Abstract
Background
The skin of atopic dermatitis (AD) patients has a high susceptibility to Staphylococcus aureus colonization, and the toxins produced by S. aureus may aggravate AD by acting as superantigens.
Objective
The purpose of this study was to evaluate the relationship of the skin barrier function, colonization of S. aureus, and the clinical severity of AD. We also examined the predominant toxin genes produced in Korean AD patients.
Methods
Thirty-nine patients with AD were evaluated for clinical severity and skin barrier function by using Severity Scoring of Atopic Dermatitis (SCORAD) index and transepidermal water loss (TEWL). S. aureus was isolated from the forearm, popliteal fossa, and anterior nares of AD patients (n=39) and age-matched controls (n=40); the toxin genes were analyzed by performing multiplex polymerase chain reaction.
Results
TEWL showed a statistically significant correlation with clinical severity in patients with AD (p<0.05). TEWL was correlated with the number of S. aureus colonization sites and the presence of nasal colonization, but these results were not statistically significant. S. aureus strains were isolated in 64.1% of the 39 AD patients. The SCORAD index and AD severity were strongly correlated with the number of colonization sites. The predominant toxin gene found in AD patients was staphylococcal enterotoxin a (sea) only, which was produced in 52.6% of patients. The toxin genes sea and toxic shock syndrome toxin-1 (tsst-1) were found together in 42.1%, while tsst-1 only was found in 5.3% of the patients.
Conclusion
S. aureus strains were isolated in 64.1% of the 39 AD patients. Skin barrier function, as measured by TEWL, revealed a statistically significant correlation with clinical severity in AD patients. The SCORAD index and severity of AD was strongly correlated with the number of colonization. The most common toxin gene was sea in the Korean AD patients and this gene might have an important role in the pathogenesis of AD.
Atopic dermatitis (AD) is a chronic, relapsing and pruritic skin disease that occurs most commonly during early infancy and childhood. It is a multifactorial disease that is caused by the interaction of various factors, such as genetic background, allergic and immunologic abnormalities, pharmacologic and physiologic factors, environmental factors, and skin barrier defects. Its prevalence has recently been increasing at up to 10 to 20% of children and 1 to 3% of adults in industrialized countries1.
Recent genetic studies have demonstrated that defective skin barrier function is an important factor in the pathogenesis of AD2,3. Several studies have shown that transepidermal water loss (TEWL), which may represent skin barrier function, was increased in AD patients compared to those of the normal control group4,5.
On the other hand, the role of Staphylococcus aureus in the pathogenesis of AD is currently receiving attention. Actually, 90% of the skin of AD patients has shown colonization with S. aureus, and the number of S. aureus colonies in the sample from AD patients was significantly higher than those of normal controls6. Nasal colonization of S. aureus in infants with AD was also associated with the prevalence and clinical severity of AD7.
The theory that the toxin produced by S. aureus exacerbates the disease activity of AD has been widely accepted, yet various rates of detecting toxin genes or commonly related genes have been reported by many researchers.
In the present study, we evaluated TEWL according to the clinical severity of AD, and we also examined the relationship between the clinical severity of AD and the colonization rates of S. aureus on the skin and the anterior nares. We also investigated the common toxin genes that are involved in the pathogenesis of AD in Korea.
From June to August 2009, we acquired patients with AD and normal control group participants through an advertisement. All 39 patients with AD, according to the accepted diagnostic criteria in Korea8, were enrolled and compared with healthy controls of a similar age (n=40). Those patients had no other systemic disease, no recent history of taking antibiotics and immunosuppressants for the month before the study, and no history of invasive infections, such as erysipelas and cellulitis. Voluntary informed consent in written form was obtained from all participants and/or legal guardians, and all the study protocols were reviewed and authorized by the Gil Hospital Institutional Review Board.
The AD severity was assessed by using Severity Scoring of Atopic Dermatitis (SCORAD) index9.
TEWL was measured using a TEWAmeter TM210® (Courage & Khazaka, Cologne, Germany) for the evaluation of the skin barrier function. Before a measurement, the patient was made comfortable with exposing the examination site by sitting in a silent, draft-free, and direct light-free room at normal temperature for 10 minutes. The examination site was the most severe lesion, usually the antecubital area or popliteal fossa, as chosen by the same dermatologist who examined all the subjects. The measurement was performed for 1 minute.
To culture and separate S. aureus, we used transport media (Micromedia, Busan, Korea). The samples were taken from the antecubital area, the popliteal fossa, and the nasal mucosa, using sterile cotton tips. Within 24 hours, the sample was incubated on mannitol salt agar for 48 hours, and a single colony was pure-cultured and used for further experiments.
The identification of S. aureus was confirmed by a coagulase test and DNase test. We also performed multiplex polymerase chain reaction (PCR) to confirm the molecular identification of S. aureus and to check for methicillin-resistant S. aureus. The primers, including nuc and mecA, were provided by Sigma Genosys (Sydney, Australia), and they are shown in Table 1. DNA extraction was performed as follows: a single colony that had been incubated overnight was taken and added to 50 µl of proteinase K (100 mg/L; Sigma Genosys) and 150 µl of TE buffer (1 mM EDTA/10 mM Tris, pH 7.5). This was then incubated for 20 minutes at 37℃ and then at 95℃ for 5 minutes to deactivate the proteinase K. The suspension was centrifuged, and only the upper layer of the centrifuge tube was used for multiplex PCR. Multiplex PCR was performed on a Corbett Research PC-960 air-cooled thermocycler, using 50 µl of reaction mixture that included 1.25 U Amplitaq DNA polymerase (Perkin Elmer, Foster City, CA, USA), 200 µM dNTPs (Phamacia, Sydney, Australia), 20 ρmol primer, 1.5 mM MgCl2, 100 mM Tris-HCl (pH 8.3), 500 mM KCl, and 50 µl of DNA template. The extracted DNA was amplified for 30 cycles, and each cycle consisted of 60 s at 94℃ for denaturation, 30 s at 50℃ for annealing and 90 s at 72℃ for primer extension. Twenty microliter of PCR product was then analyzed by agarose gel electrophoresis (2% agarose prepared in TAE [1 mM EDTA/40 mM Tris acetate, pH 8] buffer). The gel was stained with ethidium bromide and photographed under ultraviolet light.
We performed multiplex PCR, as described by Breuer et al.10, to detect the staphylococcal toxin genes carried by the S. aureus strains: staphylococcal enterotoxins sea, seb, sec, sed, and see and toxic shock syndrome toxin (tsst)-1. All primers were supplied by MWG Biotech (Ebersberg, Germany), and are shown in Table 2. A single colony was taken and suspended in 185 µl TE buffer (20 mM Tris chloride, 2 mM EDTA [pH 8.0]) with 15 µl of recombinant lysostaphin (15 mg/ml; AMBI, New York, NY, USA), and this was incubated at 37℃ for 30 minutes. The sample was subsequently extracted with a QIAamp tissue kit (QIAGEN, Hilden, Germany), according to the manufacturer's recommendations. Multiplex PCR was performed on an automated thermocycler with a hot bonnet (Hybaid, Teddington, UK), using a 50 µl reaction mixture that consisted of 1.25 U of Taq DNA polymerase (Boehringer, Mannheim, Germany), 200 µM of dATP, dCTP and dGTP, 600 µM UTP, 10 mM Tris-HCl (pH 8.9), 50 mM KCl, 3 mM MgCl2, 1.0 U of UNG and 0.5 ρmol of primer. The thermal cycling conditions for multiplex PCR were 30 cycles of denaturation at 95℃ for 1 minute (2 minutes for the first cycle), annealing at 55℃ for 1 minute and polymerization at 72℃ for 2 minutes (5 minutes for the final extension cycle). Ten microliter of the amplified products were analyzed by agarose gel electrophoresis.
The statistical package for the social sciences (SPSS Version 12.0; SPSS Inc., Chicago, IL, USA) was used for all analyses. Chi-square tests were used to compare the S. aureus colonization rates between the AD patients and the normal controls. Kruskall-Wallis test was used to examine the correlation of TEWL, according to the number of colonization sites and the presence of nasal colonization. A p-value of less than 0.05 was defined as statistically significant.
AD patients consisted of 18 males and 21 females (age: 1~40 years, median: 11.8 years). We classified the SCORAD index as follows: less than 25 as mild, 25~49 as moderate, and more than 50 as severe. The number of patients and their features are summarized in Table 3. The normal control group included 40 individuals (16 males and 24 females, age range: 1~20 years, median age: 10 years).
The mean TEWL (g/m2 per hour), according to the severity of AD was 30.95 in mild, 37.00 in moderate, and 67.55 in severe patients. TEWL showed a statistically significant correlation with the clinical severity of AD (Fig. 1, p=0.002).
Of the 39 patients, 25 patients (64.1%) had S. aureus on one or more sites of the skin. The mean colonization rates for AD patients were 25.6% on the antecubital area, 35.8% on the popliteal fossa, and 46.1% on the anterior nares. The overall mean colonization rate for the normal controls was 20%, and the anterior nares colonization rates of the normal controls were 17.5%. The results of the normal control subjects were lower than those of the AD patients, but the differences were not statistically significant (Table 4). Sixty-five percent of patients (13/20), who had S. aureus isolated from the anterior nares, also had S. aureus on the skin. Of 8 patients with S. aureus isolated from both the skin and anterior nares, 6 patients had the same toxins.
For the relationship of TEWL and the number of colonization sites, we found that the greater the number of colonization sites, the higher the TEWL (Fig. 2). However, this was not statistically significant (p>0.05).
For the relationship of TEWL and the colonization of the anterior nares, positive colonization of the anterior nares showed higher TEWL than the negative colonization in each of the groups (Fig. 3). However, these were not statistically significant (p>0.05).
For the relationship of colonization of S. aureus and the SCORAD index, we found that the higher SCORAD index group had more colonization of S. aureus. However, this was not statistically significant (p>0.05, Table 5).
S. aureus was isolated from 19 patients, and there were 32 total samples from the skin or the anterior nares. Among them, nuc was identified in all, except one sample, and mecA was not produced. Of the 19 patients with S. aureus, the predominant toxin gene was sea only in 10 patients, and the others were in co-existence with sea and tsst-1 in 8 patients and tsst-1 only in 1 patient (Table 6).
Of the 8 patients who had S. aureus on both the skin and nasal mucosa, 6 patients had the same toxin genes identified on both areas.
The clinical expression of AD is the result of the complex interaction of environmental factors and skin barrier dysfunction, susceptibility genes, and immunologic abnormalities. In recent years, an impaired skin barrier function has been receiving attention as one of the important factors in the pathogenesis of AD. Normally, the skin barrier has roles as a permeability barrier, which guards against excessive water loss, and an immunologic barrier, which prevents the entry of harmful substances from the environment such as irritants, allergens, and micro-organisms9. However, a disturbed skin barrier causes the entry of various bacteria and viruses and hypersensitivity reactions, as well as abnormal immunologic reactions against micro-organisms and secondary infection6,9. More than 90% of AD patients have S. aureus as the most common cutaneous infection. In contrast, S. aureus can be found on the skin of only 5% of healthy individuals9,10. The increased level of S. aureus on AD patients is likely the result of combined processes, which includes altered lipid composition within the stratum corneum, exposed extracellular matrix adhesins, a defective immune response, bacterial superantigens, and increased specific immunoglobulin E production9.
In our study, TEWL was significantly increased according to the clinical severity of AD. We also observed that TEWL was increased with an increased number of colonization sites in each of the clinical severity group, but this was not statistically significant. Through theses results, we can suggest that S. aureus colonization and damage of skin barrier function may be the important factors for the development and aggravation of AD.
Breuer et al.11 cultured bacterial samples from the skin and nasal mucosa of adult AD patients, and they isolated S. aureus from 94% of AD patients. Of these patients, S. aureus was isolated on both the skin and nasal mucosa in 77.3%; however, 10.6% and 6.1% of AD patients had S. aureus isolated on the skin and nasal mucosa only, respectively. These results led to the hypothesis that the nose may be an important reservoir of S. aureus strains that are spread over the surface of the skin by autotransmission. They performed toxin gene analyses of S. aureus strains, isolated from the skin and anterior nares of 10 patients, and found that the nasal and cutaneous strains produced the same toxins in most cases, results which support this hypothesis.
In our results, S. aureus was isolated from 64.1% of AD patients (25/39), and this colonization rate was lower than the previously reported high colonization rates in atopic skin11. The reasons for lower colonization compared to other studies are geographical difference and a difference in the composition of the patients' group-- we included AD patients with less severe disease because we gathered the patients through an advertisement and did not include AD patients from the out-patient clinics. However, the colonization rate of S. aureus was strongly related to AD severity and the SCORAD index. We also observed that 65% of the patients (13/20) who had S. aureus isolated from the anterior nares also had S. aureus on the skin. Of the 8 patients with S. aureus isolated from both the skin and anterior nares, 6 had the same toxins; therefore, our results corresponded with those of Breuer et al.11.
Indeed, S. aureus colonization is both a cause and a consequence of allergic skin inflammation in AD. Skin barrier dysfunction promotes increased S. aureus colonization; additionally, the exotoxins secreted by S. aureus are superantigens that are involved in the persistence and exacerbation of AD. Superantigens have several important distinguishing features that are different from the conventional peptide antigens in the pathogenesis of AD. The previously reported staphylococcal superantigens include sea, seb, sec, sed, see, tsst-1, and exfoliative toxins.
The most commonly used methods for assessing S. aureus toxins in the previous reports included reversed passive latex agglutination (RPLA) and multiplex PCR. In one study, ELISA had been used to separate the staphylococcal toxins, and the detection rate was 92%. The detection rates of RPLA had been reported to be 45.5%, 50%, and 67% in 3 different reports12-14. Yagi et al.15 reported an 81% detection rate by PCR, and Kim et al.14 also reported a 100% of detection rate by using a multiplex PCR in their study. Similar to Kim et al.14, we also detected toxins in all the patients who were cultured for S. aureus. Zouharova and Rysanek16 recently reported that multiplex PCR is a more useful tool for the detection of enterotoxin genes than RPLA. These results reflect that toxin gene assays by PCR are becoming more prevalent as a substitute for a latex test14.
There are several reports about the colonization of S. aureus in AD in several countries (Table 7)12-18. Each study reported various colonization rates of S. aureus and various dominant toxins of S. aureus. The difference in the predominant toxin carried by the S. aureus strains isolated from AD patients between the reports could be associated with different geographic regions14. In our study, the colonization rate was 64.1% (25/39), and sea was frequently detected. This result was similar to other studies on Korean people14,17. We did not perform the assay for exfoliative toxins.
In conclusion, we ascertained the correlation between skin barrier function and the clinical severity of AD, and we observed that TEWL was increased with increasing colonization sites and positive colonization of the nasal mucosa in each clinical severity group. S. aureus was isolated from 64.1% of the AD patients (25/39). The rate of colonization of S. aureus was related to the SCORAD index. According to these results, we may assume that colonization of S. aureus and damage of skin barrier function may be important factors in the development and aggravation of AD. The colonization of S. aureus on the nasal mucosa was double that of the antecubital area and popliteal fossa, and 65% of AD patients with nasal colonization also showed colonization on the skin. In addition, the same toxins were detected in 6 of the 8 patients who had S. aureus on both the nasal mucosa and skin. These results imply that the nasal mucosa may be the major reservoir of S. aureus strains. The most common toxin gene was sea in our Korean AD patients, which is similar to that of other studies performed in Korea. Therefore, sea may be an important factor in the pathogenesis of AD in Korean patients, and further investigation should be done on the role of superantigens in the pathogenesis in AD.
Figures and Tables
Fig. 1
Comparison of TEWL according to the SCORAD index (p=0.002). TEWL: transepidermal water loss, SCORAD: Scoring in Atopic Dermatitis.
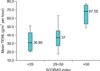
Fig. 2
TEWL according to the number of Staphylococcus aureus colonization sites (p>0.05). TEWL: transepidermal water loss, SCORAD: Severity Scoring of Atopic Dermatitis.
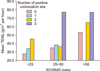
Fig. 3
TEWL according to Staphylococcus aureus colonization of the anterior nares (p>0.05). TEWL: transepidermal water loss, SCORAD: Severity Scoring of Atopic Dermatitis.

Table 2
Base sequences and the predicted sizes of the PCR products for the Staphylococcus aureus exotoxin-specific oligonucleotiede primers

References
1. Laughter D, Istvan JA, Tofte SJ, Hanifin JM. The prevalence of atopic dermatitis in Oregon schoolchildren. J Am Acad Dermatol. 2000. 43:649–655.

2. Weidinger S, Illig T, Baurecht H, Irvine AD, Rodriguez E, Diaz-Lacava A, et al. Loss-of-function variations within the filaggrin gene predispose for atopic dermatitis with allergic sensitizations. J Allergy Clin Immunol. 2006. 118:214–219.

3. Nomura T, Sandilands A, Akiyama M, Liao H, Evans AT, Sakai K, et al. Unique mutations in the filaggrin gene in Japanese patients with ichthyosis vulgaris and atopic dermatitis. J Allergy Clin Immunol. 2007. 119:434–440.

4. Gupta J, Grube E, Ericksen MB, Stevenson MD, Lucky AW, Sheth AP, et al. Intrinsically defective skin barrier function in children with atopic dermatitis correlates with disease severity. J Allergy Clin Immunol. 2008. 121:725–730.

5. Kim DW, Park JY, Na GY, Lee SJ, Lee WJ. Correlation of clinical features and skin barrier function in adolescent and adult patients with atopic dermatitis. Int J Dermatol. 2006. 45:698–701.

6. Sung HC, Jung HD, Park KD, Lee WJ, Lee SJ, Kim DW. A quantitative culture study of Staphylococcus aureus in adolescent and adult patients with atopic dermatitis using the contact-plate sampling technique. Korean J Dermatol. 2007. 45:673–679.
7. Lebon A, Labout JA, Verbrugh HA, Jaddoe VW, Hofman A, van Wamel WJ, et al. Role of Staphylococcus aureus nasal colonization in atopic dermatitis in infants: the Generation R Study. Arch Pediatr Adolesc Med. 2009. 163:745–749.
8. Park YL, Kim HD, Kim KH, Kim MN, Kim JW, Ro YS, et al. Report from ADRG: a study on the diagnostic criteria of Korean atopic dermatitis. Korean J Dermatol. 2006. 44:659–663.
9. Roll A, Cozzio A, Fischer B, Schmid-Grendelmeier P. Microbial colonization and atopic dermatitis. Curr Opin Allergy Clin Immunol. 2004. 4:373–378.

10. Breuer K, Kapp A, Werfel T. Bacterial infections and atopic dermatitis. Allergy. 2001. 56:1034–1041.

11. Breuer K, HAussler S, Kapp A, Werfel T. Staphylococcus aureus: colonizing features and influence of an antibacterial treatment in adults with atopic dermatitis. Br J Dermatol. 2002. 147:55–61.

12. Tomi NS, Kränke B, Aberer E. Staphylococcal toxins in patients with psoriasis, atopic dermatitis, and erythroderma, and in healthy control subjects. J Am Acad Dermatol. 2005. 53:67–72.

13. Bunikowski R, Mielke ME, Skarabis H, Worm M, Anagnostopoulos I, Kolde G, et al. Evidence for a disease-promoting effect of Staphylococcus aureus-derived exotoxins in atopic dermatitis. J Allergy Clin Immunol. 2000. 105:814–819.

14. Kim DW, Park JY, Park KD, Kim TH, Lee WJ, Lee SJ, et al. Are there predominant strains and toxins of Staphylococcus aureus in atopic dermatitis patients? Genotypic characterization and toxin determination of S. aureus isolated in adolescent and adult patients with atopic dermatitis. J Dermatol. 2009. 36:75–81.

15. Yagi S, Wakaki N, Ikeda N, Takagi Y, Uchida H, Kato Y, et al. Presence of staphylococcal exfoliative toxin A in sera of patients with atopic dermatitis. Clin Exp Allergy. 2004. 34:984–993.

16. Zouharova M, Rysanek D. Multiplex PCR and RPLA Identification of Staphylococcus aureus enterotoxigenic strains from bulk tank milk. Zoonoses Public Health. 2008. 55:313–319.

17. Yim YS, Park CW, Lee CH, Song WK. A study on the evaluation of the staphylococcal exotoxins and staphylococcal enterotoxin a-specific ige antibody in childhood atopic dermatitis. Korean J Dermatol. 2002. 40:607–615.




 PDF
PDF ePub
ePub Citation
Citation Print
Print







 XML Download
XML Download