Abstract
Purpose
To estimate the prevalence of periodontitis in Korean adults and to examine the associations between periodontitis and known risk factors.
Methods
Using Korean National Oral Health Survey 2006 data, a total of 4,263 people who had taken oral examination, interviewed by questionnaire, and aged 18 or older were sampled. The prevalence of periodontitis measured by Community Periodontal Index (CPI) was calculated and the differences in prevalence according to known risk factors (age, sex, monthly income, education, residential area, tooth-brushing frequency, regular dental visit, smoking, and diabetes) were examined with chi-square test. Logistic regression analysis was performed to see the effects of each risk factor on the risk of having periodontitis. All statistical approaches were reflected national sampling design using Survey procedures in SAS 9.1.
Results
The overall prevalence of periodontitis in Korean adults was 10.3%. There existed statistically significant differences in crude prevalence for periodontitis according to the all risk factors. In logistic regression, older age groups (O.R.:2.94-3.71), people living in rural area (O.R.:1.87), and current smokers (O.R.:1.77) were significantly prone to have periodontitis. People who earned monthly income of more than 2 million Korean won (O.R.:0.64) and brushed their teeth two or more times per day (O.R.:0.60-0.62) had significantly lower risk of having periodontitis.
Periodontitis which has been viewed for years as a primary dental outcome from chronic bacterial infection is now seen as resulting from a complex interplay between bacterial infection and host response, often modified by behavioral factors1,2). Knowing that periodontitis is a preventable disease and is irreversible when it occurs, many developed countries have been concerned about the prevalence of periodontitis in their population and made policy efforts to reduce it.
Prevalence is defined as the number of affected persons present in the population at a specific time divided by the number of persons in the population at that time3). Despite the inconsistency in case definition and measurement method of periodontitis, the prevalence of severe periodontitis is known to be relatively high in the population (11-15% in adult populations) in both developed1,4) and developing countries5-7). However, accurate assessment of the prevalence of periodontitis is complicated by multiple factors8); different operational definitions of clinical periodontitis, characteristics of the study populations, measurement of clinical parameters (e.g. probing depth, clinical attachment loss, bleeding on probing, presence of dental calculus or plaque, etc.), and choice of partial vs. whole-mouth examination. Moreover, most prevalence data from foreign countries are either estimates obtained from relatively small observational (cross-sectional) studies or are derived from larger epidemiological studies that do not permit an in-depth diagnostic assessment of subjects8).
The national prevalence of periodontitis in Korean population has not been reported yet because of the above mentioned reasons. However, after Oral Health Act was enacted in 2000, Korea has conducted Korean National Oral Health Survey (KNOHS) which surveyed national representative sample of Korean population at 3 years interval9,10). The data from KNOHS include periodontal health status measured by Community Periodontal Index (CPI), so that calculation of prevalence of periodontitis would be possible. KNOHS also surveyed various risk factors known to be related with periodontitis such as smoking and diabetes. In this study, we estimated the prevalence of periodontitis in Korean adults and examined the strength of association between periodontitis and well known risk factors.
The data for this study came from Korean National Oral Health Survey (KNOHS) 2006. The KNOHS 2006 surveyed 15,777 persons 2 years or older, of whom 4,263 who were 18 years or older and had completed oral health examination were included in this study. Sampling design for KNOHS 2006 was a proportionally stratified by region (metropolis, urban, and rural) and clustered sampling. People who lived in urban area were oversampled than those in other two regions. Full description of the sample design of KNOHS 2006 was reported elsewhere10).
The periodontal health status was measured using CPI recommended by WHO as standard epidemiological examination method for periodontal disease with dental mirror and WHO periodontal probe11). People were classified into five groups by CPI; 0 - healthy periodontium, 1- gingival bleeding on probing, 2 - dental calculus presence, 3-shallow pocket depth (4-5 mm), 4-deep pocket depth (6 mm or above). A person coded with more than 2 in any of six sextants was defined as having periodontitis.
From the various variables surveyed in KNOHS 2006, nine variables were included in this study as periodontitis-related risk factors; demographics (age and sex), socio-economic factors (monthly income, education, and residential area), dental behavioral factors (tooth-brushing and regular dental clinic visit), and general health related factors (smoking and diabetes)
Data management procedures were carried out with Surveyfreq and Surveylogistic procedures in SAS 9.1 (SAS Institute Inc., Cary, USA) to take into account the national complex sampling design and to yield unbiased standard error estimates. To investigate the distribution of socio-demographic characteristics of the population and the CPI categories, and estimate the prevalence of periodontitis, weighted percents, 95% confidence limits, and P-values for determination of significant differences were calculated using Surveyfreq procedure. To examine the strength of the association between the prevalence of periodontitis and related risk factors such as demographic, socioeconomic, and behavioral determinants after controlling the effects of each risk factor, we built three logistic regression models. In model I, age and gender were included. In model II, resident area, household income, education were added to model I. Lastly, in model III, regular dental visit, smoking, tooth-brushing frequency, and diabetic status were added into model II. Adjusted odds ratios (OR) and 95% confidence intervals were calculated with Surveylogistic procedure.
The general characteristics of study population are
shown in Table 1. Weighted gender and age distribution is likely representative for Korean population in 2006 due to using national data. Only 14.3% of study subjects had visited dental clinic regularly and 11.0% brushed their teeth one or less times per day. Current smokers were 25.8% and 4.6% had diabetes. The proportion of residential area of whom lived in metropolis, urban, and rural areas were 47.6%, 37.2%, and 15.2%, respectively.
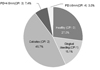
The weighted distribution of CPI index is shown in Fig. 1. About 10% of population had periodontitis (CPI index 3 or 4) and 27% were healthy. Gingival bleeding was found in 16.1% and 46.7% had calculus.
The overall prevalence of periodontitis in Korean adults in 2006 was 10.3% (95% C.I. 9.3%-11.4%) (Table 2). The prevalence was higher in men (13.3%) than women (7.6%) and showed increasing tendency as age increased. There existed statistically significant differences in periodontitis prevalence according to the all related risk factors included in this study. The person who had 9 years or less education, brushed their teeth one or less time per day, had diabetes, and lived rural area exhibited worse periodontal clinical conditions with prevalence of 18.0%, 20.1%, 19.0%, and 20.2% respectively.
The results from logistic regression analysis are shown in Table 3. In model I, which contains only gender and age, men were 1.98 times more likely to have periodontitis than women and odds ratios increased significantly as age group getting older. However, this increasing trend through age group was disappeared when other risk factors added to the model. In model III, which included all 9 risk factors, there existed no risk differences in having periodontitis between men and women (95% C.I. 0.86-1.86). Compared with young age group (18-39 years), all the other age groups had significantly higher odds of having periodontitis and the person living in rural area had 1.87 times more risk of p eriodontitis than the person living in metropolis area. More education, regular dental visit, and not having diabetes did not exhibited significant positive effect on periodontitis after controlling other risk factors. However, the person who brushed their teeth two or more times showed about 60% of risk to have periodontitis compared with one or less times tooth-brushing per day. Current smokers were 1.77 times more likely to have periodontitis than non-smokers after controlling other risk factors.
In this study, we estimated the prevalence of periodontitis using national data representing Korean adult population and examined the associations between periodontitis and known risk factors. As far as we know of, this would be the first report about the prevalence of periodontitis and associated risk factors in Korea calculated from nationally representative data with large sample size. Most of national surveys12-15) use stratified sampling technique to effectively sample the study population that could represent the original population. KNOHS 2006 also used a stratified cluster sampling that stratified by residential area such as metropolis, urban, and rural. Because the survey units were two times oversampled from urban area, to accurately measure the prevalence, one should take into account the weight of each survey unit to account for the sample design. For this reason, this study took into account all the weights used in KNOHS 2006 in prevalence calculation and risk estimation.
To define periodontitis operationally, the basic clinical measures, apart from gingival bleeding and radiographic assessment of bone loss, are clinical attachment loss (CAL) and probing depth (PD)1). Although CAL which could measure accumulated past status at a site of a tooth rather than current inflammatory activity, has been used as a diagnostic "gold standard" for periodontitis, the absence of consensus on how best to incorporate CAL and PD into a case definition of periodontitis continues in the field of clinical and epidemiological research16). A case definition for periodontitis needs to establish 1) what depth of CAL at any one site constitutes evidence of disease processes; 2) how many such sites need to be present in a mouth to establish disease presence17). The first problem also has to have measurement variation by dental examiners, who can be confused to detect CAL progression. Even though measurements of probing depth are repeatable to within 1 mm more than 90% of the time, the standard deviation of repeated CAL measurements of the same site by an experienced examiner with a manual probe is around 0.8 mm18). On the other hand, CPI used in this study probed pocket depth, so it can only reflect current periodontal status. The estimation of the prevalence of periodontitis can vary by different case definition.
Determining the prevalence of periodontitis is practically complicated by the various case definitions used. If periodontitis in the United States (US) population is defined as the identification of at least one site with CAL of ≥2 mm, around 80% of all adults are affected, and around 90% of those aged 55 to 641). Using pockets of ≥4 mm as a case definition, 30% of adults had met that criterion on at least three to four teeth. The prevalence of attachment loss ≥3 mm was 53.1% for the population of dentated U.S. adults 30 to 90 years of age and, fourteen percent of these persons had furcation involvement in one or more teeth. It was estimated that at least 35% of the dentated U.S. adults aged 30 to 90 have periodontitis with 21.8% having a mild form and 1 2.6% having a moderate or severe form19). As with prevalence, measures of periodontitis incidence will vary according to the case definition of the disease. CPI also has been used to measure periodontal health clinically20) and epidemiologically21). Japanese national survey21) where CPI also used, the prevalence of periodontitis among adults in 1999 was around 40%. However, the prevalence of periodontitis among Korean adults in 2006 was 10.3% which has not been expected compared to that of other countries. This finding could be explained as follows; 1) Korean population may have better periodontal health as Korea has developed economically. 2) The examiners participated in the KNOHS were prone to be conservative to define periodontal case and/or might miss deep pockets because it had been within one year since all examiners had graduated their dental schools.
Contemporary epidemiological studies including this study employ partial mouth examination system that could estimate biased values, especially when the prevalence of the disease is underestimated22). CPI is also based on partial-mouth recordings: pocket probing was performed at buccal sites in the maxillary and at lingual sites in the mandible. The highest value of CPI scores among the six section21).
It has been well-known that socio-demographic factors4,23,24) and other behavioral factors such as smoking21,25-27), diabetes22,28,29), and tooth brushing30,31) could affect the development and/or prevention of periodontitis. The findings in the present study partially support the associations between periodontitis and related risk factors as above. In Korean adults, age and smoking were positively associated with the prevalence of periodontitis after adjusted other confounders whereas income and tooth-brushing frequency were negatively related. The periodontitis was more prevalent in people who were men and living in rural area. Based on this information, the periodontal health of those susceptible groups should be considered regarding prevention of the disease and periodontal health promotion.
This study has a couple of important limitations. CPI with the measurement of pocket depth has an inherent weakness to detect periodontal destruction via; 1) national surveys is the use of partial-mouth recording, with only selected one or two selected teeth in 6 sextants examined, under the assumption that these measurements are representative of the full mouth4), 2) pocket depth could not detect the past periodontitis. These issues make the epithelial destruction of periodontium underestimated. The other limitation was that the study design was cross-sectional which cannot surely explain temporal precedence of risk factors over periodontal health condition. However, the results showed definite associations between age, residence, income, smoking, tooth brushing and periodontitis. In the future, the studies investigating measurement issue of defining periodontitis and related risk factors should be conducted to identify the independent pathways through which risk factors can impact periodontal health using longitudinal epidemiologic approaches.
In conclusion, one Korean adult person out of ten had periodontitis diagnosed by CPI in 2006. People who were older, living in rural region, in lower income status, smoking, less tooth brushing were more likely to have periodontitis.
Figures and Tables
Figure 1
Weighted distribution of CPI index in study population Korean National Oral Health Survey 2006
References
1. Burt B. Position paper: epidemiology of periodontal diseases. J Periodontol. 2005. 76:1406–1419.
3. Gordis L. Measuring the occurrence of disease: I. Morbidity. Epidemiology. 2009. 4th ed. Philadelphia: Saunders Elsevier;43.
4. Albandar JM, Brunelle JA, Kingman A. Destructive periodontal disease in adults 30 years of age and older in the United States, 1988-1994. J Periodontol. 1999. 70:13–29.

5. Dowsett SA, Archila L, Kowolik MJ. Oral health status of an indigenous adult population of Central America. Community Dent Health. 2001. 18:162–166.
6. Gjermo P, Rosing CK, Susin C, Oppermann R. Periodontal diseases in Central and South America. Periodontol 2000. 2002. 29:70–78.

8. Cobb CM, Williams KB, Gerkovitch MM. Is the prevalence of periodontitis in the USA in decline? Periodontol 2000. 2009. 50:13–24.

9. Ministry of Health and Welfare. Korean National Oral Health Survey 2000. 2001. Seoul: Ministry of Health and Welfare;28–30.
10. Ministry of Health and Welfare. Korean National Oral Health Survey 2006. 2007. Seoul: Ministry of Health and Welfare;23–24.
11. Ainamo J, Barmes D, Beagrie G, et al. Development of the World Health Organization (WHO) community periodontal index of treatment needs (CPITN). Int Dent J. 1982. 32:281–291.
12. Borrell LN, Kunzel C, Lamster I, Lalla E. Diabetes in the dental office: using NHANES III to estimate the probability of undiagnosed disease. J Periodontal Res. 2007. 42:559–565.

13. Cook S, Auinger P, Li C, Ford ES. Metabolic syndrome rates in United States adolescents, from the National Health and Nutrition Examination Survey, 1999-2002. J Pediatr. 2008. 152:165–170.

14. Demmer RT, Jacobs DR Jr., Desvarieux M. Periodontal disease and incident type 2 diabetes: results from the First National Health and Nutrition Examination Survey and its epidemiologic follow-up study. Diabetes Care. 2008. 31:1373–1379.
15. Noble JM, Borrell LN, Papapanou PN, et al. Periodontitis is associated with cognitive impairment among older adults: analysis of NHANES-III. J Neurol Neurosurg Psychiatry. 2009. Forthcoming.

16. Goodson JM. Diagnosis of periodontitis by physical measurement: interpretation from episodic disease hypothesis. J Periodontol. 1992. 63:373–382.

17. Jeffcoat MK, Reddy MS. Advances in measurements of periodontal bone and attachment loss. Monogr Oral Sci. 2000. 17:56–72.

18. Haffajee AD, Socransky SS. Attachment level changes in destructive periodontal diseases. J Clin Periodontol. 1986. 13:461–475.

19. Oliver RC, Brown LJ, Löe H. Periodontal diseases in the United States population. J Periodontol. 1998. 69:269–278.

20. Bacić M, Plancak D, Granić M. CPITN assessment of periodontal disease in diabetic patients. J Periodontol. 1988. 59:816–822.

21. Ojima M, Hanioka T, Tanaka K, Inoshita E, Aoyama H. Relationship between smoking status and periodontal conditions: findings from national databases in Japan. J Periodontal Res. 2006. 41:573–579.

23. Nunn ME. Understanding the etiology of periodontitis: an overview of periodontal risk factors. Periodontol 2000. 2003. 32:11–23.

24. Taylor GW, Borgnakke WS. Self-reported periodontal disease: validation in an epidemiological survey. J Periodontol. 2007. 78:1407–1420.

25. Haber J, Wattles J, Crowley M, et al. Evidence for cigarette smoking as a major risk factor for periodontitis. J Periodontol. 1993. 64:16–23.

26. Hujoel PP, Drangsholt M, Spiekerman C, DeRouen TA. Periodontitis-systemic disease associations in the presence of smoking--causal or coincidental? Periodontol 2000. 2002. 30:51–60.

27. Laxman VK, Annaji S. Tobacco use and its effects on the periodontium and periodontal therapy. J Contemp Dent Pract. 2008. 9:97–107.

28. Nibali L, D'Aiuto F, Griffiths G, et al. Severe periodontitis is associated with systemic inflammation and a dysmetabolic status: a case-control study. J Clin Periodontol. 2007. 34:931–937.

29. Tsai C, Hayes C, Taylor GW. Glycemic control of type 2 diabetes and severe periodontal disease in the US adult population. Community Dent Oral Epidemiol. 2002. 30:182–192.

30. Merchant A, Pitiphat W, Douglass CW, Crohin C, Joshipura K. Oral hygiene practices and periodontitis in health care professionals. J Periodontol. 2002. 73:531–535.

31. Williams KB, Cobb CM, Taylor HJ, Brown AR, Bray KK. Effect of sonic and mechanical toothbrushes on subgingival microbial flora: a comparative in vivo scanning electron microscopy study of 8 subjects. Quintessence Int. 2001. 32:147–154.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download