563–2570.

24. Kempaiah RK, Manjunatha H, Srinivasan K. Protective effect of dietary capsaicin on induced oxidation of low-density lipoprotein in rats. Mol Cell Biochem. 2005. 275:7–13.

25. Malmberg AB, Mizisin AP, Calcutt NA, von Stein T, Robbins WR, Bley KR. Reduced heat sensitivity and epidermal nerve fiber immunostaining following single applications of a high-concentration capsaicin patch. Pain. 2004. 111:360–367.

26. Surh YJ, Lee RC, Park KK, Mayne ST, Liem A, Miller JA. Chemoprotective effects of capsaicin and diallyl sulfide against mutagenesis or tumorigenesis by vinyl carbamate and N-nitrosodimethylamine. Carcinogenesis. 1995. 16:2467–2471.

27. Reddy AC, Lokesh BR. Studies on anti-inflammatory activity of spice principles and dietary n-3 polyunsaturated fatty acids on carrageenan-induced inflammation in rats. Ann Nutr Metab. 1994. 38:349–358.

28. Jeon G, Choi Y, Lee SM, Kim Y, Jeong HS, Lee J. Anti-obesity activity of methanol extract from hot pepper (Capsicum annuum L.) seeds in 3T3-L1 adipocyte. Food Sci Biotechnol. 2010. 19:1123–1127.

29. Lee JJ, Choi MS, Chung CS, Choi BD. Effects of food restriction on rat adipose tissue lipoprotein lipase activity and lipogenesis. Korean J Exerc Nutr. 2003. 7:135–141.
30. Lee SK, Kim OS, Kim JY, Lee KH, Baik HW, Jo YS, Kim HJ, Kim BJ, Park KS, Ahn KJ. Lipoprotein lipase hind 3 polymorphism, blood lipid levels and obesity in Korean women. 2007. In : 2007 Annual Meeting of Korean Endocrine Society; 2007 May 3-4; Seoul, Korea. Seoul: Korean Endocrine Society;133.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download