Dear Editor,
Posterior reversible encephalopathy syndrome (PRES) is a clinicoradiologic syndrome characterized by predominant parietal and occipital lobe edema that is mainly reversible within a few days.12 However, many atypical patterns have been identified.3 Reversibility is not always achieved,3 which exposes a contradiction in this supposedly benign entity. We report the case of a 14-year-old patient who presented with classical signs of PRES, but whose condition evolved into a malignant phenotype.
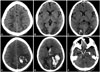
A 14-year-old patient received a renal transplant from a cadaveric donor and took tacrolimus, corticosteroids, and antithymocyte immunoglobulin. On the third day, his renal function had not improved and hemodialysis was started. He then immediately developed headache, vomiting, and experienced a tonic-clonic seizure. His blood pressure (BP) peaked at 240/150 mm Hg. Brain CT revealed bilateral edema in the parieto-occipital regions associated with a small hemorrhagic transformation, representing PRES (Fig. 1A, B, and C). Laboratory tests revealed thrombocytopenia (66,000/µL) and elevated serum creatinine (2.52 mg/dL). Tacrolimus was suspended and sodium nitroprusside was administered, which fully controlled his hypertension.
On the following day his BP was 147/90 mm Hg but his neurological function had deteriorated. Follow-up brain CT revealed extensive cortical and white-matter edema associated with parenchymatous, subarachnoid, and intraventricular hemorrhage (Fig. 1D, E, and F). Brain death was later confirmed, leading to the suspension of life support.
PRES pathophysiology remains a mystery even after almost 20 years since its initial description.1 There are two prevailing hypotheses: cytotoxic and vasogenic.45 Eclampsia, renal failure, autoimmune diseases, and chemotherapy treatment and other factors may trigger PRES.15 Despite its classically favorable outcome, PRES is associated with direct mortality in 5-15% of cases.67 Important prognostic factors within PRES have been suggested, such as the anatomical distribution of edema, hemorrhage, and cytotoxic edema in diffusion-weighted imaging sequences, but they have yet to be proven.78 Alhilali et al.7 further analyzed the prognostic factors in PRES and found that hemorrhage, even if minor, is the most relevant factor associated with a poor outcome, which may indicate more extensive endothelial dysfunction.
Hefzy et al.6 found that hemorrhagic transformation occurred in 17% of PRES cases, mainly small or petechial hemorrhages (<5 mm) and hematomas, but also subarachnoid and intraventricular bleeding. The rates found in other studies have varied from 6.4% to 19.4%, with mortality reaching 26-29% in this group.67 The precipitating illness is usually severe, and PRES may be only a marker of such severity, but novel reports unveil the malignant evolution of this condition.78 Although many terms have been used to describe this event,7 malignant PRES seems to best encompass the permanent neurological damage or even death that sometimes accompanies this endothelial breakdown in the brain.
The optimal management of PRES has yet to be elucidated.8 Better outcomes in malignant PRES have been attributed to aggressive neurointensive care, decompressive craniectomy, and intracranial pressure management.8 These results are promising, but larger studies are needed to confirm the efficacy of this approach.
PRES is usually a benign entity; however, it represents a small outlier in the clinical spectrum of this syndrome. A wider range of neurological disabilities encompasses this syndrome and leads to a malignant evolution, suggesting that a good prognosis cannot be assumed with PRES, but rather its occurrence should prompt immediate action to avoid disability.
Figures and Tables
Fig. 1
Brain CT in the first presentation. A: Subcortical edema extends to the parietal and frontal region. B and C: Bilateral occipital edema is demonstrated, with minor hemorrhagic transformation, suggesting posterior reversible encephalopathy syndrome. D, E, and F: Control brain CT 24 hours later shows extensive intraparenquimatous bleeding (D), associated to intraventricular (E) and subarachnoid hemorrhage (F).
References
1. Hinchey J, Chaves C, Appignani B, Breen J, Pao L, Wang A, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996; 334:494–500.

2. Casey SO, Sampaio RC, Michel E, Truwit CL. Posterior reversible encephalopathy syndrome: utility of fluid-attenuated inversion recovery MR imaging in the detection of cortical and subcortical lesions. AJNR Am J Neuroradiol. 2000; 21:1199–1206.
3. McKinney AM, Short J, Truwit CL, McKinney ZJ, Kozak OS, SantaCruz KS, et al. Posterior reversible encephalopathy syndrome: incidence of atypical regions of involvement and imaging findings. AJR Am J Roentgenol. 2007; 189:904–912.

4. Porcello Marrone LC, Gadonski G, de Oliveira Laguna G, Poli-de-Figueiredo CE, Pinheiro da Costa BE, Lopes MF, et al. Blood-brain barrier breakdown in reduced uterine perfusion pressure: a possible model of posterior reversible encephalopathy syndrome. J Stroke Cerebrovasc Dis. 2014; 23:2075–2079.

5. Marrone LC, Gadonski G, Diogo LP, Brunelli JP, Martins WA, Laguna Gde O, et al. Posterior reversible encephalopathy syndrome: differences between pregnant and non-pregnant patients. Neurol Int. 2014; 6:5376.

6. Hefzy HM, Bartynski WS, Boardman JF, Lacomis D. Hemorrhage in posterior reversible encephalopathy syndrome: imaging and clinical features. AJNR Am J Neuroradiol. 2009; 30:1371–1379.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download