Abstract
In Korea, chestnut production is increasing each year, but consumption is far below production. We investigated the effect of chestnut extracts on antioxidant activity and anticancer effects. Ethanol extracts of raw chestnut (RCE) or chestnut powder (CPE) had dose-dependent superoxide scavenging activity. Viable numbers of MDA-MD-231 human breast cancer cells, DU145 human prostate cancer cells, and AGS human gastric cancer cells decreased by 18, 31, and 69%, respectively, following treatment with 200 µg/mL CPE for 24 hr. CPE at various concentrations (0-200 µg/mL) markedly decreased AGS cell viability and increased apoptotic cell death dose and time dependently. CPE increased the levels of cleaved caspase-8, -7, -3, and poly (ADP-ribose) polymerase in a dose-dependent manner but not cleaved caspase-9. CPE exerted no effects on Bcl-2 and Bax levels. The level of X-linked inhibitor of apoptosis protein decreased within a narrow range following CPE treatment. The levels of Trail, DR4, and Fas-L increased dose-dependently in CPE-treated AGS cells. These results show that CPE decreases growth and induces apoptosis in AGS gastric cancer cells and that activation of the death receptor pathway contributes to CPE-induced apoptosis in AGS cells. In conclusion, CPE had more of an effect on gastric cancer cells than breast or prostate cancer cells, suggesting that chestnuts would have a positive effect against gastric cancer.
The chestnut belongs to the genus Castanea in the Fagaceae family and is native to temperate regions in Asia, Europe, North America, and North Africa. There are 13 species, and the Japanese, Korean (Castanea crenata Sieb et Zucc.), Chinese (Castanea mollissima Blume), European (Castanea sativa Mill), and American (Castanea dentate Borkhausen) chestnuts are mainly cultivated for fruit [1].
Currently, chestnut trees are cultivated on approximately 80,000 ha in Korea, yielding 94,000 tons, and most are produced in Gongju, (10,246 tons of chestnut in 4,515 ha of farmland) [2]. Chestnut trees have been cultivated throughout history, as they are used in traditional Korean ceremonies. Chestnuts are also consumed as a favorite food because of their good nutrition. However, to increase chestnut consumption, it is necessary to demonstrate that chestnuts are healthy.
According to oriental medicine, chestnuts cure stomach cancer, epistaxis, and hemafecia, but the components and their functions that cure diseases have not been systematically tested [3]. The antioxidant activity of the chestnut inner shell, leaves, and flowers has been tested [4,5]. Because the flesh and inner shell of chestnuts have antioxidant activity, we investigated whether uncooked chestnuts and processed chestnut powder extracts also have antioxidant effects. The objective of this study was to test the activity of chestnut extracts against cancer cell proliferation and to examine the molecular mechanisms underlying this phenomenon as a trial for developing a novel functional anticancer food. Thus, we investigated the effect of chestnuts on cell proliferation and apoptosis using MDA-MD-231 human breast cancer cells, DU145 human prostate cancer cells, and AGS human gastric cancer cells.
Raw chestnuts and chestnut powder were produced in Gongju, Korea. According to Gongju City, chestnut powder is obtained after the flesh and shell, including the outer and inner parts, are ground and mixed with water. This powder sells in the market under the name chestnut starch. The reagents employed in this study were purchased from the indicated suppliers: Anti-β-actin antibody and 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) were obtained from Sigma-Aldrich Co. (St. Louis, MO, USA); Dulbecco's Modified Eagle's Medium/Ham's F-12 nutrient mixture (DMEM/F-12) was purchased from Gibco BRL (Gaithersburg, MD, USA); fetal bovine serum (FBS) was obtained from Cambrex Bio Technology (Walkersville, MD, USA); horse-radish peroxidase (HRP)-conjugated anti-rabbit and anti-mouse IgG were purchased from Amersham Biosciences (Arlington Heights, IL, USA); antibodies against cleaved caspase-3, cleaved caspase-7, cleaved caspase-9, cleaved poly (ADP-ribose) polymerase (PARP), caspase-8, Bid, and X-linked inhibitor of apoptosis protein (XIAP) were obtained from Cell Signaling Technology (Beverly, MA, USA); antibody against tumor necrosis factor-related apoptosis including ligand (TRAIL) was obtained from BD Pharmingen (Franklin Lake, NJ, USA); antibodies against Fas, Fas ligand (Fas-L), Bcl-2, and Bax were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA); and antibodies against death receptor (DR) 4 and 5 were obtained from Imgenex (San Diego, CA, USA). All other materials were obtained from Sigma-Aldrich Co.
Preparation of aqueous extracts. Chestnut powder (0.5 kg) was extracted with water for 30 min using a sonication method. The extract was centrifuged at 6,000 rpm for 20 min to remove the residue. This procedure was repeated twice more with a sonication time of 30 min. The upper phase was collected and lyophilized (CPW). The CPW yield was 5.05 g (0.61%). Pulverized raw chestnut (1.5 kg) was extracted with water by refluxing at 100℃ for 3 hr. The extract was centrifuged at 6,000 rpm for 20 min to the separate residue. The upper phase was divided into a non-mucous (RCW1) and mucous (RCW2) fractions. Each fraction was lyophilized, and the yields were 78.92 g (5.35%) and 60.57 g (4.4%), respectively.
Preparation of ethanol extracts. Pulverized raw chestnut or chestnut powder (1.5 kg each) was subjected to three extractions with 95% ethanol (4.5 L) for 3 h and then filtered (Advantec filter paper, type 2; Advantec, Tokyo, Japan). Each combined filtrate was then concentrated in vacuo at 45℃ (Bchi Rotavapor R-220, Flawil, Switzerland). The yields of the pulverized raw chestnut extract (RCE) and chestnut powder extract (CPE) were 68.30 g (4.55%) and 1.89 g (0.13%), respectively.
Photochemiluminescence was used to determine the antioxidant activity of the extracts. We used an antioxidant capacity of water extracts (ACW) kit (Analytik Jena AG Co., Jena Germany) for the water soluble extracts and an antioxidant capacity of lipid extracts (ACL) kit for the water insoluble extracts (Analytik Jena), in which luminal plays a double role as a photosensitizer and an oxygen radical detection reagent, according to the manufacturer's instructions. Results were calculated in ascorbate equivalents for ACW and trolox equivalents for ACL.
MDA-MB-231 human breast cancer cells, DU145 human prostate cancer cells, and AGS human gastric cancer cells were obtained from the American Type Culture Collection (Manassas, VA, USA). All cells were maintained in DMEM/F12 containing 100 mL/L FBS with 100,000 U/L penicillin and 100,000 mg/L streptomycin at 37℃ in 5% CO2. We plated cells in 24-well plates with DMEM/F-12 containing 100 mL/L FBS for 24 h to examine the effect of the extracts on cell growth. Prior to extract treatment, the cell monolayers were rinsed and serum-deprived for 24 h with DMEM/F-12 containing 10 mL/L FBS (serum-deprivation medium). After serum-deprivation, we replaced the serum-deprivation medium with or without the indicated concentrations of extracts. Viable cell numbers were estimated by the MTT assay, as described previously [6,7].
AGS cells were plated in 24-well plates and treated with various concentration of CPE for 24, 48, or 72 hr. The cells were assayed to quantitatively determine the mono- and oligo-nucleosomes released into the cytoplasm using a Cell Death Detection ELISAPLUS kit (Roche, Mannheim, Germany), according to the manufacturer's instruction.
The cells were cultured in 100 mm dishes and treated with CPE using the method described above. Total cell lysates were prepared as previously described [8]. We determined protein content in total cell lysates using the BCA Protein Assay kit (Pierce, Rockford, IL, USA). The proteins in the total cell lysates were resolved on a sodium dodecyl sulfate (SDS) 4-20% or 10-20% polyacrylamide gels and transferred to a polyvinylidene fluoride membrane (Millipore, Bedford, MA, USA). Western blot analyses were performed as previously described [7]. We detected the signals based upon the enhanced chemiluminescence method using the Immobilon™ Western Chemiluminescent HRP Substrate (Millipore). The abundance of each band was quantified using the Bio-profile Bio-1D application (Vilber-Lourmat, Marine la Vallee, France), and expression levels were normalized to β-actin.
We performed preliminary studies to determine the antioxidant activity of the chestnut extracts. The superoxide radical scavenging effect of water soluble extracts (such as RCW1, RCW2, and CPW) or ethanol extracts (such as RCE and CPE) were compared to ascorbate or trolox, respectively. The RCE and CPE revealed dose dependent superoxide scavenging activity (Table 1).
We determined whether the chestnut extracts moderated growth of MDA-MD-231, DU145, and AGS cells. In MDA-MB-231 cells, the water soluble chestnut extracts (RCW1, RCW2, CPW) and CPE induced a mild decrease in viable cell numbers within 24 hr after adding 200 µg/mL of extract (Fig. 1A). In DU145 cells, the water soluble extracts such as RCW1, RCW2, and CPW and the ethanol extract of raw chestnut, RCE, exerted no effects on cell growth. However a 24 hr treatment of DU145 cells with 200 µg/mL CPE resulted in a 30.5% decrease in viable cell numbers (Fig. 1B). In AGS cells, the water soluble extracts significantly increased cell growth. RCE exerted no effect on AGS cell growth as in the other cancer cell lines. However, a 24 hr treatment of AGS cells with 200 µg/mL CPE resulted in a 69.4% decrease in viable cell numbers (Fig. 1C).
We found that CPE had the greatest cancer cell growth inhibiting effect, among the various chestnut extracts and that AGS cells were most affected by CPE among the various cancer cell lines. AGS cells were treated with various CPE concentrations (0, 25, 50, 75, 100, 200 µg/mL) to determine whether CPE inhibits stomach cancer cell growth. As a result, CPE markedly decreased AGS cell viability dose-dependently and time-dependently (Fig. 2A). To determine if the decrease in cell growth observed after CPE treatment was due to the induction of apoptosis, AGS cells were treated for 24, 48, or 72 hr with various CPE concentrations (0, 50, 100, or 200 µg/mL) and the degree of apoptosis was estimated using an ELISA. CPE treatment induced an increase in the numbers of apoptotic AGS cells in a dose-dependent manner (Fig. 2B).
Because CPE induced apoptosis in AGS cells, we assessed the effects of CPE on the activation of caspases and PARP cleavage. AGS cells were treated for 48 hr with various concentrations of CPE, and total cell lysates were prepared for Western blotting with antibodies that detect the cleaved forms of the enzymes. CPE increased the protein levels of cleaved caspase-8, -7, and -3 in a dose-dependent manner. The level of cleaved caspage-9 was not altered following CPE treatment (Fig. 3A). Additionally, CPE increased the PARP cleavage product in a concentration-dependent manner (Fig. 3B).
We then assessed the effects of CPE on the levels of Bcl-2, an antiapoptotic Bcl-2 family protein and Bax, a pro-apoptotic Bcl-2 family protein. CPE exerted no effects on the levels of Bcl-2 and Bax. (Fig. 4A).
XIAP is an inhibitor of apoptotic proteins (IAP) and inhibits apoptosis by inhibiting caspase activity. We determined whether CPE decreased XIAP protein levels and found that the XIAP protein levels decreased slightly with CPE treatment (Fig. 4B).
CPE had no effect on caspase-9 activity (Fig. 3A) or Bcl-2 and Bax expression (Fig. 4A). However, CPE effectively increased caspase-8 activity (Fig. 3A). These results imply that CPE induced apoptosis by a death receptor mediated pathway. To confirm these results, we determined whether CPE increased the levels of TRAIL, DR4, Fas, and Fas-L proteins using Western blotting of total cell lysates. As a result, the levels of TRAIL, DR4, and Fas-L dose-dependently increased in CPE treated cells, but CPE did not affect the Fas level (Fig. 5).
Oriental medicine and folk remedies have found that chestnuts promote digestion and strengthen the stomach. The dictionary of traditional Chinese medicine states that "Chestnut remedies gastric cancer, nosebleeds, and bloody excrement". The Donguibogam states that chestnuts "fortify oneself, strengthen the stomach, reinforce vigor, and can be used as food". Furthermore, the starch in chestnuts is an easily-digested, high quality sugar that contains gastric function enzymes, and a folk remedy suggests that chewing roasted chestnuts helps reduce stomach aches and diarrhea [4]. In the current study, we attempted to determine whether chestnut extracts have healthy functions. First, we detected the superoxide radical scavenging effects of aqueous and ethanol extracts from raw chestnuts and chestnut powder. As a result, we found that the ethanol extract increased antioxidant activity in a dose dependent manner. Several studies have indicated the antioxidant activities of chestnut bark, flower, leaf, and fruit extracts [5,6]. We investigated the effect of CPE on cell proliferation in several cell lines to determine whether this antioxidant effect could be extended to an anticancer effect, and CPE restrained cancer cell proliferation. In particular, CPE strongly retarded cell proliferation and promoted apoptosis in the AGS gastric cancer cell line.
Cancer development and progression is a very complex process. Cancer chemoprevention has gained more attention, presumably because it involves the use of dietary bioactive compounds to prevent, inhibit, or reverse the development of cancer [9]. Previous investigations have shown that cancer cell proliferation decreases by about 40% after injecting L1210 cells into the peritoneal cavity of mice and transplanting cancer cells [10]. In the present study, CPE had an antioxidant effect and promoted apoptosis in cancer cells, particularly in gastric cancer cells, and this result supports the folk remedies.
We studied the mechanism of apoptosis in CPE (0-200 µg/mL), which had the most antioxidant and anti-proliferation activity in AGS cells. Apoptosis occurs through an intrinsic pathway (also known as the mitochondrial pathway) and an extrinsic pathway (also known as the death receptor pathway) [11]. Apoptosis is initiated through the mitochondrial pathway following physiological conditions, such as oxidative stress, mitochondrial disturbance, and DNA damage. This pathway is involved in the permeabilization of the outer mitochondrial membrane, which facilitates cytochrome c release into the cytoplasm. Cytochrome c binds the caspase adaptor apoptotic protease-activating factor-1 (Apaf-1), thereby triggering the apoptotic cascade by activating procaspase 9 and forming an apoptosome complex. This apoptosome, in turn, activates the downstream effector caspases-3, -6, and -7, leading to apoptosis [12,13]. The typical executioners of apoptosis are proteolytic enzymes called caspases. Our results clearly demonstrate that CPE increased the activation of caspase-8, 7, and 3 and induced PARP cleavage. Caspase activation may be one of the major mechanisms by which CPE induced apoptosis.
Another important key player in the mitochondrial pathway of apoptosis is the Bcl-2 family, which consists of pro-apoptotic proteins and anti-apoptotic proteins. Bax, a pro-apoptotic member of the Bcl-2 family, induces the release of cytochrome c from mitochondria, thereby activating caspase-9. Bax promotes apoptosis by stimulating the release of the Smac/DIABLO protein from mitochondria, thereby inactivating IAPs, such as XIAP, cIAP1, cIAP2, and survivin [14]. However, Bcl-2 is an anti-apoptotic member of the Bcl-2 family that binds and inactivates Apaf-1. In the present study, we observed that CPE did not alter the levels of Bcl-2 and Bax. Moreover, the caspase-9 levels were not affected by CPE treatment. These results suggest that Bcl-2 family is not the main cause of the apoptotic effect of CPE.
The extrinsic pathway of apoptosis is induced by ligand binding death receptors. The most important ligand-death receptor system includes tumor necrosis factor (TNF)-TNF receptor-1, Fas-L-Fas (also called CD95 or Apo1) and TRAIL-TRAIL receptor (including TRAIL-R1, also called DR4 and TRAIL-R2 or DR5). Binding of their receptors by the respective ligands leads to receptor oligomerization and recruitment of death signal adaptor proteins. For example, binding of Fas-L to Fas leads to recruitment of Fas-associated death domain (FADD) [15]. The oligomerized receptors and FADD form a death-inducing signaling complex, which binds to initiator caspases (caspase 8 and 10), thereby triggering the caspase cascade such as activation of caspase 3, which leads to apoptotic events. Fas and Fas-L are constitutively expressed in the gastrointestinal tract [16] and in gastric [17,18] and colon cancers [19]. Thus, the Fas/Fas-L system is an important target for apoptosis induction in gastric and colon cancers. Importantly, TRAIL preferentially induces apoptosis in cancer cells while exhibiting little or no toxicity in normal cells [20]. The TRAIL receptors, DR4 and DR5, are highly expressed in many cancers including gastric and colorectal cancers [14,20].
Initiator caspases, caspase-8 and -9, are activated by other pathways. Caspse-8 is activated by a combined cell death receptor, such as Fas, with a cell death receptor ligand in the external pathway, whereas caspase-9 is activated by the release of cytochrome c from the mitochondrial membrane, which activates the internal pathway [21-23]. In this study, CPE had no effect on caspase-9 activity (Fig. 3A) or Bcl-2 and Bax expression (Fig. 4). As shown in Fig. 3A, CPE effectively increased caspase-8 activity. These results suggest that CPE induced apoptosis by the death receptor mediated pathway. To confirm these results, we determined whether CPE increased the levels of death receptors using Western blotting of total cell lysates. As a result, Fas-L, DR4, and Trail levels were dose-dependently increased in the CPE-treated cells, indicating that activation of the death receptor pathway contributed to CPE-induced apoptosis in AGS cells. Further studies will be required to delineate the mechanisms by which CPE induced caspase-8 activation in AGS cells.
IAPs are a group of important apoptosis regulatory proteins. XIAP directly binds and inhibits key effector caspases, such as caspase 3, 7, and 9, thereby inhibiting apoptosis [24]. Downregulation of XIAP sensitizes gastric cancer cells to Trail and chemotherapeutic drug-induced apoptosis [25,26]. In the present study, the levels of XIAP decreased slightly with CPE treatment (Fig. 4).
In conclusion, considering that CPE had more of an effect on gastric cancer cells than breast cancer or prostate cancer cells in this study, chestnuts may function more effectively to aid in the treatment of gastric cancer. We demonstrated that CPE decreased growth and induced apoptosis in AGS gastric cancer cells, which was mediated by caspase activation. Our results indicate that CPE activates caspases mainly through the extrinsic pathway. Further studies should explore the anticancer effect of chestnuts and the mechanism through in vivo studies and investigate the active ingredients in chestnut extracts that have an anticancer effect.
Figures and Tables
Fig. 1
Effect of chestnut extracts on the viable cell numbers of MDA-MD-231, DU145, and AGS cells. (A) MDA-MB-231, (B) DU145, and (C) AGS cells were plated at a density of 50,000 cells/well in 24-well plates with DMEM/F12 supplemented with 10% FBS. One day after plating, the monolayers were serum-deprived with DMEM/F12 supplemented with 1% FBS serum-deprivation medium for 24 h. After serum deprivation, the cells were incubated in serum-deprivation medium in the absence or presence of 200 µg/mL viscous, aqueous extract of raw chestnut (RCW1), non-viscous, aqueous extract of raw chestnut (RCW2), aqueous extract of chestnut powder (CPW), ethanol extract of raw chestnut (RCE), or an ethanol extract of chestnut powder (CPE) for 24 hr. Cell numbers were estimated by the MTT assay. Each bar represents the mean ± SEM (n = 6). Means at a time without a common letter differ significantly, P < 0.05.

Fig. 2
Effect of an ethanol extract of chestnut powder (CPE) on AGS cell viability and apoptosis. AGS cells were plated at a density of 50,000 cells/well in 24-well plates with DMEM/F12 supplemented with 10% FBS. One day after plating, the monolayers were serum-deprived with DMEM/F12 supplemented with 1% FBS serum-deprivation medium for 24 h. After serum deprivation, cells were incubated in serum-deprivation medium in the absence or presence of various concentrations of CPE. (A) Cell numbers were estimated by the MTT assay. (B) Apoptotic cells were detected with the Cell death detection ELISAPLUS. Each bar represents the mean ± SEM (n = 6). Means without a common letter differ significantly, P < 0.05.

Fig. 3
Effect of an ethanol extract of chestnut powder (CPE) on the levels of cleaved caspases (A) and poly (ADP-ribose) polymerase (PARP) (B) in AGS cells. Cells were plated and treated with various concentrations of CPE for 48 h as described in Fig. 2. Total cell lysates were analyzed by Western blotting with the indicated antibodies. Photographs of chemiluminescent detection of the blots, which are representative of three independent experiments, are shown. The relative abundance of each band to their own β-actin concentration was quantified and control levels were set to 1. The adjusted mean ± SEM is shown above each blot. Means without a common letter differ significantly, P < 0.05.
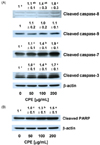
Fig. 4
Effect of an ethanol extract of chestnut powder (CPE) on the levels of Bcl-2, Bax, and an inhibitor of apoptosis proteins (XIAP) in AGS cells. Cells were plated and treated with various concentrations of CPE for 48 h as described in Fig. 2. Total cell lysates were analyzed by Western blotting with the indicated antibodies. Photographs of chemiluminescent detection of the blots, which are representative of three independent experiments, are shown. The relative abundance of each band to their own β-actin concentration was quantified, and the control levels were set to 1. The adjusted mean ± SEM is shown above each blot. Means without a common letter differ significantly, P < 0.05.
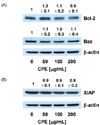
Fig. 5
Effect of an ethanol extract of chestnut powder (CPE) on the levels of tumor necrosis factor-related apoptosis including ligand (TRAIL), DR4, Fas, and Fas-L in AGS cells. Cells were plated and treated with various concentrations of CPE for 48 h as described in Fig. 2. Total cell lysates were analyzed by Western blotting with the indicated antibodies. Photographs of chemiluminescent detection of the blots, which are representative of three independent experiments, are shown. The relative abundance of each band to their own β-actin concentration was quantified, and the control levels were set to 1. The adjusted mean ± SEM is shown above each blot. Means without a common letter differ significantly, P < 0.05.
References
1. Paillet FL. Chestnut: history and ecology of a transformed species. J Biogeogr. 2002. 29:1517–1530.

2. Kim JS, Jung BH, Joo RW, Choi SI. Marketing of chestnut and economic analysis of chestnut cultivation. Korean J For Econ. 2004. 12:12–21.
3. Heo J. Doguibogam. 2001. Seoul: Hanmibook press;1153.
4. Vekiari SA, Gordon MH, García-Macías P, Labrinea H. Extraction and determination of ellagic acid content in chestnut bark and fruit. Food Chem. 2008. 110:1007–1011.

5. Barreira JCM, Ferreira ICFR, Oliveira MBPP, Pereira JA. Antioxidant activities of the extracts from chestnut flower, leaf, skins and fruit. Food Chem. 2008. 107:1106–1113.

6. Kim EJ, Kang IJ, Cho HJ, Kim WK, Ha YL, Park JH. Conjugated linoleic acid downregulates insulin-like growth factor-I receptor levels in HT-29 human colon cancer cells. J Nutr. 2003. 133:2675–2681.

7. Cho HJ, Kim WK, Kim EJ, Jung KC, Park S, Lee HS, Tyner AL, Park JH. Conjugated linoleic acid inhibits cell proliferation and ErbB3 signaling in HT-29 human colon cell line. Am J Physiol Gastrointest Liver Physiol. 2003. 284:G996–G1005.
8. Na MH, Seo EY, Kim WK. Effects of α-lipoic acid on cell proliferation and apoptosis in MDA-MB-231 human breast cells. Nutr Res Pract. 2009. 3:265–271.

9. Chen C, Kong AN. Dietary cancer-chemopreventive compounds: from signaling and gene expression to pharmacological effects. Trends Pharmacol Sci. 2005. 26:318–326.

10. Jeon BG, Jeong HW, Lee JR, Ji JM. The development of functional beverage from the inner skin of the chestnut Castanea crenata (II) - Physiological effects of chestnut inner skin tea, brown rice-green tea and Cassia tora tea in mouse and rat. Korean J Food Nutr. 2000. 13:411–418.
11. Lorenzo HK, Susin SA. Therapeutic potential of AIF-mediated caspase-independent programmed cell death. Drug Resist Updat. 2007. 10:235–255.

12. Oliver L, Vallette FM. The role of caspases in cell death and differentiation. Drug Resist Updat. 2005. 8:163–170.

13. Iannolo G, Conticello C, Memeo L, De Maria R. Apoptosis in normal and cancer stem cells. Crit Rev Oncol Hematol. 2008. 66:42–51.

14. Qiao L, Wong BC. Targeting apoptosis as an approach for gastrointestinal cancer therapy. Drug Resist Updat. 2009. 12:55–64.

15. Thorburn A, Behbakht K, Ford H. TRAIL receptor-targeted therapeutics: resistance mechanisms and strategies to avoid them. Drug Resist Updat. 2008. 11:17–24.

16. Sträter J, Möller P. CD95 (Fas/APO-1)/CD95L in the gastrointestinal tract: fictions and facts. Virchows Arch. 2003. 442:218–225.

17. Nagashima H, Mori M, Sadanaga N, Mashino K, Yoshikawa Y, Sugimachi K. Expression of Fas ligand in gastric carcinoma relates to lymph node metastasis. Int J Oncol. 2001. 18:1157–1162.

19. Korkolopoulou P, Saetta AA, Levidou G, Gigelou F, Lazaris A, Thymara I, Scliri M, Bousboukea K, Michalopoulos NV, Apostolikas N, Konstantinidou A, Tzivras M, Patsouris E. c-FLIP expression in colorectal carcinomas: association with Fas/FasL expression and prognostic implications. Histopathology. 2007. 51:150–156.

20. Mahalingam D, Szegezdi E, Keane M, de Jong S, Samali A. TRAIL receptor signalling and modulation: are we on the right TRAIL? Cancer Treat Rev. 2009. 35:280–288.

21. Budihardjo I, Oliver H, Lutter M, Luo X, Wang X. Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol. 1999. 15:269–290.

23. Baker SJ, Reddy EP. Modulation of life and death by the TNF receptor superfamily. Oncogene. 1998. 17:3261–3270.

24. Verhagen AM, Silke J, Ekert PG, Pakusch M, Kaufmann H, Connolly LM, Day CL, Tikoo A, Burke R, Wrobel C, Moritz RL, Simpson RJ, Vaux DL. HtrA2 promotes cell death through its serine protease activity and its ability to antagonize inhibitor of apoptosis proteins. J Biol Chem. 2002. 277:445–454.

25. LaCasse EC, Cherton-Horvat GG, Hewitt KE, Jerome LJ, Morris SJ, Kandimalla ER, Yu D, Wang H, Wang W, Zhang R, Agrawal S, Gillard JW, Durkin JP. Preclinical characterization of AEG35156/GEM 640, a second-generation antisense oligonucleotide targeting X-linked inhibitor of apoptosis. Clin Cancer Res. 2006. 12:5231–5241.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download