Abstract
Purpose
Increasing evidences have indicated the critical role of HO-1 in cytoprotection and more diverse biological functions. HO-1 has been reported to stimulate tumor cell growth and proliferation. And several tumors, including renal cell carcinoma, prostate tumor, hepatoma, and sarcoma, express a high level of HO-1. Indeed, inhibition of HO-1 by using specific HO inhibitors demonstrated in vivo antitumor activity. However, the precise mechanism of HO-1 induction and signals in lung cancer is not clearly known yet. We aimed to analyze the role of HO-1, characterize the mechanism of HO-1 induction, the role of Nrf2 in the induction, and investigated whether inhibition of HO-1 may induce apoptosis in lung cancer cells.
Materials and Methods:
Western blot and immunostaining analyses were performed to ascertain whether HO-1, Nrf2, and NFkB were expressed or not in various lung cancer cell lines. Apoptosis by HO-1 inhibition through siRNA transfectiori was measured by flow cytometric analysis and Western blot. And the expression of HO-1 by siRNA of Nrf2 and NFkB was examined by ARE-driven luciferase activity and Western blot.
Results:
We demonstrated that HO-1 was expressed highly in A549 cells than other lung cancer cells. And A549 cells were transfected by HO-1 small interfering RNA (siRNA) induced apoptosis. Nrf2 siRNA, next, resulted in a decrease of HO-1 expression. However, NFkB siRNA had no influence on the expression of HO-1.
Conclusion:
Increasing HO-1 expression in A549 cells may be resulted from the transcriptional activation of Nrf2, and inhibition of Nrf2-HO-1 pathway induces apoptosis. Therefore it provides new important insights into the possible molecular mechanism of the antitumor therapy. (J Lung Cancer 2006;5(1):39-46)
Go to : 
REFERENCES
1.Tenhunen R: Marver HS: Schmid R. The enzymatic catabolism of hemoglobin: stimulation of microsomal heme oxygenase by hemin. J Lab Clin Med. 1970. 75:410–421.
2.Baranano DE: Rao M: Ferris CD., Snyder SH. Biliverdin reductase: a major physiologic cytoprotectant. Proc Natl Acad Sci USA. 2002. 99:16093–16098.
3.Maines MD. The heme oxygenase system: a regulator of second messenger gases. Annu Rev Pharmacol Toxicol. 1997. 37:517–554.
4.Tenhunen R., Marver HS., Schmid R. The enzymatic catabolism of hemoglobin: stimulation of microsomal heme oxygenase by hemin. J Lab Clin Med. 1970. 75:410–421.
5.Motterlini R., Foresti R., Bassi R., Calabrese V: Clark JE., Green CJ. Endothelial heme oxygenase-1 induction by hypoxia. Modulation by inducible nitric-oxide synthase and S-nitrosothiols. J Biol Chem. 2000. 275:13613–13620.
6.Stuhlmeier KM. Activation and regulation of Hsp32 and Hsp 70. Eur J Biochem. 2000. 267:1161–1167.
7.Doi K., Akaike T., Fujii S, et al. Induction of haem oxygenase-1 nitric oxide and ischaemia in experimental solid tumours and implications for tumour growth. Br J Cancer. 1999. 80:1945–1954.
8.Elbirt KK: Whitmarsh AJ., Davis RJ: Bonkovsky HL. Mechanism of sodium arsenite-mediated induction of heme oxygenase-1 in hepatoma cells. Role of mitogen-activated protein kinases. J Biol Chem. 1998. 273:8922–8931.
9.Eyssen-Hemandez R: Ladoux A., Frelin C. Differential regulation of cardiac heme oxygenase-1 and vascular endothelial growth factor mRNA expressions by hemin, heavy metals, heat shock and anoxia. FEBS Lett. 1996. 382:229–233.
10.Keyse SM., Tyrrell RM. Heme oxygenase is the major 32-kDa stress protein induced in human skin fibroblasts by UVA radiation, hydrogen peroxide, and sodium arsenite. Proc Natl Acad Sci USA. 1989. 86:99–103.

11.Lautier D: Luscher P: Tyrrell RM. Endogenous glutathione levels modulate both constitutive and UVA radiation/hydrogen peroxide inducible expression of the human heme oxygenase gene. Carcinogenesis. 1992. 13:227–232.

12.Hara E., Takahashi K., Takeda K, et al. Induction of heme oxygenase-1 as a response in sensing the signals evoked by distinct nitric oxide donors. Biochem Pharmacol. 1999. 58:227–236.

13.Rizzardini M., Terao M., Falciani F., Cantoni L. Cytokine induction of haem oxygenase mRNA in mouse liver. Interleukin 1 transcriptionally activates the haem oxygenase gene. Biochem J. 1993. 290:343–347.

14.Agarwal A., Shiraishi F., Visner GA., Nick HS. Linoleyl hydroperoxide transcriptionally upregulates heme oxygenase-1 gene expression in human renal epithelial and aortic endothelial cells. J Am Soc Nephrol. 1998. 9:1990–1997.

15.Fogg S., Agarwal A., Nick HS., Visner GA. Iron regulates hyperoxia-dependent human heme oxygenase 1 gene expression in pulmonary endothelial cells. Am J Respir Cell Mol Biol. 1999. 20:797–804. ,.

16.Alam J: Stewart D., Touchard C: Boinapally S: Choi AM: Cook JL. Nrf2, a Cap'n'Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J Bio Chem. 1999. 274:26071–26078.
17.Wagner M., Cadetg P., Ruf R., Mazzucchelli L., Ferrari P., Redaelli CA. Heme oxygenase-1 attenuates ischemia/reperfusion-induced apoptosis and improves survival in rat renal allografts. Kidney Int. 2003. 63:1564–1573.

18.Choi BM., Pae HO., Chung HT. Nitric oxide priming protects nitric oxide-mediated apoptosis via heme oxygenase-1 induction. Free Radic Biol Med. 2003. 34:1136–1145.

19.Amon M., Menger MD., Vollmar B. Heme oxygenase and nitric oxide synthase mediate cooling-associated protection against TNF-alpha-induced microcirculatory dysfunction and apoptotic cell death. F ASEB J. 2003. 17:175–185.
20.Ke B., Shen XD., Zhai Y, et al. Heme oxygenase 1 mediates the immunomodulatory and antiapoptotic effects of interleukin 13 gene therapy in vivo and in vitro. Hum Gene Ther. 2002. 13:1845–1857.
21.Goodman Al., Choudhury M., da Silva JL., Schwartzman ML., Abraham NG. Overexpression of the heme oxygenase gene in renal cell carcinoma, Proc Soc Exp Biol Med. 1997. 214:54–61.
22.Maines MD., Abrahamsson PA. Expression of heme oxygenase-1 (HSP32) in human prostate: normal, hyperplastic, and tumor tissue distribution. Urology. 1996. 47:727–733.

23.Sahoo SK., Sawa T., Fang J, et al. Pegylated zinc protopo卜phyrin: a water-soluble heme oxygenase inhibitor with tumortargeting capacity. Bioconjug Chem. 2002. 13:1031–1038.
24.Crissman HA., Steinkamp JA. Cell cycle-related changes in chromatin structure detected by flow cytometry using multiple DNA fluorochromes. Eur J Histochem. 1993. 37:129–138.
25.Elbashir SM: Harborth J: Lendeckel W., Yalcin A: Weber K., Tuschl T. Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 2001. 411:494–498.

26.Alam J: Wicks C: Stewart D, et al. Mechanism of heme oxygenase-1 gene activation by cadmium in MCF-7 mammary epithelial cells. Role of p38 kinase and Nrf2 transcription factor. J Biol Chem. 2000. 275:27694–27702.
27.He CH., Gong P., Hu B, et al. Identification of activating transcription factor 4 (ATF4) as an Nif2-interacting protein. Implication for heme oxygenase-1 gene regulation. J Biol Chem. 2001. 276:20858–20865.
28.Banning A: Brigelius-Flohe R. NF-kappaB, Nrf2, and HO-1 interplay in redox reguhted VC AM 니expression. Antioxid Redox Signal. 2005. 7:889–899.
29.Deramaudt BM., Braunstein S: Remy P: Abraham NG. Gene transfer of human heme oxygenase into coronary endothelial cells potentially promotes angiogenesis. J Cell Biochem. 1998. 68:121–127.

30.Hanselmann C: Mauch C., Werner S. Heme oxygenase-1: a novel player in cutaneous wound repair and psoriasis? Biochem J. 2001. 353:459–466.
31.Fang J: Akaike T: Maeda H. Antiapoptotic role of heme oxygenase (HO) and the potential of HO as a target in anticancer treatment. Apoptosis. 2004. 9:27–35.

32.Fang J., Sawa T: Akaike T, et al. In vivo antitumor activity of pegylated zinc protoporphyrin: targeted inhibition of heme oxygenase in solid tumor. Cancer Res. 2003. 63:3567–3574.
33.Sunamura M., Duda DG., Ghattas MH, et al. Heme oxygenase-1 accelerates tumor angiogenesis of human pancreatic cancer. Angiogenesis. 2003. 6:15–24.
34.Tanaka S: Akaike T: Fang J, et al. Antiapoptotic effect of haem oxygenase-1 induced by nitric oxide in experimental solid tumor. Br J Cancer. 2003. 88:902–909.
35.Alam J: Stewart D., Touchard C., Boinapally S: Choi AM., Cook JL. Nrf2, a Cap'n'Cdlar transcription factor, regulates induction of the heme oxygenase-1 gene. J Biol Chem. 1999. 274:26071–26078.
36.Zipper LM: Mulcahy RT. The Keapl BTB/POZ dimerization function is required to sequester Nrf2 in cytoplasm. J Biol Chem. 2002. 277:36544–36552.
37.Barkett M., Gilmore TD. Control of apoptosis by Rel/NF-kappa B transcription factors. Oncogene. 1999. 18:6910–6924.
Go to : 
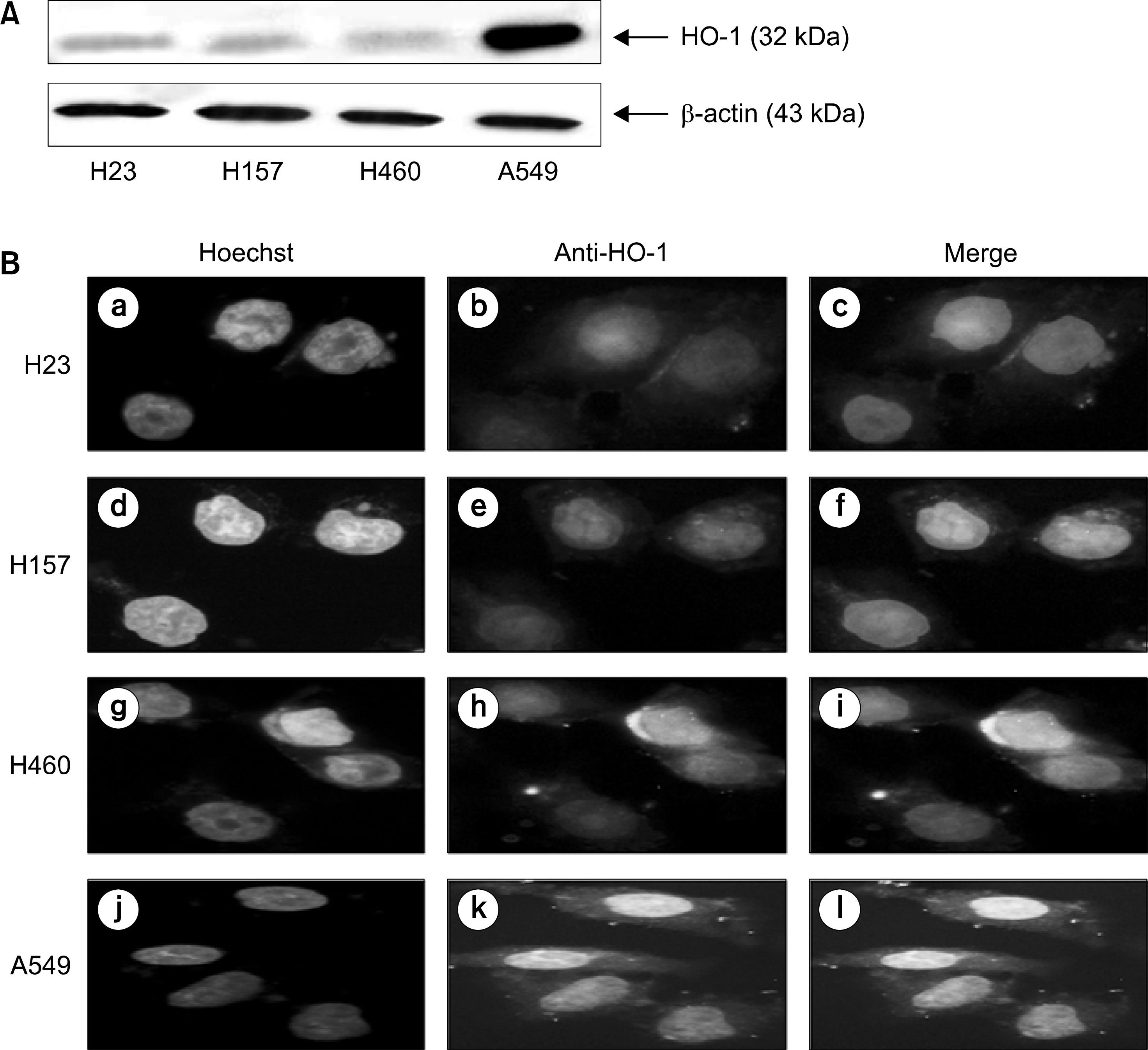 | Fig. 1.Expression of HO-1 in endogenous lung cancer cells. (A) Expression levels of HO-1 protein were measured by Western blotting using ant卜HO-1 antibody. Lysates of untreated lung cancer cells were subjected to 12.5% SDS-PAGE to measure the expression of HO-1 protein. (B) Untreated lung cancer cells were reacted with Hoechst and ariti-HO-1 antibody. Then, cells were visualized under fluorescent microscope. |
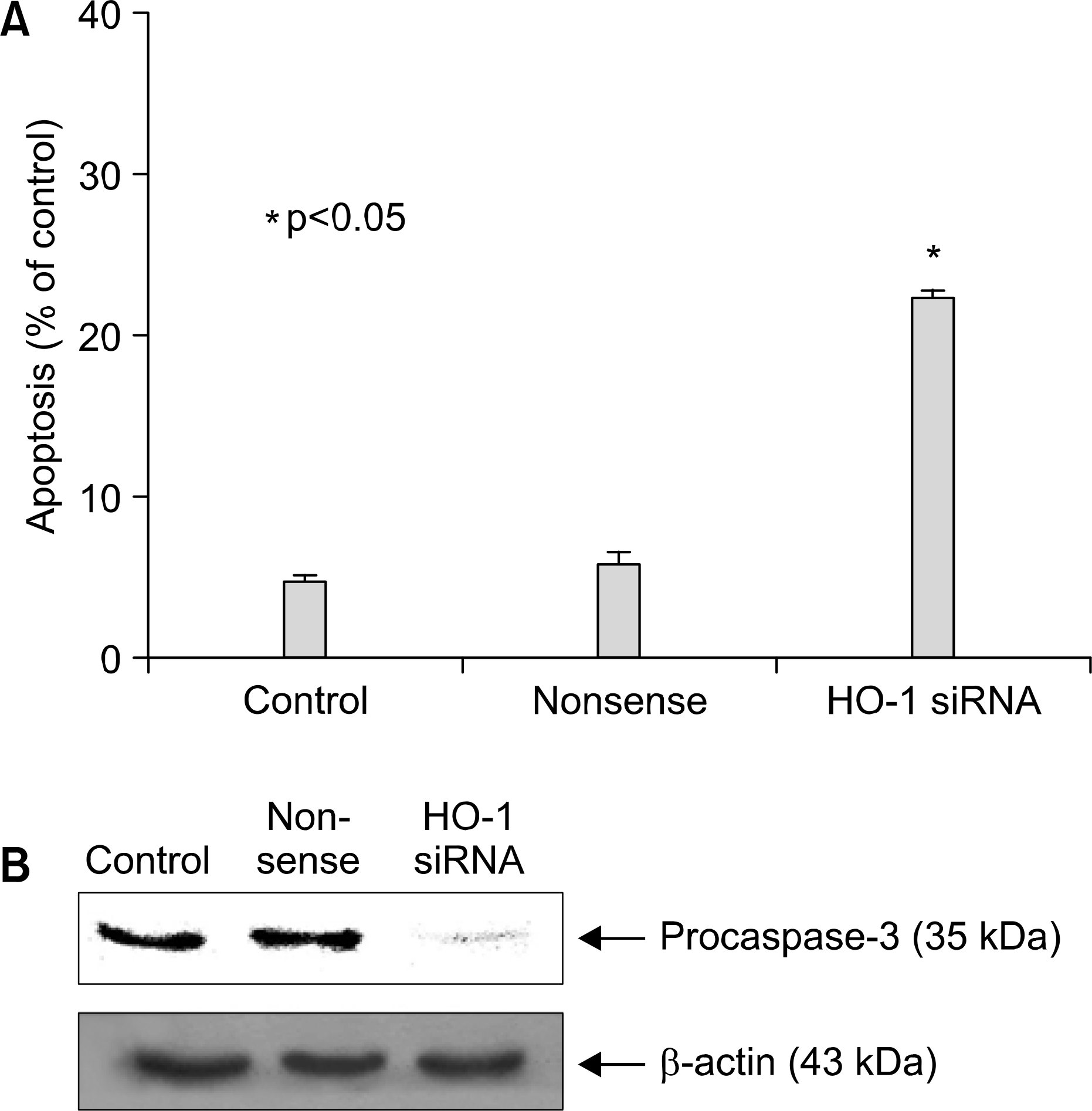 | Fig. 2.Apoptosis in A549 celts after HO-1 siRNA transfection. Nonsense indicates the result for cells treated with Silencer Negative Control siRNA. Cells were transfected with 0.5 ᆻg siRNA of HO-1 and control plasmid. (A) Cellular DNA was stained with PI and celt cycle was analyzed by flow cytometry. The data represent the mean 士 S.D. of triplicate. ∗p<0.05. (B) Cells were transfected with 0.5 ᆻg siRNA of HO-1 and control plasmid. After 48 hrs, lysate was subjected to 12.5% SDS-PAGE to measure the expression of pro- caspase-3. |
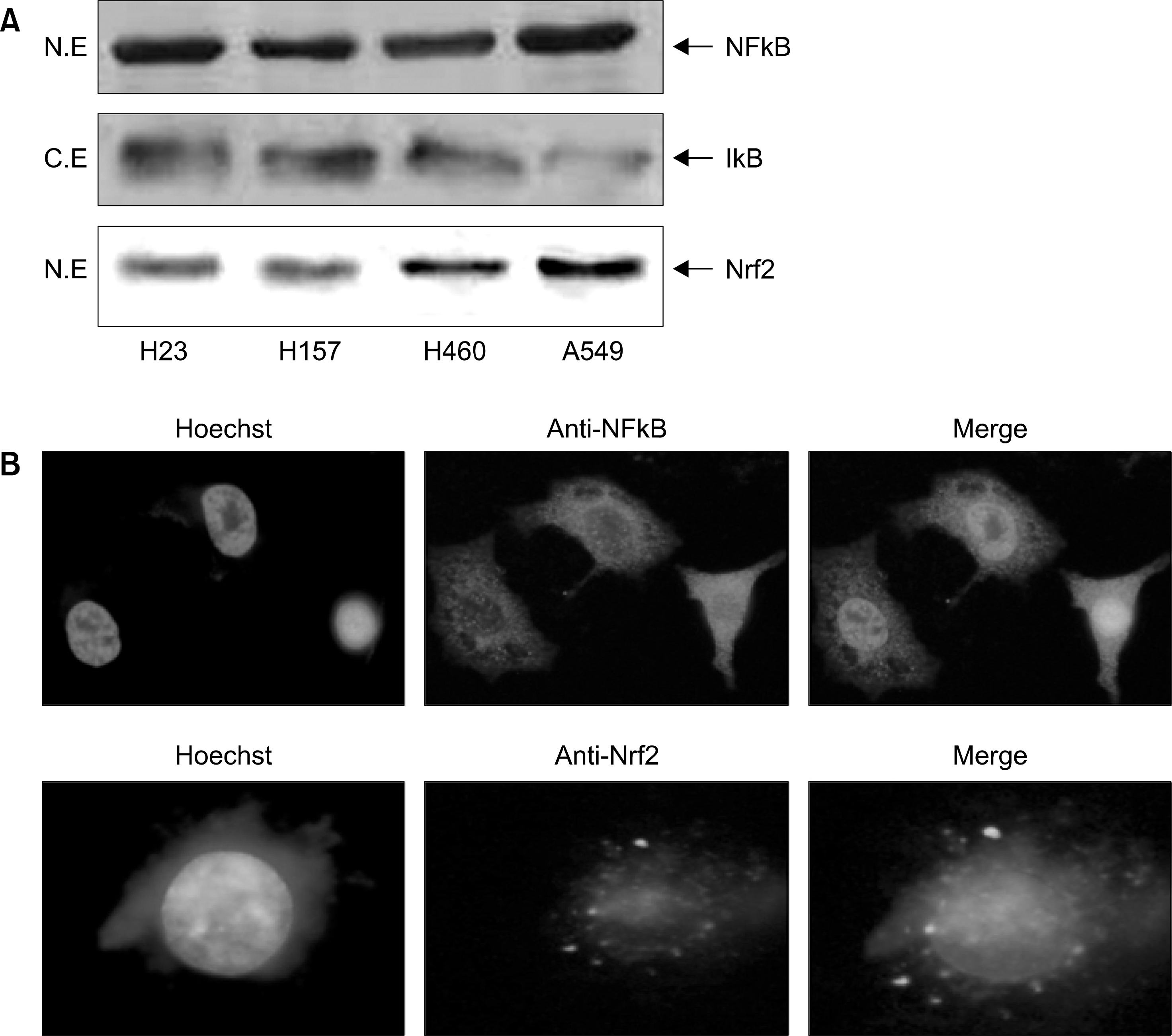 | Fig. 3.Expression of Nrf2 and NFkB in endogenous lung cancer cells. (A) Expression levels in both cytosolic and nuclear fractions of Nrf2 and NFkB protein were measured by Western blotting. Lysate was fractionated into cytosolic and nuclear portions and proteins were separated on 15% SDS-PAGE to immunoblot for Nrf2 and NFkB in nucleus and IkB in cytoplasm. (B) Untreated A549 cells were reacted with Hoechst and ant卜HO-1 antl· body. Then, cells were visualized under fluorescent microscope. |
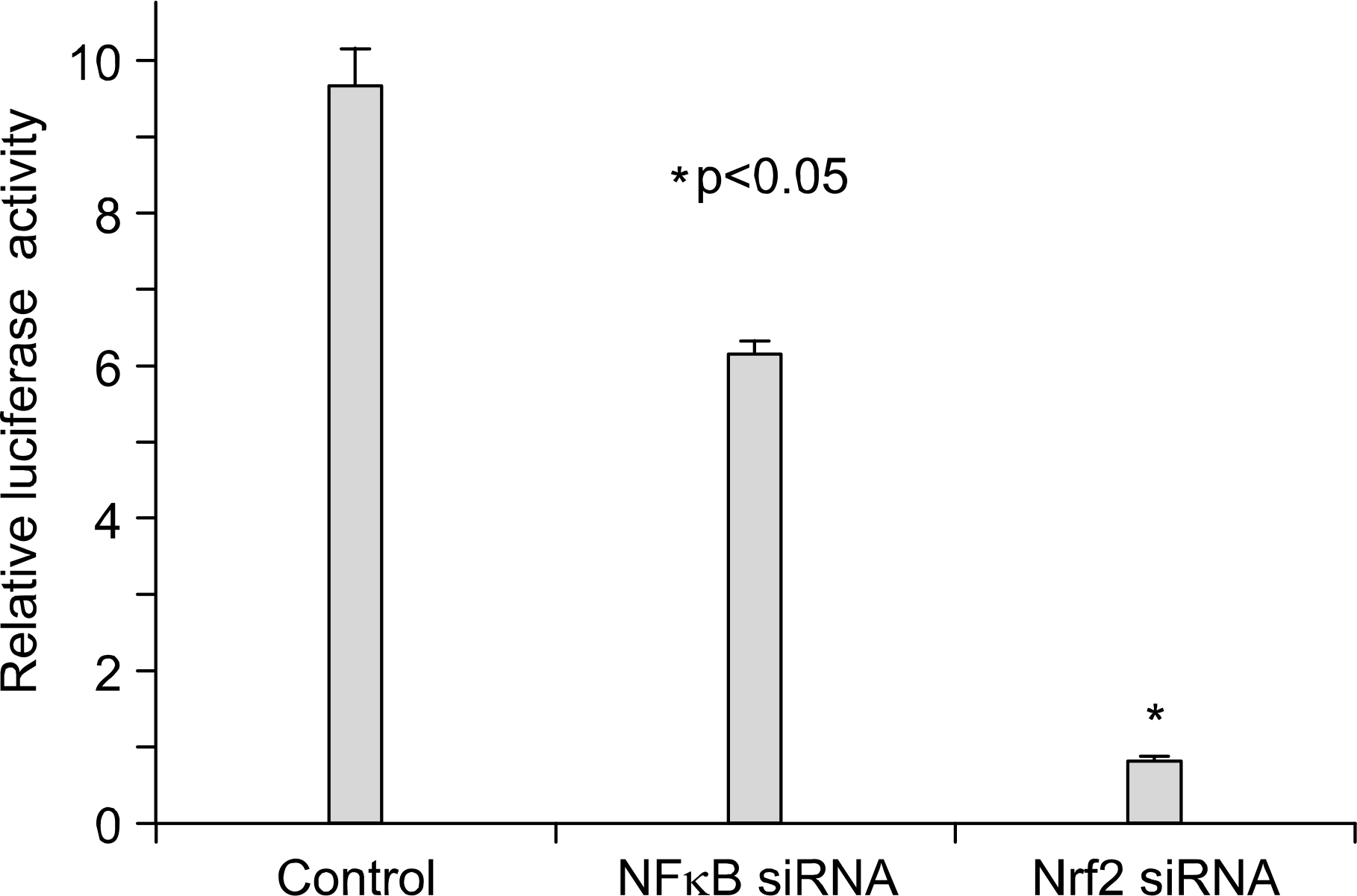 | Fig. 4.Expression of Nrf2 and NFkB siRNA transfection on HO-1 promotor activity. In A549 cells, Nrf2 and NFkB expression vectors were respectively cotransfected with ARE—luciferase reporter gene for 24 hrs, and then luciferase activity was measured at 18 h later. The data represent the mean 土 S.D. of triplicate. ∗p<0.05. |
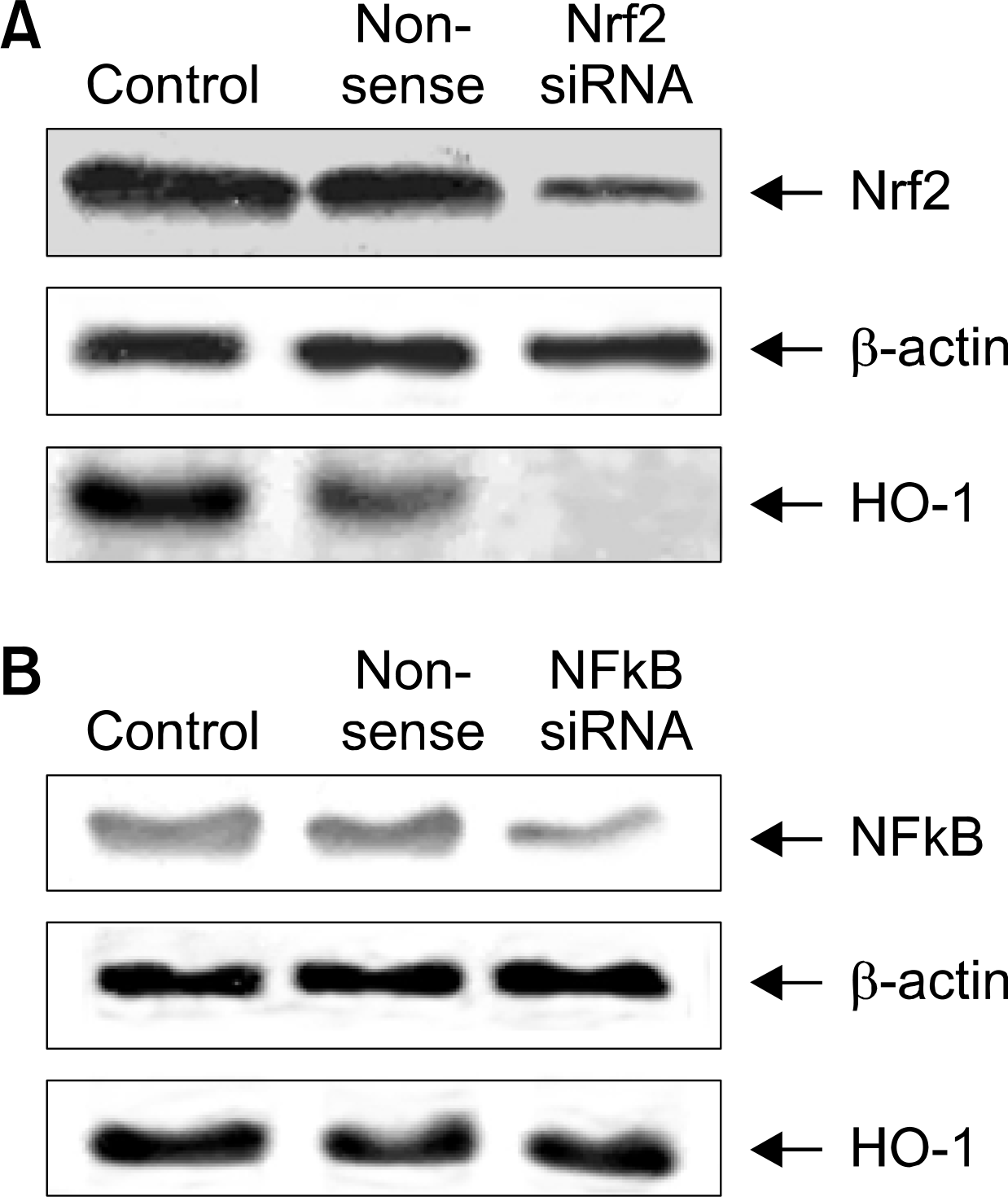 | Fig. 5.Expression of Nrf2 and NFkB siRNA transfectiori on HO-1 protein expression. Nonsense indicates the result for cells treated with Silencer Negative Control siRNA. (A) Cells were transfected with 0,5 μ g siRNA of Nrf2 and control plasmid. Suppression of Nrf2 levels after siRNA treatment after 48 hrs was respectively confirmed by Western blot. Simultaneously lysates were subjected to 12.5% SOS-PAGE to measure the expression of HO-1 protein, (B) Cells were transfected with 0.5μ g siRNA of NFkB and control plasmid Suppression of NFkB levels after siRNA treatment after 48 hrs was respectively confirmed by Western blot Simultaneously lysates were subjected to 12.5% SDS-PAGE to measure the expression of HO-1 protein. |




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download