Abstract
Zinc is an essential trace element required for bone formation, however not much has been clarified yet for its role in osteoblast. We hypothesized that zinc would increase osteogenetic function in osteoblasts. To test this, we investigated whether zinc treatment enhances bone formation by stimulating osteoblast proliferation, bone marker protein alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells. MC3T3-E1 cells were cultured and treated with various concentrations of zinc (0, 1, 3, 15, 25 uM) along with a normal osteogenic medium (OSM) as control for 1, 5, 10 days. As measured by MTT assay for mitochondrial metabolic activity, cell proliferation was stimulated even at low zinc treatment (1-3 µM) compared to OSM, and it was stimulated in a zinc concentration-dependent manner during 5 and 10 days, with the most pronounced effect at 15 and 25 uM Zn. Cellular (synthesized) alkaline phosphatase (ALP) activity was increased in a zinc concentration-dependent manner, so did medium (secreted) ALP activity. Cellular collagen concentration was increased by zinc as time went by, therefore with the maximum zinc stimulatory effect in 10 days, and medium collagen concentration showed the same pattern even on 1 and 5 day. This zinc stimulatory effect of collagen synthesis was observed in cell matrix collagen staining. The study results imply that zinc can increase osteogenic effect by stimulating cell proliferation, ALP activity and collagen synthesis in osteoblastic cells.
Bone mass is maintained through the repeated cycle of destruction and rebuilding to maintain the balance between bone formation and bone resorption which are controlled by osteoblasts and osteoclasts [1]. Bone formation is a complex biological process and involves several tightly regulated gene expression patterns of bone-related proteins [2]. The basic processes that involve bone formation are considerably similar to the development of other organs except for the unique mineralization phase involving the attachment of calcium and phosphate, along with collagenous and other noncollagenous proteins, to form the structural integrity and framework of mature skeletal bone. Normal maturation of bone cell relies upon complex cell-cell and cell matrix interactions in a highly temporally and spatially coordinated manner [3] that involves successive developmental stages including proliferation, bone matrix maturation, mineralization and bone-remodeling [4]. The regulation of hormones and other molecules secreted by either osteoblast or osteoclast tightly controls the dynamic and complex process of bone remodeling thus bone formation [5].
Zinc is an essential trace element to the structure and function of metabolism [6]. Zinc deficiency decreases bone weight and delays growth in bone metabolism. Zinc deficiency results in a retardation of bone growth, development, and maintenance of bone health [7,8]. Zinc has a stimulatory effect on bone formation and mineralization in vivo and vitro [9,10]. Zinc is also implicated in the stimulation of collagen production in rat femora and calvaria [11]. It has been shown that zinc acts to increase bone formation and mineralization, decrease bone resorption and stimulate alkaline phosphatase (ALP) activity, in both calvaral organ cultures and osteoblast cell cultures [9,11-13]. The cellular mechanism of zinc action in osteoblastic cells has not fully clarified. Zinc demonstrated stimulating cell proliferation and differentiation, as well as protein synthesis in osteoblatic cells [14,15]. Once bone matrix synthesis begins in osteoblast culture models, such as osteoblastic MC3T3-E1 cells, the cells differentiate in accordance with the gene activation and protein synthesis of osteoblast markers. Alkaline phosphatase (ALP) is one of the most representative bone marker proteins for osteoblast differentiation [16,17].
In this study, we evaluated whether zinc could increase osteoblast proliferation and differentiation which can be measured by collagen synthesis and alkaline phosphatase (ALP) activity as an indicator for osteoblast differentiation, using mouse osteoblastic MC3T3-E1 cells. In addition, matrix Ca deposits by zinc treatment were also examined for matrix mineralization. The study results showed the anabolic role of zinc on bone formation in osteoblast model.
Mouse calvariae origin osteoblastic MC3T3-E1 subclone 4 (ATCC, CRL-2593, subclone 4, highly differentiating osteoblast) was obtained from ATCC Cell Bank (Manassas, VA, USA). Cell culture reagents (α-MEM, penicillin & streptomycin and fetal bovine serum) were obtained from Gibco Laboratories (Grand Island, NY, USA). Chemicals were obtained from Sigma (St Louis, MO), unless otherwise noted. All consumables reagents and plastic wares used were trace element free analysis grade.
MC3T3-E1 cells were seeded at a density of 1×105 cells/mL and maintained in regular growth medium (α-MEM with 10% FBS, 1mM sodium pyruvate and 1% penicillin and streptomycin) in a humidified atmosphere of 5% CO2 at 37℃. At 80% confluence, cells were treated with various concentrations of zinc (0, 1, 3, 15, 25 uM) in the form of ZnCl2 with the addition of 5 µM TPEN (N,N,N',N'-tetrakis-(2-pyridylmethyl)-Ethylenediamine, intracellular cell-permeable zinc-chelator) for the depletion of zinc in the media serum. TPEN is the intercellular zinc chelating agent and each TPEN molecule binds a single zinc atom through 4 pyridine rings and so the molar ratio is one [18]. Zinc addition at these levels also ensured maintenance of cell viability and the physiological relevance of the zinc treatment conditions. Normal physiological zinc level in mammalian animals and humans is about 15 µM [6,19]. ZnCl2 or ZnSO4 is normally used for the cellular zinc treatment, and in the present study, ZnCl2 was used for external zinc treatment for the cell culture. Normal osteogenic differentiation medium (OSM, growth media plus 10 mM β-glycerophosphate and 50 µg/mL L-ascorbic acid without TPEN addition) was used as control group. Cells were cultured with zinc-treated media for 1, 5 and 10 days and the media were changed every 2-3 days.
Cell proliferation by treatment of 0-25 µM zinc under 5 µM TPEN during 1, 5 and 10 days was determined by MTT assay on the metabolic reduction of 3-[4,5-dimethylthiazol-2-y]-2,5-diphenyltetrazolium bromide (MTT). Briefly, cells were plated at seeding MC3T3-E1 cells (1×104 cells/well in 96-well plate) and maintained in growth media for 24 hours at 5% CO2 at 37℃. At 80% confluence, the cells were treated with zinc for 1, 5 and 10 days. Then, 10 µl of MTT solution was added in each well and incubated at 37℃ for 3 hours to allow for the formation of formazan crystals. After formation of formazan crystals, MTT medium was then aspirated and replaced with solubilization solution (DMSO) for dissolving the formazan crystals. The plates were read on Micro Elisa reader (Asys Hitee, Expert 96, Asys Co, Austria) at 570 nm.
ALP activity of MC3T3-E1 cells and media were measured. Cells were washed with PBS and lysed in 1 mL of 0.02% Triton-X in 0.9% NaCl before subjecting to sonication for 30s twice on ice. The sonicated lysates were centrifuged for 15 min at 12,000 rpm. The supernatant was kept at -20℃ until analysis. The activity of ALP in cell lysates and media was measured using ρ-nitrophenylphosphate (PNPP) as the substrate and the optical density for the enzyme products was measured at 405 nm. Cellular protein concentration was measured by the Bradford method, with bovine serum albumin as the standard. The activity of cellular ALP expressed as nmol PNPP/mg of protein/min. The activity of medium ALP was expressed as nmol PNPP/mL/min.
Collagen concentration in the cells and the media were measured. The cell lysates and medium samples were centrifuged and the supernatants were stored at -70℃ until use. Collagen standards were prepared using collagen type I, and calibration was obtained for their absorbance values in a range of 0 to 400 µg/ml. After the samples being thawed, the collected supernatants were centrifuged again. Fifty microliters of sample and collagen standard were added in each well of a 96-well plate. The plates containing the supernatant, sample, and standard were incubated at 37℃ consecutively for 16 h in a humidified atmosphere, and 24 h in a dry oven. After incubation, all the wells were washed 3 times with 200 µl of distilled water. Clean wells were filled with 100 µl of 0.1% (w/v) Sirius red F3BA in saturated picric acid. Staining lasted 1 h at 37℃ in a dry incubator. The plates were finally washed 5 times for 10 seconds with 200 µl of 10 mM HCl solution. The samples underwent a further step in 0.1 N NaOH for 5 minutes at room temperature to dissolve the adsorbed collagen. The solution was mixed several times and transferred into a clean plate. Finally, the absorbance at 540 nm was measured using an ELISA reader (TECAN, Austria). Collagen concentration was normalized to the protein concentration which was measured by Bradford method.
To evaluate extracellular matrix collagen synthesis, cellular matrix was stained using Van Gieson's stain method. At days of 1, 5 and 10, cells were washed with PBS twice, and then fixed with 2% formaldehyde for 15 min at 4 C. The cells were stained with Van Gieson's stain for 5 min at room temperature and rinsed with deionized water twice. The images of stained cells were captured using a phase contrast microscope with digital camera (IM50, Leica, Germany).
To measure extracellular matrix Ca deposits for bone nodule formation, cellular matrix was stained using Alizarin red S dye which combines with Ca on matrix. At the differentiation days of 1, 5 and 10, cells were washed with PBS twice, and then fixed with 4.0% formaldehyde. The cells were stained with 40 mM of Alizarin red S solution at pH 4.4 for 40 min at room temperature and rinsed with deionized water twice. The images of stained cells were captured using a phase contrast microscope with digital camera (IM50, Leica, Germany).
Data were analyzed using software SPSS-17.0. Values for cell proliferation by MTT assay for absorbance are presented as the mean ± SEM. The data analysis was performed using one way ANOVA and Tukey's HSD test as post hoc test was applied if significance was determined. Differences were considered as being significant at P < 0.05.
MC3T3-E1 cell proliferation was increased as time went by in a zinc concentration-dependent manner (Fig. 1). Cell proliferation was stimulated even at low zinc treatment (1-3 M) at day 1, compared to OSM. As time went by, this zinc-stimulated cell proliferation was more prominent at high range of zinc concentration at 15 and 25 uM Zn on day 5 and 10, while low zinc level showed the adverse effect of cell proliferation during this culture period.
Results show that the stimulatory effect for cell proliferation was shown by zinc treatment. MC3T3-E1 cells cultured in 0 µM Zn had decrease in cell proliferation as compared to OSM during 1 day. And MC3T3-E1 cells cultured in 0 and 1 µM Zn also had decrease in cell proliferation as compared to OSM during osteogenic period (5 and 10 days). This suggests that 0 and 1 µM Zn medium with the 5 µM TPEN treatment causes an adverse effect in the cell proliferation which may not be appropriate for prolonged cell culture. During 5 and 10 days of zinc treatment, the significant increase of cell proliferation was shown with a zinc-concentration dependent manner.
To evaluate the effect of zinc on the differentiation of osteoblastic MC3T3-E1 cells, we examined the activity of ALP, a marker of osteoblastic differentiation, on day 1, 5, and 10 of osteoblast proliferation and differentiation period both in cells (Fig. 2A) and media (Fig. 2B). Cellular (synthesized) and medium (secreted) ALP activity was stimulated by zinc in a concentration-dependent manner at day 1, 5, and 10. Cellular ALP activity was increased in a zinc concentration-dependent manner, especially day 1, at the range of 15 and 25 µM Zn treatments, compared to normal osteogenic differentiation media (OSM). Medium ALP activity was also shown as the same pattern with cellular ALP activity. Medium ALP activity by zinc treatment was shown more prominent zinc concentration-dependent manner up to 10 days, which can be interpreted that it might be due that ALP is the secretory enzyme out of the cell to the extracellular matrix. Medium ALP was increased by zinc treatment during 1, 5 and 10 days particularly at the range of 15 and 25 µM Zn, compared to OSM. These data showed that zinc treatment stimulated cellular and medium ALP activity.
Collagen synthesis is also a major marker of osteoblastic differentiation therefore we assessed the effect of zinc on collagen concentration in osteoblastic MC3T3-E1 cells both in cells (Fig. 3A) and media (Fig. 3B) during 10 days. Cellular collagen concentration was increased at higher zinc concentrations as time went by, especially at day 10 at the zinc concentration of 3-25 µM Zn treatment. Since collagen is cell membrane-attached and secretory protein, medium collagen concentration was also measured. Medium collagen concentration was increased with zinc concentration-dependent manner at day 1 and the pattern in which zinc treatment increased collagen concentration up to 10 days.
In addition to osteoblast proliferation and differentiation, such as collagen synthesis and ALP activity, extracellular matrix mineralization is also one of the major part of bone formation, and we assessed whether zinc treatment to MC3T3-E1 cells stimulated matrix mineralization which increases the anabolic activity in bone metabolism. Extracellular matrix Ca deposits for mineralised nodule formation were stained with Alizarin red S dye which combines with Ca ions and the calcified nodules appeared bright red colour by Alizarin red S staining (Fig. 5). Alizarin red S staining showed the sign that zinc treatment has the tendency to increase extracellular matrix Ca accumulation. However, in the present study, due to the shortage of zinc treatment duration for inducing calcification (since normally Ca deposits are very active around 10-15 days), Ca deposits in matrix was not prominent yet, up to 10 days. It was observed that zinc increased matrix Ca deposits with zinc concentration- and time-dependent manner during 5-10 days on MC3T3-E1 cells.
Zinc is a trace element, essential for the function of normal mammalian cell processes such as DNA [20,21] and protein synthesis [12,22,23]. In the present study, we investigated the possible effect of zinc on osteoblast proliferation and differentiation which was measured for the indicators of osteoblast differentiation, collagen concentration and alkaline phosphatase (ALP) activity, in MC3T3-E1 cells. Pre-osteoblasts, like the MC3T3-E1 cells, differentiate into osteoblasts, which is accompanied by a coordinated expression of a series of extracellular matrix (ECM) proteins, collagenous and noncollagenous proteins such as collagen type I and ALP, respectively [3]. At the proliferation phase of pre-osteoblastic to osteoblastic cells, the cells undergo DNA synthesis and cell division resulting in a rapid increase in cell number, which finally increases the expression of the osteogenic phenotype as well. In the present study, zinc increases cellular proliferation in osteoblatic MC3T3- E1 cells as time went by during 1-10 days, which is considered as stimulating osteoblast differentiation by bone marker protein synthesis by osteoblasts.
The presence of mature osteoblasts during the proliferation phase is indicated by the production of ALP, processing of procollagen to collagen, and the deposition of extracellular matrix containing additional proteins which would later be mineralized [24,25]. Thus, ALP activity and collagen synthesis are the most widely recognised biochemical markers for osteoblastic activity. These two proteins are typical proteins for osteoblast-phenotype and osteoblast differentiation [24,26]. Therefore, in general, ALP synthesis and ALP activity, and collagen synthesis are increased as osteoblasts are differentiating especially from the early stage of osteogenesis. In addition, ALP also contains zinc as part of its homodimeric metalloenzymes which contain zinc as the component [19,27].
ALP protein is known to be secreted from the cells after it is being synthesized and its expression heralds the stage at which differentiation is believed to be nonreversible in most systems studied. This agrees with the result of this study, since an increased ALP activity pattern by zinc treatment was observed from the proliferation (1 and 5 days) to matrix maturation and early mineralization phase (10 days). This pattern was more prominent in medium ALP activity (Fig. 2B), which is matched with the normal pattern of secretory protein like ALP. In this study, zinc increased ALP activity at cellular level at day 1 and 10 (Fig. 2A), which is considered as that zinc stimulates ALP synthesis, therefore can stimulate osteoblast differentiation. Also this increased ALP protein in cells is secreted outside the cells, which is confirmed by the same pattern of zinc-stimulated ALP activity in media (Fig. 2B). Therefore, these increased ALP protein synthesis and enzyme activity in cells and medium can be interpreted as the indication of osteoblast differentiation and bone matrix maturation.
Collagen synthesis is also well known as a sign of osteoblast differentiation. It has been reported by Bremner and Beattie [19] and Wallwork and Sandstead [28] that zinc deficiency affects both the biosynthesis and degradation of all type of collagen. In the present study, cellular collagen concentration was increased by zinc treatment as time went by. Cellular collagen level was not immediately increased by zinc treatment on day 1. But it was increased as time went by, therefore collagen level in cells were the highest at the highest zinc treatment level (25 mM) at day 10, with the higher collagen concentration (Fig. 3A). This pattern of the increased collagen concentration at 10 days would be considered that collagen proteins as secretory protein were accumulated in cells and cell layers as time went by. Also, collagen is cell membrane-attached and secretory protein, medium collagen synthesis was also measured [19,28]. While cellular collagen level was accumulated as zinc- and time-dependent manner, medium collagen concentration showed the more prominent zinc stimulator effect for collagen synthesis at day 1 with the zinc-dependent manner. In addition, collagen synthesis measured by staining collagen protein in cell layers showed the same pattern of cellular collagen level by zinc treatment (Fig. 4). These increased collagen synthesis and accumulation by zinc treatment in osteoblasts can be considered as the sign of stimulating the bone matrix and osteoblast maturation. Mineralization of extracellular matrix as Ca deposits for mineralised nodule formation was also assessed by staining by Alizarin red S dye which combines with Ca ions (Fig. 5). Even there is a sign of zinc stimulatory effect for ECM Ca deposits, however, Ca deposits were not prominently observed probably due to the immature ECM calcification yet at day 10 in this study.
Taken all together, it is suggested that zinc can increase osteogenic effect by increasing cell proliferation and stimulating ALP and collagen synthesis during the proliferation and differentiation phase in osteoblastic MC3T3-E1 cells. Therefore, zinc can increase bone formation through increasing osteoblast cell growth and cellular ALP and collagen synthesis in MC3T3-E1 cells. The future work needs to be focused on how and where-by these ALP and collagen synthesis modulated by zinc to clarify the osteogenic role of zinc.
Figures and Tables
 | Fig. 1
Proliferation of osteoblastic MC3T3-E1 cells by zinc treatment for 1, 5 and 10 days. Cell proliferation of MC3T3-E1 cells by zinc treatment were measured by MTT assay. The values are presented as %OSM as normal osteogenic medium. Different superscripts mean significant difference by zinc treatments at P < 0.05 by Tukey, ANOVA. Data presented as mean ± SEM (n = 10). |
 | Fig. 2
ALP activity in osteoblastic MC3T3-E1 cells by zinc. Alkaline phosphatase (ALP) activity in MC3T3-E1 cells (synthesized ALP, A) and the media (secreted ALP, B) treated with zinc. Different superscripts mean significant different among zinc treatments at P < 0.05 by Tukey, one-way ANOVA. Data presented as mean ± SEM (n = 3). ALP one unit of ρ-nitrophenol as products being converted from p-nitrophenyl phosphate as substrate for 30 minute reaction time. (cellular ALP 1 unit = a nmole ρ-nitrophenol/mg of protein/30 min, medium ALP 1 unit = 1 nmole p-nitrophenol/ml/30 min) |
 | Fig. 3
Collagen synthesis in osteoblastic MC3T3-E1 cells by zinc. Cellular and medium collagen concentration was measured as described in Methods. Different superscripts mean significant difference among zinc treatments at P < 0.05 by Tukey, one way ANOVA. Data presented as mean ± SEM (n = 3). |
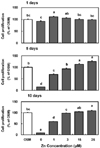
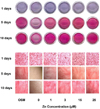 | Fig. 4
Collagen staining by zinc treatment in MC3T3-E1 cells for 1, 5 and 10 days. Cell matrix collagen was stained using Van Gieson's staining method as described on Method. Magnification rate, ×100 (1days) or ×40 (5 and 10 days). Representative image of 8 replicates in 24-well plate. |
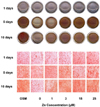 | Fig. 5
Alizarin Red S staining for Ca deposits (bone nodule) by zinc treatment in MC3T3-E1 cells for 1, 5 and 10 days. Cells were cultured at 24-well plates for 1, 5, and 10 days with the 0-25 mM zinc treatment. Extracellular matrix Ca deposits for matrix mineralization was measured using Alizarin red S dye which binds with Ca. Magnification rate, ×100. Representative image of 8 replicates in 24-well plate. |
References
1. Harada S, Rodan GA. Control of osteoblast function and regulation of bone mass. Nature. 2003. 423:349–355.

2. Noda M. Stein GS, Lian JB, editors. Cellular and molecular biology of bone. Molecular mechanisms mediating developmental and hormone-regulated expression of genes in osteoblasts: An integrated relationship of cell growth and differentiation. 1993. San Diego: Academic Press;48–97.
3. Koeneman KS, Yeung F, Chung LW. Osteomimetic properties of prostate cancer cells: a hypothesis supporting the predilection of prostate cancer metastasis and growth in the bone environment. Prostate. 1999. 39:246–261.

4. Choi JY, Lee BH, Song KB, Park RW, Kim IS, Sohn KY, Jo JS, Ryoo HM. Expression patterns of bone-related proteins during osteoblastic differentiation in MC3T3-E1 cells. J Cell Biochem. 1996. 61:609–618.

5. Chien KR, Karsenty G. Longevity and lineages: toward the integrative biology of degenerative diseases in heart, muscle, and bone. Cell. 2005. 120:533–544.

6. Tapiero H, Tew KD. Trace elements in human physiology and pathology: zinc and metallothioneins. Biomed Pharmacother. 2003. 57:399–411.

7. Hsieh HS, Navia JM. Zinc deficiency and bone formation in guinea pig alveolar implants. J Nutr. 1980. 110:1581–1588.

8. Oner G, Bhaumick B, Bala RM. Effect of zinc deficiency on serum somatomedin levels and skeletal growth in young rats. Endocrinology. 1984. 114:1860–1863.

9. Yamaguchi M, Yamaguchi R. Action of zinc on bone metabolism in rats: increases in alkaline phosphatase activity and DNA content. Biochem Pharmacol. 1986. 35:773–777.
10. Hall SL, Dimai HP, Farley JR. Effects of zinc on human skeletal alkaline phosphatase activity in vitro. Calcif Tissue Int. 1999. 64:163–172.

11. Yamaguchi M, Oishi H, Suketa Y. Stimulatory effect of zinc on bone formation in tissue culture. Biochem Pharmacol. 1987. 36:4007–4012.

12. Yamaguchi M. beta-Alanyl-L-histidinato zinc and bone resorption. Gen Pharmacol. 1995. 26:1179–1183.
13. Yamaguchi M. Role of zinc in bone formation and bone resorption. Journal of Trace Elements in Experimental Medicine. 1998. 11:119–135.

14. Hashizume M, Yamaguchi M. Stimulatory effect of beta-alanyl-L-histidinato zinc on cell proliferation is dependent on protein synthesis in osteoblastic MC3T3-E1 cells. Mol Cell Biochem. 1993. 122:59–64.

15. Hashizume M, Yamaguchi M. Effect of beta-alanyl-L-histidinato zinc on differentiation of osteoblastic MC3T3-E1 cells: increases in alkaline phosphatase activity and protein concentration. Mol Cell Biochem. 1994. 131:19–24.

16. Franceschi RT, Iyer BS. Relationship between collagen synthesis and expression of the osteoblast phenotype in MC3T3-E1 cells. J Bone Miner Res. 1992. 7:235–246.

17. Stein GS, Lian JB, Owen TA. Relationship of cell growth to the regulation of tissue-specific gene expression during osteoblast differentiation. FASEB J. 1990. 4:3111–3123.

18. Cao J, Bobo JA, Liuzzi JP, Cousins RJ. Effects of intracellular zinc depletion on metallothionein and ZIP2 transporter expression and apoptosis. J Leukoc Biol. 2001. 70:559–566.
19. Bremner I, Beattie JH. Copper and zinc metabolism in health and disease: speciation and interactions. Proc Nutr Soc. 1995. 54:489–499.

20. Beyersmann D, Haase H. Functions of zinc in signaling, proliferation and differentiation of mammalian cells. Biometals. 2001. 14:331–341.

21. Yamaguchi M, Matsui T. Stimulatory effect of zinc-chelating dipeptide on deoxyribonucleic acid synthesis in osteoblastic MC3T3-E1 cells. Peptides. 1996. 17:1207–1211.

22. Ma ZJ, Yamaguchi M. Role of endogenous zinc in the enhancement of bone protein synthesis associated with bone growth of newborn rats. J Bone Miner Metab. 2001. 19:38–44.

23. Ma ZJ, Misawa H, Yamaguchi M. Stimulatory effect of zinc on insulin-like growth factor-I and transforming growth factor-beta 1 production with bone growth of newborn rats. Int J Mol Med. 2001. 8:623–628.
24. Magnusson P, Larsson L, Magnusson M, Davie MW, Sharp CA. Isoforms of bone alkaline phosphatase: characterization and origin in human trabecular and cortical bone. J Bone Miner Res. 1999. 14:1926–1933.

25. Peterson WJ. A method to assess the proliferative activity of small numbers of murine peripheral blood mononuclear cells. J Immunol Methods. 1987. 96:171–177.

26. Lian JB, Stein GS. Transcriptional control of vitamin D-regulated proteins. J Cell Biochem. 1992. 49:37–45.

27. Fernley HN, Walker PG. Inhibition of alkaline phosphatase by L-phenylalanine. Biochem J. 1970. 116:543–544.

28. Simmons DJ. Wallwork JC, Sandstead HH, editors. Nutrition and Bone Development. Zinc. 1990. New York: Oxford University Press;316–342.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download