Abstract
Background
Psoriasis is an autoimmune disease that is caused by a shift in the Th1/Th2 balance toward Th1-dominant immunity. It has been established as an effective treatment to counteract psoriasis by subcutaneous injection of recombinant interleukin (IL)-4, and IL-4 gene therapy by topical transdermal penetration has shown its antipsoriatic effect in mice. Retinoic acid (RA) and dimethylsulfoxide can increase the efficiency of gene transfection in the topical transdermal delivery system.
Objective
We investigated whether RA could improve anti-psoriasis efficiency using IL-4 expression plasmid pORF-mIL-4 (pIL-4) via transdermal delivery system in K14-vascular endothelial growth (K14-VEGF) factor transgenic mice.
Methods
After pretreatment with RA, plasmid pIL-4 in 10% dimethylsulfoxide was applied to the ear skin by topical transdermal penetration. Hematoxylin- eosin staining and immunohistochemistry were performed with ear samples to evaluate anti-psoriasis efficiency in mice.
Psoriasis is an autoimmune disease that is caused by a shift in Th1/Th2 balance toward Th1-dominant immunity. Histological hallmarks of psoriatic skin include the infiltration of multiple immune cells, keratinocyte proliferation and increased dermal vascularization. A combination of genetic predisposition and environmental factors would probably contribute to its autoimmune process1. Fibroblasts, keratinocytes, antigen-presenting cells, T cells, and endothelial cells have all been proposed as candidates for the primary defect2. It is likely that abnormal regulation of T cell-keratinocyte interaction with a complex cytokine network is involved in the pathogenesis of psoriasis3,4. It has been identified that Th17 cells, which can produce specific cytokine interleukin (IL)-17 and IL-22, are distinct from Th1 cells in the dermis of psoriatic subjects. IL-17 has proinflammatory effects in a variety of cells including keratinocytes, macrophages, and endothelial cells5. It has been confirmed that subcutaneous injection with IL-4 can induce Th2 differentiation in CD4+ T cells and has been proved as a potential treatment for psoriasis6,7.
As previously described8, we have confirmed that efficient delivery of transgenes to mouse skin was increased by the use of retinoic acid (RA) and dimethylsulfoxide (DMSO). DMSO has been shown to promote the permeation of, for example, antiviral agents, steroids and antibiotics9. We also confirmed that 10% DMSO was the optimal transdermal delivery formula for further examination in vivo8,10. These results provide a foundation for increasing the efficiency of gene transdermal penetration with DMSO.
RA carries out most of the biological activities of vitamin A and is required for normal growth, cell differentiation, and immune functions11. Therapeutic RA has profound effects on psoriatic skin pathology due to its interactions with various retinoid-binding proteins in keratinocytes12. Some researchers have demonstrated that RA could also markedly increase the transfection efficiency of gene to skin13,14. And we also have developed a novel method for topical skin gene delivery that utilized RA pretreatment and transdermal delivery formulation containing 10% DMSO8. The transfection efficiency of report plasmid pORF-LacZ was higher with the pretreatment of RA in our delivery system where DMSO was used as a gene intermediary. All these findings provide a more effective approach for the subsequent treatment of psoriasis with topical gene therapy.
In this paper, we investigated whether the antipsoriatic effect of IL-4 could be improved by transportation across the stratum corneum via the RA-DMSO mediated transdermal penetration delivery system.
The pORF-mIL-4 (pIL-4) plasmid, containing the murine IL-4 gene, and pORF-mcs (the empty vector) plasmid, the empty vector plasmid, were purchased from InvitroGen (San Diego, CA, USA). All these two plasmids were purified with an Endo-Free plasmid purification kit (Qiagen, Hilden, Germany).
K14-vascular endothelial growth factor (K14-VEGF) transgenic mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA)15. K14-VEGF transgenic homozygous mice (3 months old) with moderate psoriasis were chosen for the experiment. All studies involving mice (n=24) were approved by the Animal Experimental Center of Sichuan University.
To evaluate the anti-psoriasis effect of the IL-4 gene in the psoriasis mouse model, pIL-4 was prepared for the pharmacodynamic study as a therapeutic plasmid, and MCS (the empty vector plasmid), as a control. A total of five formulas (Table 1) were prepared in the in vivo gene delivery assay as follows: (a) pretreatment with 0.05% RA, pIL-4/10% DMSO complex (mixture contained pIL-4 plasmid and 10% DMSO); (b) pIL-4/10% DMSO complex, without RA pretreatment; (c) pretreatment with RA, MCS-10% DMSO (mixture contained empty vector plasmid and 10% DMSO); (d) MCS-10% DMSO, without RA pretreatment; and (e) saline solution. Formulas (c) and (d) were used as controls, and formula (e) as a negative control. The quantity of pIL-4 and empty vector plasmid respectively was 10 µg, and the total drug delivery volume was 10 µl.
Twenty-five female K14-VEGF mice were picked out for our experiment. There were five mice in each group. After treating with 0.09% physiological saline for 15 minutes, ears of the mice in group (a) and (c) were treated with 20 µl 0.05% RA (dissolved in 0.09% physiological saline) every other day for one week. At the seventh day, four hours after the ears were pretreated with RA; the mixture containing pIL-4 or MCS and 10% DMSO was pipetted topically with 10 µl aliquots on the ear skin (plasmid concentration was 1 µg/µl) with a micropipette once a day, respectively, for one month. On day 31, photos of the ears were obtained, All of the ears were harvested and subsequently subjected to histological and immunohistochemical analyses, as described subsequently.
After being photographed, the ears of all mice for the experiments were harvested. They were fixed with 4% paraformaldehyde, and the paraffin-embedded ear samples were sectioned into 5 µm thickness. After they were deparaffinized, the sections were stained with H&E and analyzed immunohistochemically. The pathological score based on the Baker score system was obtained to assess the severity of the psoriasis16. Ear sections were immunostained with E-Selectin (CD62E, diluted 1 : 100; Boster, Wuhan, China), anti-mouse intracellular adhesion molecule-1 (ICAM-1) (CD54, diluted 1 : 100; Boster), anti-mouse vascular cell adhesion molecule-1 (VCAM-1) (CD106, diluted 1 : 100; Boster), anti-mouse platelet-endothelial cell adhesion molecule-1 (PECAM-1) (CD31, diluted 1 : 100; Santa Cruz Biotechnology, Santa Cruz, CA, USA), antimouse VEGF (diluted 1 : 100; Boster) and anti-mouse VEGF receptor-2 (VEGFR-2, diluted 1 : 100; BD Pharmingen, San Diego, CA, USA) in accordance with the manufacturers' instructions.
Data are provided as the means. Differences between the groups were tested by using analysis of one-way ANOVA with a post hoc Student-Newman-Keuls multiple comparisons test. Statistical analyses were performed by SPSS software (version 17.0; SPSS Inc., Chicago, IL, USA). p< 0.05 was considered statistically significant.
We have confirmed that pretreatment of RA can promote efficient delivery of transgenes in 10% DMSO to mouse skin by topically transdermal penetration8. Here, we determined the therapeutic effect when pIL-4 was applied to K14-VEGF transgenic homozygous mice with a serious inflammatory skin condition, with many of the cellular and molecular features of psoriasis, including the characteristic vascular changes, epidermal alterations, and inflammatory infiltration15. After the treatment with pIL-4 in 10% DMSO for one month, psoriasis symptoms including erythematous, scaly and thickened skin lesions (Fig. 1) were clearly relieved with the RA pretreatment. Ears that were pretreated with RA and then treated with pIL-4 in 10% DMSO showed only hyperemia and slight skin thickness, and there was also a conspicuous decrease in inflammatory cells (1B in Fig. 1). In the treatment group of pIL-4/10% DMSO complex, in the defect of RA pretreatment, the epidermis of the ears exhibited more thickened skin lesions and rete ridges, and a definite inflammatory cell infiltration was observed in the dermis. Obviously, RA treatment could make a significant impact on the therapeutic effect of pIL-4/10% DMSO complex for psoriasis. Obviously, regardless of whether RA pretreatment was applied or not, mice in the groups treated with MCS-DMSO and with saline all showed typical psoriasiform features in the ear tissue sections (Fig. 1).
The pathological scores of the mice treated with these five formulas are shown in Table 2. The data verified that there were significant differences between the RA and pIL-4/10% DMSO complex group and the other four groups. No significant difference was observed among the control groups and the negative group. While pIL-4/10% DMSO complex treatment with RA absence also produced a certain degree of relief in psoriatic symptoms in our study, these results coincided with previous research10.
The expressions of E-selectin17,18, CD54 (ICAM-1) and CD 106 (VCAM-1) are a hallmark of the hyperplastic and inflamed vessels in human psoriatic skin lesions. Similar to human psoriasis, the expressions of these cell adhesion molecules were prominent in the hyperplastic vessels in the psoriasiform skin of K14-VEGF transgenic mice15. In our research, compared to the control groups, expressions of E-selectin, CD54, and CD106 were down regulated in the treatment groups of pIL-4/10% DMSO complex, with or without RA pretreatment. We also found that the expression of these cell adhesion molecules in the group treated with RA-pIL-4/10% DMSO complex was lower than the group treated with pIL-4/10% DMSO complex only (Fig. 2). Our results illustrated that inflammation was relieved with the treatment of pIL-4-DMSO, and more importantly, RA pretreatment enhanced the anti-inflammatory effects of pIL-4-DMSO in gene therapy for psoriasis in K14-VEGF transgenic mouse.
Furthermore, CD31 was also decreased in vascular endothelial cells in the groups of RA-pIL-4-DMSO and pIL-4-DMSO, but there was no significant difference in the inhibition of hyperplastic cutaneous blood vessels in the treatment groups of pIL-4-DMSO, regardless of whether RA pretreatment was applied or not. By contrast, much more elongated and enlarged vessels was detected in the control mice of the other three groups (Fig. 2), with this feature being similar to the defining characteristic of human psoriatic skin15. VEGF and VEGFR-2 were also observed in all immunohistochemically stained ear sections with special antibodies; nevertheless, there were no significant variations in any of their expressions in the mice (Fig. 2).
All the data obtained in our study suggested that RA pretreatment promoted the antipsoriasis effect of pIL-4-DMSO treatment via the topical transdermal delivery system.
Psoriasis is an immune-mediated skin condition that affects 2% to 3% of the world's population at present19,20. It not only affects patients' health-related quality of life but it also results in a significant financial burden. The pathogenesis of psoriasis is complex and both genetic and environmental factors are involved in its pathogenesis21. Although the mechanism of trigger in psoriasis is unclear yet, many researchers7,10,22 have verified that IL-4 is one of the great potential candidates for the treatment of psoriasis. As previously described8, we have developed a novel method for topical gene delivery that utilized RA pretreatment and DMSO as enhancers of gene transfer into skin by transdermal penetration, and the result indicated that RA pre-treatment can increase the efficiency of transfection. In other studies, researchers have demonstrated that RA can markedly increase the transfection efficiency of gene to skin13. So, in this study, we investigated whether RA could improve the effect of IL-4 gene therapy for psoriasis by use of a delivery system of transgenes to mouse skin.
Psoriasis is associated with inappropriate vascular expansion within the superficial dermis, abnormal keratinocyte proliferation and differentiation, and inflammation. In our study, under the premise of RA pretreatment, these pathological characteristics of psoriasis were improved markedly in K14-VEGF transgenic mice after the treatment of RA-pIL-4-DMSO. The pathological score of the mice treated with RA-pIL-4-DMSO was decreased nearly 50% compared to mice treated only with pIL-4-DMSO. Obviously, RA pretreatment made the treatment of pIL-4-DMSO for psoriasis more effective.
Vessels were highly aberrant in the dermis of psoriatic skin22,23. Some researchers have proved that E-selectin CD54 and CD106 are over expressed in inflamed vasculature in psoriatic lesions24,25. In our experiment, in contrast with the pIL-4-DMSO application formula without RA, more significant reductions of the expression of these cell adhesion molecules were presented in the formula using RA-pIL-4-DMSO (Fig. 2).
Former studies have reported that psoriatic vasculogenesis is one of the pathological phenotypes in human psoriasis26. Formation of new blood vessels starts with early psoriatic changes and disappears with disease clearance27. In our study, although both pIL-4-DMSO and RA-pIL-4-DMSO remarkably suppressed the expression of CD31, there were no significant differences in the inhibition of hyperplastic cutaneous blood vessels whether RA pretreatment was applied or not (Fig. 2). Psoriasis pathogenesis lies at the crossroads linking the pathways of angiogenesis and inflammation. Several angiogenic mediators like VEGF, and angiopoietins are up-regulated in psoriasis development. These mediators may play significant roles in the pathophysiology of psoriasis and may even account for the maintenance of the chronic inflammatory state27. However, compared to control mice, there was no obvious inhibition of over-expressed VEGF in the mice treated by RA-pIL-4-DMSO or pIL-4-DMSO in our study, and the expression of VEGFR-2 in all groups was nearly at the same level. These results possibly reveal that there was no interaction between mIL-4 and VEGF. Otherwise the inconsistency may also be attributed to the psoriasis model, VEGF transgenic mouse. VEGF is widely and constitutively expressed in this psoriasis mouse model15, and the effect of IL-4 aiming at VEGF may be hard to detect.
RA and its derivatives have been used successfully for the treatment of psoriasis and other dermatoses28. RA exerts its therapeutic effects through binding to two groups of nuclear receptors, the RA receptors (RAR) and the retinoid X receptors (RXR). RAR usually form heterodimers with RXR, and these dimers bind to specific DNA sequences, the RA-responsive elements. In this way, RA down regulates the production of cytokines such as interferon-γ, tumor necrosis factor-α, IL-1. These cytokines play an important role in inflammatory and immune responses and are involved in the pathogenesis of psoriasis29,30. In addition, DMSO also has a potentially anti-inflammatory property31. In Table 2, the clinical scores of RA-DMSO-MCS and DMSO-MCS appear to significantly differ; we predict this may be the result of the considerable anti-inflammatory effect of RA in psoriasis.
The penetration and the expression level of plasmid IL-4 were important in our research. We have evaluated the secreted expression of IL-4 in mouse serum by ELISA (data not shown). Maybe the level of IL-4 in the serum was too low to be detected. The IL-4 protein expressed by pIL-4 may directly target other inflammatory cells and regulate the immune response in the K14-VEGF transgenic mice. It has been well established for many years that Th1 cells and cytokines are elevated in psoriatic tissue32. According to recent data, a new type of T cell, Th17 has been discovered in the pathogenesis of autoimmune inflammation. Th1 mediated responses have been shown to contain mixed populations of Th1 and Th17 cells in psoriasis tissues, especially in the dermis33,34. So, the relationship between IL-4 and Th17 is worth studying in the treatment of psoriasis.
In conclusion, this work strongly demonstrates that topical application of IL-4 combined with 10% DMSO after pretreatment with RA has significantly greater efficacy against psoriasis in K14-VEGF transgenic mice. These results may provide a foundation for the treatment of psoriasis and other skin disorders that uses a more efficient approach of topical gene therapy.
Figures and Tables
 | Fig. 1Histological phenotype improvement of the ear after administration in a pharmacodynamic experiment. (A1~A5) Macroscopic characteristics of the ears. Images show macroscopic findings of the mice after treatment with A1: RA+DMSO+IL-4; A2: DMSO+IL-4; A3: RA+DMSO+MCS; A4: DMSO+MCS; A5: saline. (B1~B5) Pathological characteristics of the ears. Images show H&E staining of the ears after treatment with B1: RA+DMSO+IL-4; B2: DMSO+IL-4; B3: RA+DMSO+ MCS; B4: DMSO+MCS; B5: saline (original magnification ×200). Thickness A refers to the epidermis thickness of the other four groups; thickness B was the epidermis thickness of RA+DMSO+pIL-4. Vertical ordinate was the ratio of other group to RA+DMSO+pIL-4. For the rectangle, the top and bottom respectively means the epidermal thickness ratio of the maximum to the minimum. Thick solid lines mean the mean value. Thickness A: RA+DMSO+IL-4; DMSO+IL-4; RA+DMSO+ MCS; DMSO+MCS; and saline. Thickness B: RA+DMSO+IL-4. Bars show mean±standard error of the mean (n=4). RA: retinoic acid, DMSO: dimethylsulfoxide, IL-4: interleukin-4, MCS: pORF -mcs (multiple cloning site). *p<0.05. |
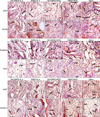 | Fig. 2Immunohistochemical staining of the ears after antipsoriasis treatment experiment. Sections were stained with special antibodies (Psoriasis-associated immunological factors were significant reduced after treatment). Immunohistochemical staining with CD54, CD106, E-selectin, CD31, VEGF and VEGFR-2 of the ears after being treated with the five formulations (RA+DMSO+IL-4; DMSO+IL-4; RA+DMSO+MCS; DMSO+MCS; and saline); Arrows in the images indicate positive cells stained with the corresponding antibody (original magnification ×400). RA: retinoic acid, DMSO: dimethylsulfoxide, IL-4: interleukin-4, VEGF: vascular endothelial growth factor, MCS: pORF-mcs (multiple cloning site). |
Table 2
Pathological score showing the more remarkable antipsoriasis effects of RA+DMSO+IL-4 treatment in K14-VEGF transgenic mice

Values are presented as mean±standard deviation. RA: retinoic acid, DMSO: dimethylsulfoxide, IL-4: interleukin-4, VEGF: vascular endothelial growth factor, MCS: pORF-mcs (multiple cloning site). *p<0.01 compared to the groups of DMSO+MCS and saline. †p<0.05 compared to the groups of DMSO+IL-4 and RA+DMSO+MCS (n=4).
ACKNOWLEDGMENT
This work was supported by a grant from the National Science and Technology Major Projects (2012ZX1000 2006-003-001) and Natural Science Foundation of China (31271483/C0706). We thank members of the State Key Laboratory of Biotherapy for help and discussion. The authors declare that they have no competing interests.
References
1. Reich A, Szepietowski J. Genetic and immunological aspects of the pathogenesis of psoriasis. Wiad Lek. 2007; 60:270–276.
2. Boniface K, Lecron JC, Bernard FX, Dagregorio G, Guillet G, Nau F, et al. Keratinocytes as targets for interleukin-10-related cytokines: a putative role in the pathogenesis of psoriasis. Eur Cytokine Netw. 2005; 16:309–319.
3. Lowes MA, Bowcock AM, Krueger JG. Pathogenesis and therapy of psoriasis. Nature. 2007; 445:866–873.

4. Pastore S, Mascia F, Mariotti F, Dattilo C, Girolomoni G. Chemokine networks in inflammatory skin diseases. Eur J Dermatol. 2004; 14:203–208.
5. Harrington LE, Hatton RD, Mangan PR, Turner H, Murphy TL, Murphy KM, et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat Immunol. 2005; 6:1123–1132.

6. Ghoreschi K, Thomas P, Breit S, Dugas M, Mailhammer R, van Eden W, et al. Interleukin-4 therapy of psoriasis induces Th2 responses and improves human autoimmune disease. Nat Med. 2003; 9:40–46.

7. Ren X, Li J, Zhou X, Luo X, Huang N, Wang Y, et al. Recombinant murine interleukin 4 protein therapy for psoriasis in a transgenic VEGF mouse model. Dermatology. 2009; 219:232–238.

8. Chen X, Zhang Y, Liu C, Zhang Y, Zhou X, Zhou T, et al. Retinoic acid and dimethyl sulfoxide promote efficient delivery of transgenes to mouse skin by topically transdermal penetration. Drug Deliv. 2010; 17:385–390.

9. Lee VH. Advanced drug delivery reviews: cornerstone in the stimulation and dissemination of innovative drug delivery research. Adv Drug Deliv Rev. 2004; 56:1–2.

10. Zhang Y, Li J, Liu CY, Zhou XK, Qiu J, Zhang YB, et al. A novel transdermal plasmid-dimethylsulfoxide delivery technique for treatment of psoriasis. Dermatology. 2010; 221:84–92.

11. Chen Q, Ross AC. Retinoic acid regulates cell cycle progression and cell differentiation in human monocytic THP-1 cells. Exp Cell Res. 2004; 297:68–81.

12. Karlsson T, Virtanen M, Sirsjö A, Rollman O, Vahlquist A, Törmä H. Topical retinoic acid alters the expression of cellular retinoic acid-binding protein-I and cellular retinoic acid-binding protein-II in non-lesional but not lesional psoriatic skin. Exp Dermatol. 2002; 11:143–152.

13. Domashenko A, Gupta S, Cotsarelis G. Efficient delivery of transgenes to human hair follicle progenitor cells using topical lipoplex. Nat Biotechnol. 2000; 18:420–423.

14. Park KM, Kang HC, Cho JK, Chung IJ, Cho SH, Bae YH, et al. All-trans-retinoic acid (ATRA)-grafted polymeric gene carriers for nuclear translocation and cell growth control. Biomaterials. 2009; 30:2642–2652.

15. Xia YP, Li B, Hylton D, Detmar M, Yancopoulos GD, Rudge JS. Transgenic delivery of VEGF to mouse skin leads to an inflammatory condition resembling human psoriasis. Blood. 2003; 102:161–168.

16. Baker BS, Brent L, Valdimarsson H, Powles AV, al-Imara L, Walker M, et al. Is epidermal cell proliferation in psoriatic skin grafts on nude mice driven by T-cell derived cytokines? Br J Dermatol. 1992; 126:105–110.

17. Schön MP, Krahn T, Schön M, Rodriguez ML, Antonicek H, Schultz JE, et al. Efomycine M, a new specific inhibitor of selectin, impairs leukocyte adhesion and alleviates cutaneous inflammation. Nat Med. 2002; 8:366–372.

18. Grailer JJ, Kodera M, Steeber DA. L-selectin: role in regulating homeostasis and cutaneous inflammation. J Dermatol Sci. 2009; 56:141–147.

19. Nelson AA, Pearce DJ, Fleischer AB, Balkrishnan R, Feldman SR. New treatments for psoriasis: which biologic is best? J Dermatolog Treat. 2006; 17:96–107.

20. Pariser DM, Bagel J, Gelfand JM, Korman NJ, Ritchlin CT, Strober BE, et al. National Psoriasis Foundation. National Psoriasis Foundation clinical consensus on disease severity. Arch Dermatol. 2007; 143:239–242.

21. Liu Y, Helms C, Liao W, Zaba LC, Duan S, Gardner J, et al. A genome-wide association study of psoriasis and psoriatic arthritis identifies new disease loci. PLoS Genet. 2008; 4:e1000041.

22. Gonze D, Halloy J, Leloup JC, Goldbeter A. Stochastic models for circadian rhythms: effect of molecular noise on periodic and chaotic behaviour. C R Biol. 2003; 326:189–203.

23. Catalina MD, Estess P, Siegelman MH. Selective requirements for leukocyte adhesion molecules in models of acute and chronic cutaneous inflammation: participation of E- and P- but not L-selectin. Blood. 1999; 93:580–589.

24. Sigmundsdottir H, Gudjonsson JE, Valdimarsson H. The effects of ultraviolet B treatment on the expression of adhesion molecules by circulating T lymphocytes in psoriasis. Br J Dermatol. 2003; 148:996–1000.

25. Eşrefoğlu M, Gül M, Seyhan M. Ultrastructural findings and tumor necrosis factor-alpha and intercellular adhesion molecule-1 expression in psoriasis patients before and after oral cyclosporin A therapy. Ultrastruct Pathol. 2006; 30:95–102.

26. Creamer D, Allen MH, Sousa A, Poston R, Barker JN. Localization of endothelial proliferation and microvascular expansion in active plaque psoriasis. Br J Dermatol. 1997; 136:859–865.

27. Heidenreich R, Röcken M, Ghoreschi K. Angiogenesis drives psoriasis pathogenesis. Int J Exp Pathol. 2009; 90:232–248.

28. Bovenschen HJ, Otero ME, Langewouters AM, van Vlijmen-Willems IM, van Rens DW, Seyger MM, et al. Oral retinoic acid metabolism blocking agent Rambazole for plaque psoriasis: an immunohistochemical study. Br J Dermatol. 2007; 156:263–270.

29. Geria AN, Scheinfeld NS. Talarozole, a selective inhibitor of P450-mediated all-trans retinoic acid for the treatment of psoriasis and acne. Curr Opin Investig Drugs. 2008; 9:1228–1237.
30. Mrass P, Rendl M, Mildner M, Gruber F, Lengauer B, Ballaun C, et al. Retinoic acid increases the expression of p53 and proapoptotic caspases and sensitizes keratinocytes to apoptosis: a possible explanation for tumor preventive action of retinoids. Cancer Res. 2004; 64:6542–6548.

31. Colucci M, Maione F, Bonito MC, Piscopo A, Di Giannuario A, Pieretti S. New insights of dimethyl sulphoxide effects (DMSO) on experimental in vivo models of nociception and inflammation. Pharmacol Res. 2008; 57:419–425.

32. Li J, Chen X, Liu Z, Yue Q, Liu H. Expression of Th17 cytokines in skin lesions of patients with psoriasis. J Huazhong Univ Sci Technolog Med Sci. 2007; 27:330–332.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download