Abstract
Background
The clinical and pathological heterogeneity of progressive supranuclear palsy (PSP) is well established. Even with a well-defined clinical phenotype and a thorough laboratory workup, PSP can be misdiagnosed, especially in its early stages.
Frontotemporal lobar degeneration, progressive supranuclear palsy (PSP), and corticobasal degeneration (CBD) have overlapping clinical and pathological features. PSP is a clinicopathological entity that typically presents as an akinetic rigid syndrome with early postural instability, axial rigidity, and variable supranuclear gaze palsy. In particular, supranuclear vertical gaze palsy, moderate or severe postural instability, and falls during the first year after onset are the main characteristics of PSP and they affect the accuracy of the clinical diagnosis (9% error according to logistic regression).1,2 We report a pathologically confirmed case of PSP that presented with behavioral changes including agitation and irritability, which eventually progressed to cardinal symptoms, such as frequent falls and supranuclear gaze palsy.
In 2004, a 52-year-old Korean woman was evaluated for sleep disturbance and behavioral changes over the previous two years. She walked around her neighborhood all day, meddling in other people's affairs and starting arguments with them. Her neighbors did not want to spend time with her, and her inappropriate behavior was worsening progressively. She visited a psychiatry clinic initiated by her husband, and was diagnosed with severe anxiety neurosis of unknown etiology. She was prescribed medicines for her insomnia and behavioral problems, but her symptoms worsened. Her husband reported that her memory had been impaired for 6 months before her first visit to us.
Her past medical and family histories were unremarkable. She graduated from high school and then worked as a secretary for a small company until she got married. She was a very kind and careful housewife.
On neurological examination, she was alert and oriented. However, she could not stay seated in a chair for more than a few minutes, instead walking around the outpatient clinic. Her speech was fluent and cranial nerve examinations produced normal results. The motor and sensory examinations were unremarkable. She complained of mild lower back pain but no gait disturbance.
She scored 23/30 on the Korean version of the Mini-Mental Status Examination3 and had a clinical dementia rating4 of 0.5. The Neuropsychiatric inventory5 revealed agitation, irritability, and disinhibition, and her husband reported her sleep disorder. Her behavioral symptoms prevented the application of detailed neuropsychological tests because of her behavioral symptoms. The laboratory data, including thyroid function tests, were normal. Brain magnetic resonance imaging (MRI) showed diffuse atrophy changes (Fig. 1A) and brain 18F-fluorodeoxyglucose positron emission tomography (18F-FDG PET) revealed subtly decreased glucose metabolism in the right prefrontal and both posterior frontal areas (Fig. 1B).
We diagnosed this patient with a behavioral variant of frontotemporal dementia at that time. Six months later, she returned to our hospital due to severe lower back pain and gait disturbance. She underwent lumbar MRI in the orthopedic clinic, but no specific abnormalities were detected. Neurological examinations revealed mild features of Parkinson's disease. Her facial expression was slightly decreased and her speech was monotonous compared to at the previous examoination. She had no tremor, but showed bradykinesia and mild symmetric rigidity. Limb apraxia and alien hand syndrome were not revealed. Her gait was short-stepped and slightly festinating, but without postural instability. Her extraocular movements showed no definite abnormalities. Our working diagnosis was FTD with parkinsonism. We requested a genetic study for FTD and parkinsonism linked to chromosome 17 (FTDP-17), but this did not reveal a genetic mutation of microtubule associated protein tau.
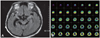
We prescribed the maximum allowable doses of the dopamine agonist and L-dopa, but thios resulted in her symptoms waxing and waning, rather than improving decisively. She gradually developed walking hesitation and was prone to falling. Vertical gaze limitation was observed in a neurological examination. We performed follow-up brain MRI and 18F-FDG PET studies. The brain MRI showed marked bilateral temporal and midbrain atrophy with ventricular dilatation, with the changes being greater in the right temporal lobe than in the left temporal lobe (Fig. 2A). Brain 18F-FDG PET revealed decreased glucose metabolism in the bilateral frontotemporal and parietal areas, with the decrease being in the right temporal lobe than in the left temporal lobe (Fig. 2B).
We made a diagnosis of probable PSP based on the National Institute of Neurological Disorders and Stroke and the Society for PSP Criteria.1 The patient started to develop difficulty swallowing and dysarthria, and these symptoms deteriorated relentlessly. Eventually, she became bedridden and required respiratory support with mechanical ventilation. Her family wanted confirmation of the diagnosis of her progressive disease, so we performed a brain biopsy and pathological evaluation. The biopsy of the temporal and frontal cortices showed tau-positive neurons (Fig. 3A) and reactive gliosis with tufted astrocytes (Fig. 3B).
Most published reports on the behavior of patients with PSP emphasize symptoms related to depression or apathy, while psychotic or obsessive features are rarely described. Litvan et al.6 investigated the behavioral symptoms of PSP, Alzheimer's disease, and controls using the NPI and noted that apathy was the most frequent behavioral abnormality; moreover, the presence of high apathy and low agitation scale scores correctly identified the patients with PSP.
In contrast, in our case, agitation and disinhibition were the earliest and most prominent behavioral symptoms. As shown previously, apathy and disinhibition are associated with progressive dysfunction of different subcortical circuits and do not proceed in parallel, and neuropsychiatric disorders are independent of cognitive dysfunction.
Five frontal-subcortical circuits unite regions of the frontal lobe with the striatum, globus pallidus, and thalamus, in functionally mediating motor activities, eye movements, cognition, and behavior. The five circuits originate in the supplementary motor area, frontal eye fields, dorsolateral prefrontal cortex, orbitofrontal cortex, and anterior cingulate cortex, which respectively mediate volitional motor activities, saccadic eye movements, executive functions, social behavior, and motivation. All five of these circuits are affected in PSP, with corresponding functional and behavioral abnormalities being evident. Apathy is associated with dysfunction of the medial frontal-subcortical circuits and disinhibition is the behavioral correlate of the orbitofrontal-subcortical circuitry.6,7
We believe that the orbitofrontal cortex, rather than the medial frontal cortex, was initially involved in our patient because her behavioral symptoms of agitation and disinhibition developed very early in her disease. The initial brain PET showed decreased glucose metabolism at right prefrontal and both posterior frontal cortices. To the best of our knowledge, agitation or disinhibition as an initial symptom of PSP is very rare, although psychosis has been reported as the presenting symptom in PSP in a few cases.8
PSP is a common cause of atypical parkinsonian syndrome with dementia. However, the diagnosis of PSP can be a difficult one, and both false-positive and false-negative misdiagnoses may occur. Pathologically confirmed cases of cardiovascular disease, diffuse Lewy body disease, multiple system atrophy, CBD, subcortical gliosis, Pick's disease, Whipple's disease, neurosyphilis, and prion disease have all been clinically misdiagnosed as PSP.9 In fact, we initially diagnosed our case as FTD before the parkinsonian features did developed. Moreover, although parkinsonian features developed, we believed that she had FTD-P-17 and did not consider PSP because the clinical hallmarks such as a falling tendency or supranuclear gaze abnormality were not evident at that time. Litvan et al.1 noted that several features should raise suspicion of PSP, including early instability and falls and vertical supranuclear palsy particularly during the first year of symptom onset.
Williams et al., recently noted that pathologically confirmed cases show two clinical phenotypes based on factor analysis, which they named Richardson's syndrome and PSP-parkinsonism (PSP-P). RS, like classical PSP, is characterized by early-onset postural instability and falls, supranuclear gaze palsy, and cognitive decline. Conversely, PSP-P initially presents with parkinsonian features and is frequently confused with Parkinson's disease. They proposed that PSP-P represented a distinct clinical phenotype because different tau isoform deposition was seen in the basal pons, and this needs to be distinguished clinically from classical PSP.10 Based on previous reports and our case, gait instability and gaze abnormality are not absolute requirements as presenting symptoms for a diagnosis of probable PSP, and the initial clinical diagnosis criteria are not mandatory.
The tau-positive neurodegenerative diseases are characterized by tau-positive inclusions and include PSP, CBD, Pick's disease, multiple system atrophy, and FTDP-17.11 However, tauopathy-neurodegenerative diseases present some unique histopathologic features. Focal asymmetric cortical atrophy with ballooned neurons, nigral degeneration and astrocytic plaques in the affected cortex are characteristic features of CBD. On the other hand, the distinctive cytopathologic findings of PSP are globose-type neurofibrillary tangles in the brainstem, and substantia nigra, and tufted astrocytes, which are abundant in the precentral gyrus and striatum.12 The pathologic results in our case revealed globose tau-positive neurons and tufted astrocytes, which are findings more specific to PSP, but not astrocytic plagues and ballooned neurons. Therefore, we consider that the pathological findings supported a diagnosis of PSP, but not CBD or FTDP-17 even though biopsy specimens were taken from the frontal and temporal cortices. We have reported here in the case of a patient who presented with unusual behavioral symptoms, along with her clinical course with pathological data.
Figures and Tables
Fig. 1
A: Brain magnetic resonance imaging shows diffuse brain atrophy on fluid-attenuated inversion recovery images. B: Brain 18F-fluorodeoxyglucose positron emission tomography reveals subtly decreased glucose metabolism in the right prefrontal and both posterior frontal areas.
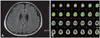
Fig. 2
A: Brain MRI shows marked bilateral temporal and midbrain atrophy with ventricular dilatation that is worse in the right temporal lobe than in the left temporal lobe on fluid-attenuated inversion recovery images. B: Brain 18F-fluorodeoxyglucose positron emission tomography reveals decreased glucose metabolism in the bilateral frontotemporal and parietal areas, with the decrease being in the right temporal lobe than in the left tempopral lobe.
Acknowledgements
This study was supported by a grant of the Korea Health 21 R&D Project, Ministry of Health & Welfare, Republic of Korea (A050079).
References
1. Litvan I, Agid Y, Calne D, Campbell G, Dubois B, Duvoisin RC, et al. Clinical research criteria for the diagnosis of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome): report of the NINDS-SPSP international workshop. Neurology. 1996. 47:1–9.

2. Pearce JM. Progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome): a short historical review. Neurologist. 2007. 13:302–304.

3. Kang Y, Na DL, Hahn S. A validity study on the Korean. Mini-mental state examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997. 15:300–308.
4. Choi SH, Na DL, Lee BH, Hahm DS, Jeong JH, Yoon SJ, et al. Estimating the validity of the Korean version of expanded clinical dementia rating (CDR) scale. J Korean Neurol Assoc. 2001. 19:585–591.
5. Cummings JL. The Neuropsychiatric inventory: assessing psychopathology in dementia patients. Neurology. 1997. 48:S10–S16.

6. Litvan I, Mega MS, Cummings JL, Fairbanks L. Neuropsychiatric aspects of progressive supranuclear palsy. Neurology. 1996. 47:1184–1189.

7. Grafman J, Litvan I, Gomez C, Chase TN. Frontal lobe function in progressive supranuclear palsy. Arch Neurol. 1990. 47:553–558.

8. Trzepacz PT, Murcko AC, Gillespie MP. Progressive supranuclear palsy misdiagnosed as schizophrenia. J Nerv Ment Dis. 1985. 173:377–378.

9. Murphy MA, Friedman JH, Tetrud JW, Factor S. Neurodegenerative disorders mimicking progressive supranuclear palsy: a report of three cases. J Clin Neurosci. 2005. 12:941–945.

10. Williams DR, de Silva R, Paviour DC, Pitman A, Watt HC, Kilford L, et al. Characteristics of two distinct clinical phenotypes in pathologically proven progressive supranuclear palsy: Richardson's syndrome and PSP-parkinsonism. Brain. 2005. 128:1247–1258.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download