Abstract
Sweet's syndrome (SS), or acute febrile neutrophilic dermatosis, is marked by fever, leukocytosis, and painful erythematous papules/plaques resulting from neutrophil migration and accumulation in the dermis. This condition has been associated with underlying hematologic as well as solid malignancies. We describe a unique case of SS in a patient with metastatic papillary follicular thyroid carcinoma and group A streptococcal pharyngitis. The distribution of the patient's SS rash was similar to the rash of neutrophilic dermatosis (pustular vasculitis) of the dorsal hands.
Sweet's syndrome (SS), also known as acute febrile neutrophilic dermatosis, is characterized by the occurrence of painful erythematous skin lesions resulting from neutrophil migration to and infiltration of the upper dermis accompanied by systemic symptoms such as fever1. First described in a series of eight women in 1964 by Robert Sweet, this condition has since been reported in both genders and in all age groups, and the youngest case was a 5-day-old neonate2.
Three distinct categories have been classified within this syndrome such as classical, drug-induced, and malignancy-associated. Classical or idiopathic SS is more commonly seen in women and most frequently occurs following an upper respiratory tract infection, although other types of infections have been described. Drug-induced SS is associated with a wide variety of medications, and is particularly seen following the administration of granulocyte-colony stimulating factor (G-CSF)3. Malignancy-associated SS, which has been reported equally in both genders, was first described in testicular carcinoma, but is now most commonly associated with acute myelogenous leukemia. Some cases of malignancy-associated SS have been reported with various solid tumors such as genitourinary, gastrointestinal, and breast malignancies4,5. One clinical variant of SS is neutrophilic dermatosis (pustular vasculitis) of the dorsal hands (NDDH), in which the cutaneous lesions of SS are present while the systemic symptoms are relatively mild6,7.
We describe a unique case of SS in a patient with metastatic papillary follicular thyroid carcinoma (PFTC) in the context of group A streptococcal pharyngitis.
A 79-year-old female presented with a sore throat and non-productive cough for 10 days, a rash for 7 days, and fever for 4 days. The rash was painful and initially began on the palms of both hands with subsequent proximal spread to the torso. At the same time, the patient developed a similar rash on the plantar aspects of both feet with progression to the lower legs. The patient also reported a rash within her mouth that she noticed on the day of presentation.
The patient's medical history was significant for PFTC (diagnosed 2 months prior to admission) with metastases to bilateral lungs, ribs, and clavicles. She underwent a total thyroidectomy 1 month prior to admission and has since been receiving radioiodine therapy. Her family history was significant for multiple first-degree family members with thyroid carcinoma of unknown pathology.
On physical exam, the vital signs were remarkable for a fever of 100.6°F. The remainder of the exam was significant for the rash that manifested as distinct and intact tender pustules of varying (5~7 mm, diameter) sizes as well as several erythematous halos with surrounding edema. These findings were present on the dorsal and palmar surfaces of the hands (Fig. 1) and extended to the mid-radial/ulnar region. The rash was also present on the abdomen, bilateral heels, and on the mucosal surface superior to the lower labial frenum.
Initial laboratory results revealed leukocytosis (17,100 cells/mm3) and neutrophilia (80%). A Group A streptococcal throat culture and anti-streptolysin O (ASLO) titers were positive with an ASLO of 894 IU/ml, and an erythrocyte sedimentation rate (ESR) of 100. Anti-neutrophil cytoplasmic antibody, anti-nuclear antibody, varicella- zoster direct fluorescent antigen, a syphilis screen, and blood/viral/wound/urine cultures were all negative. In light of the patient's painful rash on the hands, an X-ray of the left hand was performed and demonstrated osteoarthritic changes in the digits.
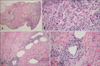
Upon admission, the patient was empirically started on vancomycin for possible methicillin-resistant Staphylococcus aureus infection. The patient remained febrile with an increase in the leukocytosis (29,200 cells/mm3), and ceftriaxone was added on hospital day 4; initial and subsequent blood and urine cultures were repeatedly negative. Given the persistent rash and fever, biopsies of the upper extremity lesions were taken and dermatopathology demonstrated superficial perivascular dermatitis with deeper sections revealing pustular vasculitis consistent with SS (Fig. 2, pathology showing SS). Following the histopathologic diagnosis of SS, prednisone was started with improvement in pain and swelling noted by the patient. Within 1 day of starting prednisone, the patient became afebrile. She received 10 days of steroid therapy, which resulted in resolution of the diffuse pustules on follow-up visits.
SS was originally described as a syndrome, including fever, neutrophilia, and erythematous plaques with neutrophilic infiltration and collection in the dermis1. Since then, the diagnostic criteria of SS have been further delineated into major and minor categories (Table 1)8,9. The major criteria include tender cutaneous lesions and collections of neutrophils without leukocytoclastic vasculitis on histopathology. Although the inflammatory infiltrate is predominately comprised of neutrophils, other cells, including eosinophils, may be found in SS lesions1. SS lesions often begin as pustules but can progress into plaques or target lesions due to the surrounding edema. They routinely occur on the face, neck, and upper extremities, although heterogeneous presentations have been noted, as was the case in our patient. Along with the 2 major criteria, at least 2 of the 4 minor criteria are required for a diagnosis of SS (Table 1). Of the minor criteria, fever is seen in 40~80% of SS cases, and leukocytosis with neutrophilia along with elevated inflammatory markers (e.g., ESR) is common. Our patient demonstrated the major criteria along with all four of the minor criteria.
Response to treatment is one of the minor SS criteria, and prednisone (1 mg/kg/day), which is the standard agent of choice to treat SS, is effective for improving both the cutaneous lesions and systemic manifestations10. Furthermore, potassium iodide is as beneficial as prednisone, and colchicine is also considered first-line therapy11,12. Other medications, including non-steroidal anti-inflammatory drugs, salicylates, clofazimine, cyclosporine, sulfones, and chemotherapeutic agents have shown promising results in patients with SS3. Surgical modalities have also been suggested as treatment options for SS. For example, O'Connor Reina et al.13 described the resolution of recurrent SS in a patient with tonsillar carcinoma following resection of a tumor.
A rare variant of SS is NDDH in which patients present with the two major criteria of SS, but the location of the lesions are predominantly on the hands6,7. Systemic symptoms are generally less severe in most cases of NDDH. In some NDDH reports, leukocytoclastic vasculitis is seen on biopsy; though many feel that this vasculitis is not a primary process, but rather a secondary vascular response to the presence of neutrophils6,7. Our patient's lesions were primarily on her hands; however, she also presented with systemic symptoms and all of the minor criteria for SS.
SS has been associated with distinct pathogenic processes, including infections, drug exposures, and malignancies. Streptococcal pharyngitis is seen in the majority of infections associated with SS, whereas gastrointestinal tract infections due to Salmonella and Yersinia are also frequently reported in conjunction with SS3. Numerous drugs have also been linked to SS, with G-CSF being the most common offending agent14. Hematologic as well as solid tumors co-occur with SS, and acute myelogenous leukemia is the predominant carcinoma in all malignancy-associated SS. Solid tumor associations were associated with SS in a case report of testicular carcinoma described by Shapiro et al.4. Since then, many different organ systems including breast and gastrointestinal carcinomas have been described. To our knowledge, we report the first case of SS in a patient with metastatic PFTC, and our patient's presentation was particularly unique as she had an underlying malignancy along with concurrent pharyngitis.
One of the many proposed theories regarding the immunopathogenesis of SS is based on a potential hypersensitivity reaction to either a bacterial or tumor antigen that leads to neutrophil build-up. This mechanism is supported by neutrophils within many different organ systems. Corticosteroid therapy is extremely effective for resolving SS3. Distinct cytokines and growth factors have also been linked to SS, including interleukin (IL)-1, 3, 6, and 8, granulocyte macrophage-colony stimulating factor, and G-CSF15. Elevated levels of both G-CSF and IL-6 cytokines, which are associated with malignancy, have been documented in patients diagnosed with SS16. Furthermore, Shinojima et al.17 localized G-CSF within a tumor in the setting of SS, and suggested that the release of such cytokines and/or growth factors by specific tumors may lead to neutrophil migration to and infiltration of the upper dermis in patients with SS. There may be a similar association in our case with concurrent follicular thyroid carcinoma and SS. In vitro studies have demonstrated the release of IL-1, IL-6, and IL-8 by follicular thyrocytes, which, in turn, are regulated by IL-1, interferon-gamma, and thyroid stimulating hormone18. As such, malignant follicular cell tumors may release significantly elevated levels of such cytokines, thereby leading to the manifestations of SS.
Understanding of SS has been rapidly evolving, particularly in light of its association with multiple other disease processes. We describe a case of SS in a patient with metastatic PFTC; thus, further adding to the possible immunopathogenic mechanisms underlying SS. A potential theory behind the development of SS may be its link to specific cytokines, and specifically as these cytokines relate to SS and thyroid pathology. Although in vitro studies of follicular thyrocytes and SS may support these ideas, further in vivo studies are clearly necessary.
Figures and Tables
Fig. 2
(A) Inflammatory infiltrate around the vessels (H&E, ×20). (B) Dermal inflammatory infiltrate (mixed populaion of cells) with neutrophils (H&E, ×400). (C) Inflammatory infiltrate within around vessel wall (H&E, ×200). (D) Vessel wall with Inflammatory infiltrate (including neutrophils) and nuclear debris from neutrophils in and around the vessels (H&E, ×400).
References
1. Cohen PR, Kurzrock R. Sweet's syndrome revisited: a review of disease concepts. Int J Dermatol. 2003. 42:761–778.

2. Barr KL, O' Connell F, Wesson S, Vincek V. Nonbullous neutrophilic dermatosis: Sweet's syndrome, neonatal lupus erythematosus, or both? Mod Rheumatol. 2009. 19:212–215.

3. Cohen PR. Sweet's syndrome--a comprehensive review of an acute febrile neutrophilic dermatosis. Orphanet J Rare Dis. 2007. 2:34.
4. Shapiro L, Baraf CS, Richheimer LL. Sweet's syndrome (acute febrile neutrophilic dermatosis). Report of a case. Arch Dermatol. 1971. 103:81–84.

5. Chan HL, Lee YS, Kuo TT. Sweet's syndrome: clinicopathologic study of eleven cases. Int J Dermatol. 1994. 33:425–432.

6. DiCaudo DJ, Connolly SM. Neutrophilic dermatosis (pustular vasculitis) of the dorsal hands: a report of 7 cases and review of the literature. Arch Dermatol. 2002. 138:361–365.
7. Byun JW, Hong WK, Song HJ, Han SH, Lee HS, Choi GS, et al. A case of neutrophilic dermatosis of the dorsal hands with concomitant involvement of the lips. Ann Dermatol. 2010. 22:106–109.

8. Su WP, Liu HN. Diagnostic criteria for Sweet's syndrome. Cutis. 1986. 37:167–174.
9. von den Driesch P. Sweet's syndrome (acute febrile neutrophilic dermatosis). J Am Acad Dermatol. 1994. 31:535–556.

10. Burrall B. Sweet's syndrome (acute febrile neutrophilic dermatosis). Dermatol Online J. 1999. 5:8.
11. Horio T, Imamura S, Danno K, Furukawa F, Ofuji S. Treatment of acute febrile neutrophilic dermatosis (Sweet's Syndrome) with potassium iodide. Dermatologica. 1980. 160:341–347.

12. Suehisa S, Tagami H. Treatment of acute febrile neutrophilic dermatosis (Sweet's syndrome) with colchicine. Br J Dermatol. 1981. 105:483.

13. O'Connor Reina C, Garcia Iriarte MT, Rodriguez Diaz A, Gomez Angel D, Garcia Monge E, Sanchez Conejo-Mir J. Tonsil cancer and Sweet's syndrome. Otolaryngol Head Neck Surg. 1998. 119:709–710.
15. Buck T, González LM, Lambert WC, Schwartz RA. Sweet's syndrome with hematologic disorders: a review and reappraisal. Int J Dermatol. 2008. 47:775–782.

16. Reuss-Borst MA, Pawelec G, Saal JG, Horny HP, Müller CA, Waller HD. Sweet's syndrome associated with myelodysplasia: possible role of cytokines in the pathogenesis of the disease. Br J Haematol. 1993. 84:356–358.





 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download