INTRODUCTION
There is a group of diseases characterized by inflammatory nodules which generally located on the lower leg. Many well-defined or debatable entities such as nodular vasculitis, erythema nodosum (EN), erythema induratum (EI), cutaneous periarteritis nodosa, and recurrent thrombophlebitis usually present as inflammatory nodules of the lower legs. Lymphoma in the form of inflammatory nodular lesions has been described1. They have monotonous appearances in common, which often makes a differential diagnosis difficult or impossible on clinical grounds alone. There is the great variation in histopathologic appearance, which depends on the duration of the lesions and sites from specimens are obtained for biopsy. Because of superficial and inadequate biopsy, it is often difficult to assess histopathologic changes in nodular lesions of the legs. Therefore, separating and subclassifying inflammatory nodule lesions of the legs, based on the subtle clinical and histological variation, is difficult. Furthermore, there has been an insufficient consensus on the definitions of many confusing terms such as chronic EN2, EN migrans3, subacute nodular migratory panniculitis4, nodular vasculitis5, and EI of Bazin, which are used to describe recurrent or persistent nodular lesions of the lower legs. In this review, I am going to summarize my clinical experiences of the patients with inflammatory nodules of the leg in Korea.
ERYTHEMA INDURATUM OF BAZIN (EIB)
Etiological relationship with tuberculosis (TB)
In 19th century, Bazin described the subcutaneous indurated plaques appearing on the back of the lower aspect of legs of middle-aged woman who had TB. The link between TB and EI was emphasized later by subsequent investigators. They included EI within the spectrum of tuberculids. The concept of tuberculids was introduced by Darier6 to designate a group of individuals with a previous history of active TB who had a tuberculoid histopathology and presented with an intense reaction to tuberculin. All the bacteriologic methods available at that time for demonstrating a tuberculous origin were negative. The tuberculids were included initially lichen scrofulosorum, papulonecrotic tuberculids, EI, and lupus miliaris disseminatus faciei (LMDF). Later, with the advent of effective treatments against TB, it was also required that these entities have a good response to antituberculous treatment. LMDF was excluded from this group.
However, the tuberculous cause of EIB has been questioned by some authors. They raised arguments against the assumption that EI is manifestation of TB. The reason is as follows. First, many authors have been unable to culture tuberculous bacilli from skin biopsy specimens. Second, active TB occurs with no greater frequency in patients with EI. Third, the degree of tuberculin sensitivity is variable. Fourth, there have been inconsistent responses to antituberculous chemotherapy and good responses to systemic corticosteroids. In 1945, Montgomery et al.5 proposed the concept of nontuberculous nodular vasculitis and described its clinical and histopathological features as different from those of EIB. Still, some authors have proposed keeping the name "EIB" for those cases in which a causative relationship with TB is demonstrated and refer to the remainder of cases as nodular vasculitis. Recently, the detection of Mycobacterium tuberculosis-specific DNA using polymerase chain reaction (PCR) methods was demonstrated in EIB7,8. Currently, however, there seems to be a consensus considering EIB and nodular vasculitis as the same entity9. Like EN, EIB is considered to be a reactive disorder related to several causative factors, one of which may be TB, especially in some geographic areas. In Korea, TB is still the main causative factor for EIB and it is important to prove latent TB infection in the diagnosis of EIB.
Until recently, the only test to identify latent TB infection was the tuberculin skin test. However, an alternative to the tuberculin skin test for diagnosing latent TB has now emerged in the form of a new in vitro test: the interferon-gamma release assay. The principle of the assay is that T cells of individuals previously sensitized with tuberculous antigen will produce interferon-gamma when they re-encounter mycobacterial antigens. Thus a high level of interferon gamma production is presumed to be indicative of tuberculous infection. The QuantiFERON-TB Gold test, which is based on a whole-blood ELISA developed in the late 1980's has been approved for in vitro diagnostics by the Food and Drug Administration (FDA). Mantoux test results were strongly positive in 90% of the patients with EIB that I have experienced. Usefulness of the interferon-gamma assay in the confirmation of latent TB in association with EI is reported by many authors10,11.
Clinical features
In early nineteen-nineties, I have reviewed the clinical records of 32 patients with EIB12. The diagnostic criteria of the EIB were as follows. The patients with chronic inflammatory nodules on the leg; (1) who had evidence of latent M. tuberculosis infection (markedly positive Mantoux test or active infection with M. tuberculosis) (2) who showed clearing of the lesions after antituberculous treatment. The patients included seven men and 25 women. Although young women were common, one half of our patients were over the age of thirty-five, and I realized that EIB is not confined to young age group. The patients had usually recurrent crops of small, tender, erythematous nodules. At times, individual lesions coalesced to form a tender plaque with or without ulcerations. Most lesions were present on the legs, but they also occurred on the forearms, thighs, feet and buttocks. When the lesions were present on the legs, they usually affected both shins and calves. There were some depressed scars and pigmentations from previous active lesions. Skin lesions were concentrated on the lower one third of the lower leg. I believe the following clinical features are helpful to distinguish EIB from other inflammatory nodules of the leg; (1) a chronic recurrent course (2) skin lesions that involve not only shins but also calves; (3) skin nodules heal with ulcerations or depressed scars; (4) skin lesions are concentrated on the lower one third of the lower legs.
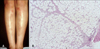
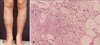
Nowadays, EIB is not common in Korea. And the clinical manifestations of newly found cases are somewhat different from the previous patients. In some patient, only one erythematous nodule on the leg was found (Fig. 1A). I have also experienced the patient whose skin lesions affected mainly the both shins (Fig. 1B). Therefore, it is difficult to make a diagnosis of EIB on the basis of clinical features alone.
Histopathology
Of the biopsy specimens taken from 32 patients of EIB I have studied, 31 were available for review. The histopathologic changes were usually limited to the subcutis and reticular dermis. The subcutis showed lobular or septolobular panniculitis with varying combinations of primary vasculitis, granulomatous inflammation, and septal fibrosis. Vasculitis was present in 87% of biopsy specimens I have studied. Although vasculitis described as a characteristic histopathologic feature of EIB, the nature, size, and location of involved vessels are not established. Segura et al.13 reported that 90.9% of their cases showed some type of venous vasculitis, whereas arteries and arterioles were only involved in 18.8% of the cases, and all the cases with arterial vasculitis, some type of venous vasculitis was concomitantly identified. Thrombosis and occlusion of the vessels are often associated with coagulation necrosis (Fig. 1C). Granulomatous inflammations occur in the majority of EIB skin biopsies14. Epithelioid cells and multinucleate giant cells are occasionally in a tubercle arrangement.
In summary, EIB is a lobular pannicultis that shows granulomatous inflammation with focal necrosis, vasculitis, and septal fibrosis in varying combinations. The histologic pattern greatly depends on the time that the biopsy was conducted during the disease evolution.
Treatment
We usually make a diagnosis of EIB on the basis of clinical feature and a positive interferon gamma release assay supplemented by histopathologic findings. The diagnosis can be confirmed by a good response to antituberculous treatment. I usually recommend a full course of 9 months (with isoniazid, rifampin and ethambutol) of antituberculous treatment. In the patient with ulcerative skin lesions, prednisolone can be combined to alleviate symptoms in the beginning.
ERYTHEMA NODOSUM (EN)
EN is characterized by the sudden onset of symmetric, painful, erythematous, nonulcerating nodules primarily affecting the lower extremities. They mostly occur on the tibial surface of the legs. However, there has been insufficient consensus on the definitions of chronic EN. I emphasized course of disease. If an attack is the first episode, duration should not exceed eight weeks. If the disease recurs or persists longer than this, association with the usual causes of classic EN must be recognized. A case which is attributable to chronic EN, I added to this category only if the effectiveness of potassium iodide was definite.
Pathogenesis
The etiology and pathogenesis of EN are unknown. There are many known associations. It is probable that EN represents a hypersensitivity reaction that may occur in response to a number of antigenic stimuli. In western country, streptococcal infection and sarcoidosis are the most common cause of EN15. Rheumatological and autoimmune diseases, inflammatory bowel diseases, medications, pregnancy and malignancies also have been cited as potential etiological agents. However, it is not easy to demonstrate the etiology of EN. The cause of EN could be suspected in only 45% of the cases15. In Korea, contraception with oral pills is less common and the incidence of sarcoidosis is lower than that of western countries. So, classic EN is infrequently encountered in Korea. EN is a rare but well known complication of the combination oral contraceptives, where estrogen dose higher than the normal physiologic level. Interestingly, I have experienced cases that EN developed in the course of estrogen replacement therapy16. The dosage of estrogen in estrogen replacement therapy is equivalent to the physiologic level.
Clinical features
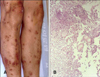
EN affects predominantly woman of young age. EN is manifest by the sudden onset of symmetric, smooth and shiny, erythematous nodules affecting the shins (Fig. 2A). The lesions are typically bilateral and symmetrical, ranging between 1 and 5 cm in diameter. The nodules may coalesce to form plaques. After several days, the erythematous nodules often flatten and become ecchymotic in color. Lesions of EN never ulcerate. Lesions erupt and are usually present for 3~6 weeks. In addition to cutaneous manifestations of EN, constitutional signs and symptoms may also be present. I have experienced a patient with Crohn's disease who had EN. This patient showed only one inflammatory nodule on the leg. When we see the patient with EN, we should keep in mind that typical clinical feature is not always found.
Histopathology
EN represents the prototype of septal panniculitis (Fig. 2B). An inflammatory infiltrate primarily involves the connective tissue septa between fat lobules of the panniculus. Early lesions of EN demonstrate septal edema and a lymphohistiocytic infiltrate, with an admixture of neutrophils and eosinophils. Miescher's radial nodules, which are clusters of macrophages around small vessels or a slit-like space are often referred to as characteristic findings. But I have seen this histologic finding in only one case. Although EN characteristically affects the septal component of the panniculitis, it should be noted that there is infrequently involvement of the lobule. Older lesions of EN demonstrate widened septae with peripheral fibrosis and, and inflammation extending into peripheral lesions of fat lobules.
ERYTHEMA NODOSUM (EN)-LIKE LESION IN BEHCET'S DISEASE
It is not easy to differentiate the nodular lesions accompanying Behcet's disease from classic EN clinically. In most textbook, Behcet's disease is described as one of the causes of EN. Korea is thought to be one of the most prevalent area of Behcet's disease and EN-like lesions have been reported more frequently. Here, I mentioned the nodular skin eruptions in the patients with Behcet's disease separately.
Clinical features
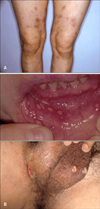
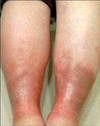
The clinical features of EN-like lesions in the patients with Behcet's disease are similar to the EN of other causes. But some patient show different clinical features of classic EN. Erythematous nodules can also be seen on the arms, thighs, neck and buttocks. There are slightly elevated erythematous plaques, with subcutaneous induration and tenderness. In order to diagnose EN-like lesion of Behcet's disease, it is also important to know the clinical information such as the history of oral and/or genital ulcers (Fig. 3B).
Histopathology
EN-like lesions of Behcet's disease are characterized by panniculitis, usually lobular or mixed septal and lobular in pattern, with variable number of neutrophils, lymphocytes, and histiocytes. Some lesions show the similar histologic features to those of classic EN. Chun et al.17 reported that there is a spectrum of histopathologic change of EN-like lesions in Behcet's syndrome, similar to that of EN secondary to other systemic disorders. However, in contrast to classic EN, vasculitis is noted in most EN-like lesions in Behcet's disease18. Because vasculitis in the EN-like lesion in Behcet's disease is extensive and not limited to areas of severe inflammation, it is believed to be a primary vasculitis. Kim and LeBoit18 suggest that vasculitis is a bascic process in EN-like lesions in Behcet's disease.
Treatment
I have observed the considerable therapeutic effect of colchicines on nodular lesions of Behcet's disease. Potassium iodide has been reported to be unsatisfactory in the treatment of cutaneous lesion of Behcet's disease19.
SUPERFICIAL THROMBOPHLEBITIS
Etiology
Superficial thrombophlebitis can develop in the patients with primary hypercoagulable state. However, these conditions are very rare. Malignancy, pregnancy, oral contraceptive use, infusion of prothrombin complex concentrates, and nephrotic syndrome are secondary hypercoagulable states caused by abnormalities of coagulation and fibrinolysis20. All these disorders may be associated with superficial thrombophlebitis except nephritic syndrome. Superficial thrombophlebitis is of particular interest to dermatologists because the variety of cutaneous disorders such as a Behcet's disease can present in this manner. However, venous insufficiency of the lower extremities is usually the only precipitating factor21.
Clinical features
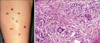
The clinical picture of superficial thrombophlebitis may vary from subtle signs of swelling to a tensely swollen, warm, tender extremity with prominent, distended collateral veins. The following clinical presentation can be a help to diagnose superficial thrombophlebitis. Multiple, tender nodules located most often on the lower extremities. The lesions progress to form palpable cords. Nodular lesions are arranged in a linear configuration are believed to be characterisitic of superficial migratory thrombophlebitis (Fig. 4A). It is important to identify conditions that may promote superficial thrombophlebitis because treatment of underlying disorder frequently results in a decreased thrombotic state. The most important question when considering the diagnosis of superficial thrombophlebitis is whether there is deep venous involvement. Patients with isolated superficial disease may have painful induration of the vein with erythema of the overlying skin.
Histopathology
For histologic examination, a deep elliptic biopsy across the vascular structure should be performed. In punch biopsy, the vein may be missed or improperly oriented on sectioning, thereby making histologic interpretation difficult. We can see the affected vein which has a thick wall with the smooth muscle and the lumen is completely occluded by the thrombus (Fig. 4B). An inflammatory infiltrate extends between the muscle bundles within the vessel wall. In early lesions, the infiltrate is composed of many neutrophils, but later it consists mainly of lymphocytes, histiocytes, and a few giant cells. The inflammation is usually restricted to the vein and rarely extends into the subcutaneous tissue.
Treatment
Superficial thrombophlebitis can be associated with a multitude of disease processes. Treatment of the underlying disease process resulting in superficial thrombophlebitis caused by secondary hypercoagulable states will frequently result in decreased thrombotic rate. Treatment of migratory thrombophlebitis is conservative, with application of a supportive stocking on the involved leg21.
CUTANEOUS POLYARTERITIS NODOSA (CPAN)
CPAN is a variant of polyarteritis nodosa that is limited primarily to the skin. Controversy exists over whether or not CPAN simply represents an early or more limited form of polyarteritis nodosa. Although some patients with CPAN may report constitutional symptoms as well as chronic mild involvement of both muscles and nerves, the course of CPAN is typically benign and relapsing22. Although recurrences are typical, remission may occur spontaneously or following steroid therapy.
Etiology
The etiology of CPAN is unknown. CPAN is probably best viewed as an immune complex-mediated disease. Direct immunofluorescence often shows immunoglobulin M and C3 deposits within affected arterial walls. CPAN may reflect underlying disease, infection, or medication use. The most common agent identified is Group A beta hemolytic Streptococcus.
Clinical features
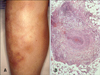
Cutaneous manifestations are the most striking feature of the disease. CPAN usually appears first as livedo reticularis (Fig. 5A), tender subcutaneous nodules, or cutaneous ulcerations. The livedo is fixed and not transient or altered by the temperature of environment. This most commonly occurs on the legs. In fact, the legs are affected 97% of the time, followed by the arms in 33%, and the trunk in 8%22. Small tender nodules that are more easily palpated than visualized are among the most common findings. These nodules with- or without livedo reticularis, are usually the first manifestation of the disease, and the predecessor to ulceration in 50% of the cases. A typical "burst" pattern of irregularly shaped livedo reticularis around an ulcer is highly suggestive of CPAN. A common clinical manifestation is a region of reticular patches, with some tenderness to palpation.
Histopathology
Deep incisional skin biopsy specimen, including the subcutaneous tissue, reveals the presence of leukocytoclastic vasculitis in the small- and medium-sized arteries of the deep dermis or hypodermis. Four stages of histologic findings in CPAN have been described: degenerative, acute inflammatory, granulation tissue, and healed end-stages. The degenerative stage is characterized by degradation of the arterial wall with deposition of fibrinoid material and partial or complete destruction of the internal and external elastic laminae. The acute inflammatory stages involves a mainly neutrophlic infiltrate with eosinophils, around and within arterial wall (Fig. 5B). In the granulation stage, this infiltrate is composed of increased lymphocytes and macrophages, with intimal proliferation and thrombosis of the artery leading to ulceration. The healed end-stage shows a fibroblastic proliferation extending perivascularly.
Treatment
Mild cases, consisting of mainly nodules and livedo, may require only non-steroidal anti-inflammatory drug (NSAID) or colchicine. In addition to their anti-inflammatory properties, NSAIDs are preferred for their analgesic effects. Patients refractory to more conservative treatment and those involving severe pain, ulcerations, necrosis, or extracutaneous symptoms such as myalgias, paresthesias, and arthralgias may require a more aggressive approach. In these instances, corticosteroids are indicated. Prednisolone 30 mg daily or less is often effective, but a dosage of 1 mg/kg/day may be required. Additional medications may be necessary to maintain control of the disease and allow reduction in steroid dosage. Agents that have been used for this purpose include NSAIDs, colchicines, hydroxychlorquine, dapsone, azathioprine, pentoxyphylline, and intravenous immunoglobulin. I have used dapsone for the treatment of many patients with CPAN23. Most patients showed the improvement of the skin lesions. Hemolytic anemia was frequent complication of dapsone treatment.
LYMPHOMA
Peripheral T-cell and natural killer (NK) cell lymphoma primarily involving subcutaneous tissue and mimicking panniculitis have been described24,25. These patients presented with inflammatory nodule on the legs (Fig. 6A) and trunk. Patients often have B symptoms-that is, weight loss, fever and fatigue. Many patients showed evidence of extracutaneous manifestations such as hepatosplenomegaly. Biopsy specimen revealed lobular panniculitis with atypical lymphoid cells. Karyorrhexis is present throughout the subcutis and large phagocytic cells with filled with cellular debris can be seen (Fig. 6B). Now, we can make a more specific diagnosis of malignant lobular panniculits. One is nasal type NK/T cell lymphoma and the other is subcutaneous panniculitic T-cell lymphoma (SPTL). In nasal type NK/T cell lymphoma, prominent angiocentricity and angiodestruction are often accompanied by extensive necrosis26. In SPTL, rimming of individual fat cells by neoplastic T cells is a helpful diagnostic feature27. Histologically, the distinction between nasal type NK/T cell lymphoma involving subcutaneous tissue and SPTL is difficult. Therefore, immunophenotyping and molecular pathological study are useful procedure for diagnosis. In nasal type NK/T cell lymphoma, neoplastic cells usually express CD56 and are characteristically positive for Epstein-Barr virus (EBV). In SPTL, neoplastic cells are usually express CD8 and are negative for EBV. The prognosis of nasal type NK/T cell lymphoma is poor. However, CD8 positive SPTL patients show an indolent clinical course27.
RARE DISEASES
Pancreatic panniculitis
Pancreatic panniculitis results from the saponification of fat as a result of the release of pancreatic enzymes. The most common pancreatic disorders associated with panniculitis are pancreatitis and pancreatic carcinoma. Patients present with multiple, tender nodules, which are erythematous or violaceous in appearance (Fig. 7A). The lower extremities, buttocks, and trunk are most often affected. Occasionally the nodules ulcerate and release a creamy or oil discharge. The main histopathologic feature is a predominantly lobular panniculitis. The pancreatic fat necrosis is characterized by a collection of ghost adipocytes (Fig. 7B). They are anucleate, composed of amorphous granular debris, and often show a rim of eosinophilia28.
Sclerosing panniculitis
Patients present with indurated plaques of wood-like consistency on the lower leg with a stocking distribution is characteristic (Fig. 8). Histopathologic findings in sclerosing panniculitis vary according to the evolution of lesions28. In early stages, there is a sparse inflammatory infiltrate of lymphocytes between the collagen bundles of the septa. In fully developed lesions, the septa appear thickened and fibrotic, resulting in atrophy of the subcutaneous tissue.
Others
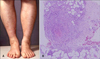
I have experienced a patient with fungal infection who showed the diffuse erythematous swelling of the lower leg. We can find many hyphae in the special stained section of skin biopsy. I have experienced a patient with sparganosis who had a skin nodule in the lower leg. Skin biopsy demonstrated heavy infiltration of eosinophils. Sometimes we can find the patient with foreign body granuloma who present erythematous nodules. Foreign material was injected to make a beautiful shape of the leg when the patient was young. In Korea, subcutaneous nodule is the most frequent specific lesion of sarcoidosis29. The cutaneous lesions in subcutaneous sarcoidosis are characterized by an insidious appearance of multiple, firm, mobile subcutaneous nodules without changes in overlying epidermis (Fig. 9A). We can find characteristic non-caseating granuloma in the biopsy specimen (Fig. 9B).
CONCLUSION
About 15 years ago, I studied one-hundred and thirty six cases featuring inflammatory nodules of the lower leg30. In this series, the most common disease of Korea was EIB. Next was Behcet's disease. Nowadays we cannot find the patients with these diseases frequently. If I survey the patients with inflammatory nodules of the leg again, I can probably get much different results. There are many causes of panniculitis. However, I have not experienced some disease such as alpha1-antitrypsin deficiency panniculitis in Korea.
There are not many tools in the diagnosis of the inflammatory nodules of the leg. Skin biopsy is the major diagnostic aid. However, there are pitfalls of pathologic examination. First, punch biopsy specimens can be interpretable only in 20% of the cases. Incisional skin biopsy is recommended in many cases. Second, we have to keep in mind that there is a variation in histologic appearances depending on the age of the lesion examined31. I believe the final diagnosis is largely based on the response to various treatments and the long-term observation of the natural course of the illness.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download