INTRODUCTION
The incidence of basal cell carcinoma (BCC) has increased because of life style changes that include more outdoor activities1-4. Since there are several histological sub-types of BCC, accurate diagnosis is essential for selection of the proper treatment5. Superficial BCC (sBCC) is one of the sub-types that can preferentially develop on the trunk or limb, while the other subtypes of BCC are mainly found on the head and neck2-4,6,7. Since sBCC can be found on parts of the body that receive only rare sun exposure, it is suggested that sBCC should be considered a rare and separate type of BCC. We report a case involving a large sBCC found on the scalp, a rare location for sBCC.
CASE REPORT
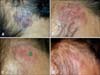
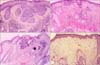
A 71-year-old female presented with a violaceous to blackish crusted plaque, approximately 6.5×6.5 cm, with slightly raised irregular borders on the parietal area of her scalp (Fig. 1A). The lesion, which had developed 4 years earlier, had begun growing rapidly for the previous 4 months prior to the patient's visit to our department. Skin biopsies of the lesion showed the typical histopathological features of sBCC with budding and peripherally palisading basaloid tumor cells in the epidermis (Fig. 2A). There was also partial stromal retraction between the tumor nest and the dermis. Although surgical excision is the usual treatment, this approach was not considered to be the best choice in this case due to the large size of the lesion and subsequent cosmetic problems with closure. Instead, the patient was instructed to apply 5% imiquimod cream once a day, at least five times a week for 12 weeks. The patient was examined every 2 weeks. Skin burning, erythema, and crust were observed as local skin reactions, but the level of this skin reaction was relatively less than expected (Fig. 1B). After the 12 weeks of topical imiquimod, the follow-up biopsy specimen showed remarkable regression of tumor cells, although they were not completely dissipated (Fig. 2B). After continued application of imiquimod for an additional 4 weeks, the biopsy showed remission of the tumor cells (Fig. 2C). Despite the re-treatment, three or four pin-point blackish macules were observed over the next 4 months. Targeted biopsy was performed on three suspicious lesions that showed remnant tumor cells around the excised tissue (Fig. 1C). Therefore, two sessions (1 week apart) of topical photodynamic therapy (PDT) with methyl 5-aminolevulinic acid (Metvix®, Galderma, Oslo, Norway) and red light (Aktilite lamp®, PhotoCure ASA, Oslo, Norway) were recommended to the patient, since the lesion was studded with sBCC. No sign of any clinically suspicious blackish areas (Fig. 1D) or histological tumor cells were found 3 months after the methyl 5-aminolevulinic acid-PDT application (Fig. 2D), and no recurrence was noted during the 2-year follow-up.
DISCUSSION
BCC is the most common cutaneous malignancy in Caucasians. Recently, the incidence of BCC has been increasing among Asians, and the overall number of BCC cases has been growing rapidly1,8,9. The clinical characteristics of BCC appear commonly on the head and neck areas, and the most common subtype of BCC is the histopathologically distinct nodular type1,6-8. sBCC is primarily developed on the trunk, in contrast to the other subtypes2-4,7,9. The different locations between sBCC and the other subtypes of BCC provides indirect favorable evidence for the hypothesis that the tumor biology and histological patterns of BCC subtypes may differ according to their anatomic locations2-4,6. It has been suggested that sBCC is a separate group within the clinical entity of BCC, of which intermittent sun exposure may be an important etiology2-4,6,7. In present case, the lesion developed on the scalp, which is often a sun-covered area, but lesional scalp may not be a fully sun-cover area because of scanty hair due to both pilofollicular recess itself and senile change. McCormack et al.2 proposed that there are some similarities between sBCC and superficial spreading melanoma in terms of the truncal predominance and occurrence in younger patients.
sBCC has been reported extremely rare in the scalp accounting for 0.7~0.8% of all sBCC6. Recently, however, some studies reported an increased incidence of sBCC, especially on the head and neck including the scalp10,11. Arits et al.10 reported a proportional increase in sBCC from 17.6% to 30.7%. They also reported that 14.3% of all sBCC was located in the head and neck region. Because of increasing tendency in sBCC on the head and neck, careful attention is necessary when examining any partial crust or erythematous plaque on the scalp. This thoroughness is necessary to distinguish it from other clinically mimicking skin diseases such as seborrheic dermatitis12 and Bowen disease13.
Treatment of sBCC can be divided into surgical and non-surgical treatment. Although the surgical approach remains the first-line therapy for BCC, it is not easy due to multifocal skip lesions histopathologically, and wide lesions14. Non-surgical treatments include topical chemotherapeutics, biological-immune-response modifiers, retinoids, and photodynamic therapy, which can be used particularly in patients with sBCC and are available for situations in which the invasive treatment is not appropriate5,13,15. In this case, the patient declined any surgical therapy, even though that was the treatment of choice. Since the lesion developed on the scalp, we considered non-surgical treatment a better cosmetic and functional result. The primary treatment was topical application of imiquimod, which acts as an immune response modifier. There are a variety of histological clearances depending on the tumor location during imiquimod treatment15. In this case, the sBCC appeared to be in clinical remission after 16 weeks of imiquimod treatment. However, a few scattered tumor cells were found on the subsequent biopsy. Although skin appendages on the scalp could be better for absorption of lipid or water soluble substances, the thicker scalp skin layer than the frequent anatomic region of sBCC may result in milder local skin reactions, which leads to insufficient histological clearance of tumor cells5,15. It was also postulated that the limited efficacy of imiquimod could be due to uneven topical application, compromised by scalp hair. We experienced complete clinical and histological remission after the application of PDT with methyl 5-aminolevulinic acid twice daily with a 1-week interval.
In conclusion, owing to recently increasing tendency of sBCC in head and neck area, dermatologists should consider this as one of differential diagnosis of a wide scaly lesion on this area.




 PDF
PDF Citation
Citation Print
Print


 XML Download
XML Download