Abstract
Corticobasal degeneration (CBD) is a progressive neurodegenerative disorder characterized by asymmetric parkinsonism associated with apraxia, cortical sensory loss, and alien-limb phenomenon. Neuropsychological testing in patients with CBD typically shows deficits in executive functions, praxis, language, and visuospatial functioning, but not in memory. We report a CBD patient with severely impaired memory function but relatively mild motor symptoms. Detailed neuropsychological assessment showed significant verbal and visual memory deficits accompanied by frontal executive dysfunctions. Our observations indicate that CBD can in rare cases present with severe episodic memory impairment associated with frontal executive dysfunctions in the early stage of illness.
Corticobasal degeneration (CBD) is a rare neurodegenerative disorder involving the cortical and subcortical structures that causes movement abnormalities with several cortical signs including cognitive impairment. CBD is clinically characterized by a distinctive levodopa-resistant progressive asymmetric dystonic-akinetic-rigid syndrome associated with cortical features such as apraxia, cortical sensory loss, myoclonus, and alien-limb phenomenon.1,5 However, speech problems and cognitive impairment are not frequent in CBD patients. Moreover, cognitive functions are usually preserved in the early stages of CBD, especially in the memory domain. A detailed neuropsychological assessment including episodic memory tasks has not yet to be reported for the typical CBD case. In general, neuropsychological assessment in patients with CBD shows deficits in executive functions, praxis, language, and visuospatial functioning, but not in episodic memory. Here we report a CBD patient who had severely impaired verbal memory functions with relatively mild motor symptoms.
A 55-year-old man presented with difficulties in hand coordination and dressing himself that had first appeared in early 2006. It took him a long time to put on his clothes because he had difficulty distinguishing between the front and back of clothing, and needed assistance with buttoning and zipping up his clothing. A severe memory disturbance that had also developed was slowly progressing.
A neurological examination showed typical cortical signs including severe apraxia, cortical sensory loss, myoclonus, and alien-limb phenomenon that predominantly affected the right arm. There was akinesia and rigidityin both extremities, with mild asymmetry. There were no prominent visuospatial problems, including simultanagnosia, visual inattention, oculomotor apraxia, or optic ataxia. A detailed neuropsychological assessment revealed prominent verbal and visual memory deficits with marked frontal executive dysfunctions (Table). The patient scored 17/30 on the Korena version of Mini-Mental State Examination, with the subscore for time orientation being 3/5 and a delayed three-word recall of 0/3. He exhibited an abnormal digit span on attention tests and showed severe ideomotor and ideational apraxia on several praxis tasks. His language functions were relatively preserved. His performance on copying in the Rey-Osterrieth Complex Figure Test was impaired. On the Seoul Verbal Learning Test, he was able to recall two items (<percentile 1) with a 20-minute delayed recall. He also scored poorly in delayed recall in the Rey-Osterrieth Complex Figure Test (0/36, percentile 2). His performances on the tasks of Controlled Oral-Verbal Fluency and Stroop Test were also severely impaired.
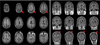
Brain MRI performed 18 months after the onset of the symptoms revealed significant cortical atrophy in both parietal areas that were more prominent on the left side with diffuse cortical atrophic changes in T1-weighted images (Fig. 1). Brain PET showed prominent asymmetric (left-dominant) hypometabolism in both parietal areas, with significant metabolic deficits in the left temporal lobes (Fig. 2). Mildly decreased metabolism was also observed in both frontal areas.
Cognitive impairments such as severe amnesia and visuospatial abnormalities were initially thought to be a rare or late presenting trait in CBD, with cognitive functions being relatively spared until the late stages of CBD and higher mental function being relatively preserved in CBD patients.1,7 Clinical descriptions of CBD (mostly from movement disorder clinics) have emphasized motor manifestations such as parkinsonian features, apraxia, myoclonus, gaze palsies, and alien-limb phenomenon. Research focused on the motor symptoms may have led to the notion that cognitive impairment or dementia occurs only in a few patients with CBD.6,7 Postmortem pathological studies of CBD show neuronal loss, swollen achromatic neurons, and diffusely stained tau-positive astrocytic plaques. These changes typically involve the cortical and subcortical areas.2,3 Asymmetric cortical atrophy involves mainly the superior parietal and frontal lobes, with smaller effects in the temporal and occipital lobes.4
Several recent studies have documented that cognitive dysfunctions and language disturbances in the early stage of the disease course are not rare manifestations in CBD patients.8,9 However, the current findings related to episodic memory functioning in CBD are not described well by comprehensive cognitive assessments. Our patient showed prominent memory impairment in several cognitive domains upon a detailed neuropsychological evaluation and history taking by his caregiver. The results of the word-list learning test as a verbal memory task indicated severe impairment of encoding, resembling the learning process frequently seen in patients with Alzheimer's disease (AD). Very few case studies have found abnormalities with respect to episodic memory test using the story recall test in patients with CBD.10-12 In general, CBD patients perform better on story recall and word list tasks than matched AD patients.11,13 The impairment of episodic memory appears to be less severe in CBD patients than in AD patients. In AD, poor strategic processes in frontal lobe dysfunctions or disruption of frontal-subcortical circuits leads to episodic memory impairment. However, the pattern of memory deficits in our patient differed from that typical of AD.12 The prominent memory deficits in our case can be explained by additional cortical hypometabolism in the left temporal area.
Posterior cortical atrophy14 can be included in the differential diagnosis of CBD. However, there were no prominent visual complaints with typical presentations of Balint's syndrome in our case. Although significant visuospatial and constructive dysfunctions were observed when our patient was asked to draw interlocking pentagons and Rey-Osterrieth figures, those deficits were augmented by severe hand apraxia. Our patient also showed severe frontal subcortical circuit deficits when asked to perform several tasks of executive functioning. The frontal lobe dysfunctions could be explained by the significant hypometabolism in both frontal areas.
In summary, our patient presented with severe episodic memory impairment and frontal executive dysfunctions at an early stage of CBD. However, other neurodegenerative diseases such as AD or other focal dementia syndromes associated with parkinsonism cannot be completely ruled out without a postmortem pathologic diagnosis.
Figures and Tables
Figure 1
Brain MRI showing significant cortical atrophy in both parietal areas, which was more prominent on the left side with diffuse cortical atrophic changes in T1-weighted images.
References
1. Riley DE, Lang AE, Lewis A, Resch L, Ashby P, Hornykiewicz O, et al. Cortical-basal ganglionic degeneration. Neurology. 1990. 40:1203–1212.

2. Boeve BF, Maraganore DM, Parisi JE, Ahlskog JE, Graff-Radford N, Caselli RJ, et al. Pathologic heterogeneity in clinically diagnosed corticobasal degeneration. Neurology. 1999. 53:795–800.

3. Piao YS, Hayashi S, Wakabayashi K, Kakita A, Aida I, Yamada M, et al. Cerebellar cortical tau pathology in progressive supranuclear palsy and corticobasal degeneration. Acta Neuropathol. 2002. 103:469–474.

4. Soliveri P, Monza D, Paridi D, Radice D, Grisoli M, Testa D, et al. Cognitive and magnetic resonance imaging aspects of corticobasal degeneration and progressive supranuclear palsy. Neurology. 1999. 53:502–507.

5. Riley D, Lang AE. Corticobasal degeneration. Clinical diagnostic criteria. Adv Neurol. 2000. 82:29–34.
6. Wenning GK, Litvan I, Jankovic J, Granata R, Mangone CA, McKee A, et al. Natural history and survival of 14 patients with corticobasal degeneration confirmed at postmortem examination. J Neurol Neurosurg Psychiatry. 1998. 64:184–189.

7. Rinne JO, Lee MS, Thompson PD, Marsden CD. Corticobasal degeneration. A clinical study of 36 cases. Brain. 1994. 117:1183–1196.

8. Grimes DA, Lang AE, Bergeron CB. Dementia as the most common presentation of cortical-basal ganglionic degeneration. Neurology. 1999. 53:1969–1974.

9. Schneider JA, Watts RL, Gearing M, Brewer RP, Mirra SS. Corticobasal degeneration: neuropathologic and clinical heterogeneity. Neurology. 1997. 48:959–969.

10. Pillon B, Blin J, Vidailhet M, Deweer B, Sirigu A, Dubois B, et al.
. Neurology. 1995. 45:1477–1483.
11. Massman PJ, Kreiter KT, Jankovic J, Doody RS. Neuropsychological functioning in cortical-basal ganglionic degeneration: Differentiation from Alzheimer's disease. Neurology. 1996. 46:720–726.

12. Pillon B, Dubois B. Memory and executive processes in corticobasal degeneration. Adv Neurol. 2000. 82:91–101.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download