Abstract
Melasma is a common acquired hyperpigmentary skin disorder characterized by light to dark brown macules and patches occurring in the sun-exposed areas of the face. Melasma lesional skin is characterized by epidermal hyperpigmentation through increased melanogenesis in epidermal melanocytes. Some patients have dermal melanin but its amount is not significant and its distribution is very heterogeneous in the whole melasma lesional skin. Melasma is not homogeneous disease and there are personal characteristics of patients with melasma. The pathogenesis of melasma is not fully understood, but several hypotheses have been suggested. Increased vascularity in melasma lesions has suggested the role of increased number of enlarged vessels in the development of melasma. Endogeneous and exogeneous stimuli such as sex hormones and ultraviolet irradiation respectively may stimulate the microenvironment leading to the release of various mediators that cause activation of melanocytes and/or these stimuli may directly activate the melanocytes. Melasma patients may have specialized melanocytes with an intrinsic sensitivity to these stimuli.
Melasma is a common acquired hyperpigmentary disorder characterized by light to dark brown macules and patches occurring in the sun-exposed areas of the face and neck. The major etiological factors include genetic influences, exposure to ultraviolet (UV) radiation, and sex hormones. The results of a recent large global survey in 324 women with melasma suggested that a combination of the known triggers including pregnancy, hormonal birth control, family history and sun-exposure do affect onset of melasma1. The results of this study showed that melasma is not always associated with pregnancy or a history of contraceptive use. In many others, a combination of factors including UV exposure, family history and age are likely to play a role in the development of melasma.
The treatment of melasma remains a challenge. Numerous treatment options, including topical agents, chemical peels and laser treatments have been tried for treating melasma. The triple fixed combination cream containing hydroquinone, retinoic acid and steroid for a duration of 8 weeks is generally recommended to treat melasma with a superior efficacy than hydroquinone monotherapy2. However, the side effect of skin irritation is very common and it is not recommended for long term use. Q-switched pigmentary lasers have not been recommended for treatment of melasma due to the high incidence of postinflammatory hyperpigmentation (PIH) and unpredictable efficacy3. Intense pulsed light has shown some efficacy in the treatment of melasma but the risk of PIH still remains high4. Fractional laser treatments have been reported to improve melasma. However, a recent report showed that the therapeutic efficacy of fractional lasers did not differ from that of conventional therapies5. Recently, a 1064-nm Q-switched Nd:YAG laser with low energy was introduced in the treatment of melasma in Asian patients with considerable results6-8. In this study, patients had minimal downtime without crust formation and significant erythema after laser treatment. However, the possibility of PIH and punctate leucoderma are the major concerns of this treatment9.
To date, no treatment for melasma has demonstrated truly satisfactory results, and so far none of these treatments have been demonstrated to prevent frequent relapses. In this review, we discuss several targets to be considered in treatment of melasma. In part I, we will discuss the findings on melanin and melanocytes in melasma. In part II, we will discuss the current understanding of the pathogenesis of increased pigmentation in melasma, i.e. melanocyte activation caused by variable stimuli.
Traditionally, melasma is classified according to the depth of melanin pigments into epidermal melasma, dermal melasma and mixed type10. As hydroquinone, a tyrosinase inhibitor, targets melanocytes, and not melanophages, it is generally believed that the determination of depth of melanin pigment is helpful in predicting the therapeutic outcome i.e. dermal melasma or mixed type melasma is difficult to treat. Hence, we propose two questions for consideration in treatment of melasma. First question is whether we have a reliable instrument to determine the depth of melanin pigment. The current clinical practice relies on Wood's light examination to determine the depth of melanin pigment. Epidermal melasma exhibits color enhancement of the lesion, whereas dermal melasma exhibits no enhancement of color. However, recent reports have suggested a poor correlation between classification based on Wood's light examination and biopsy skin samples assessed using light microscopy11,12. In a clinical study, Wood's light examination did not help predict response of melasma to treatment13. Therefore, it seems that the clinical use of Wood's light examination is not accurate in determining the level or the depth of melanin pigment. Skin biopsy may be an attractive option in clearly detecting the depth of pigment in melasma. However, our recent study suggested that previous histological classification using single skin biopsy may not be comprehensive14. When we examined the complete melasma lesion using in vivo reflectance confocal microscopy (RCM), it showed that the topographic distribution of melanophages was very heterogeneous between one region of the melasma lesion to another and even within a particular region of the melasma lesion. This finding suggested that a reliable classification should be based on the ratio of epidermal to dermal melanin involving the whole lesional skin.
The second question which may be more important is whether there exists a true dermal type of melasma. Dermal melanophages are commonly found in the sun-exposed skin and the normal facial skin has pigments in the dermis. Kang et al.15 suggested that there is no true dermal type of melasma. Melanophages were present both in melasma lesional and perilesional normal skin in 36% of Korean patients. Grimes et al.11 also reported that all the melasma patients have melanophages in lesional and nonlesional normal skin. The detection of epidermal hyperpigmentation using RCM in all 26 cases in our study of melasma supported the view that there is no true dermal type of melasma14. Therefore, melasma is characterized by epidermal hyperpigmentation with or without melanophages. There was no statistically significant difference in the amount of dermal melanin in lesional skin compared to that of perilesional normal skin, although there was an increase in the lesion11,15. Therefore, it needs to be further studied if this small amount of dermal melanin in the melasma lesional skin does really affect the therapeutic outcome of the treatment.
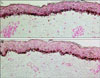
Increased epidermal pigmentation is the hallmark of melasma and must be the main target for melasma treatment (Fig. 1). Histological examinations of melasma have consistently shown that lesional skin is characterized by an increased melanin deposition in the all layers of the epidermis. Indeed, there was an 83% increase in epidermal pigmentation in lesional skin of 56 Korean melasma patients. Another study showed a 61% increase in epidermal pigmentation in lesional skin in all the 11 melasma patients of Fitzpatrick skin type IV to Vl. RCM examination of melasma also consistently detected a 35% increase in epidermal pigmentation in all the 26 melasma patients of skin type II to V. Therefore, the true primary histological target in melasma treatment is the epidermal melanin pigmentation in lesional skin. In other words, although the long wavelength laser targets the dermal melanin, the majority of the beam is absorbed by the increased epidermal pigments in the melasma lesion.
Melanocytes are responsible for increased epidermal pigmentation in melasma. Indeed, melanocytes within the affected skin are larger, intensely stained cells with prominent dendrites and contain more melanosomes than melanocytes in the unaffected skin, suggesting that melanocytes are active in melasma. Our recent transcriptomic study in 12 Korean melasma patients confirmed that higher amounts of epidermal melanin are due to activation of melanogenesis in epidermal melanocytes (Kang et al. unpublished data). There was upregulation of many melanin biosynthesis-related genes as well as melanocyte markers (Tyrosinase, TYRP1, TYRP2 and MITF) in lesional skin compared to perilesional normal skin of melasma patients. It is not clearly established whether there is an increase in the number of melanocytes in the lesional skin. NKI/beteb immunostaining showed an increase in the number of melanocytes, while Mel 5 staining did not confirm this finding11,15. In our study, we found no significant difference in the mean number of MITF-stained melanocytes in lesional skin compared to perilesional normal skin.
It appears that some melasma patients may have hyperactive melanocytes. Interestingly, RCM examination showed that 6 of 25 patients had activated dendritic melanocytes in the melasma lesional skin14. Fontana-Masson staining showed numerous melanized dropping cells in the melasma lesion and NKI/beteb immunostaining confirmed that these cells were melanocytes. This finding needs to be confirmed but it may help in explaining why some patients respond differently to the standard melasma treatment. For example, topical agents may have limitations in inhibiting the melanogenesis in these hyperactive melanocytes. In addition to this finding from our study, there is an interesting paper showing a significant number of melasma patients (11 of 30) having degraded molecules of melanin in the stratum corneum of lesional skin16. The authors suggested that melasma is not a homogeneous disease and this finding may explain the variable success rate of the standard melasma therapy. It may be true that there are personal characteristics of melasma patients.
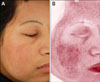
A major clinical feature of melasma is the hyperpigmented patches, but melasma patients have additional distinguishing features like pronounced telangiectatic erythema confined to the melasma lesional skin (Fig. 2). Recent data showed that melasma lesions have more vascularization as compared to the perilesional normal skin14,17. Increased expression of vascular endothelial growth factor (VEGF) in keratinocytes was suggested as the major angiogenic factor for altered vessels in melasma18. It is unclear whether the increased vasculature in melasma is just the result of chronic UV accumulation accompanying epidermal hyperpigmentation. However, recent studies have suggested that a connection between blood vessels and cutaneous pigmentation could exist. Normal human melanocytes in vitro express VEGF receptors and some of these receptors are functional, and this data suggests that VEGF may play a role in melanocyte behavior in skin17. It is also shown that there is an increase in iNOS expression in melasma lesions, providing further perspectives for pathogenesis of increased vasculature in melasma19. The biological role of cutaneous blood vessels in the pathogenesis of melasma remains an interesting topic for future studies.
Therefore the question arises: Whether is it necessary to target blood vessels along with the melanin pigment in treating melasma? Very recently, we performed a prospective, controlled, comparative split-face study evaluating the effects of pulsed dye laser (PDL) therapy in association with triple combination cream in the treatment of melasma. The combination of the Triluma cream and PDL induced a significant decrease in the pigmentation as compared to cream alone. Interestingly, the improvement induced by the combination of PDL and the cream remains significant even after one summer while relapses were observed in the group treated with only the cream. It would be tempting to think that the action of PDL on the vascularization might have played an important role in preventing the relapses. By targeting vascularization and at least some part of elastosis in the melasma lesions we might decrease the stimulation of melanocytes, and thus decrease the relapses.
UV exposure is a major triggering or aggravating factor in the development of melasma. Very recently, it was suggested that visible light also induces hyperpigmentation on type IV-VI skin.20 Recent histological and immunohistochemical studies have shown that melasma skin shows features of prominent solar damaged skin21. Increased solar elastosis in the lesional skin was shown and it was suggested that the process of solar damage may influence the development of hyperpigmentation in the melasma skin. During sun exposure, network of cellular interactions between keratinocytes, fibroblasts and perhaps vasculature and melanocytes may play an important role in epidermal hyperpigmentation in melasma.
The role of neighboring keratinocytes during UV exposure has been suggested in melasma. UV irradiation is known to increase the synthesis of alpha-MSH and ACTH derived from POMC in keratinocytes. These peptides lead to proliferation of melanocytes as well as increase in melanin synthesis via stimulation of tyrosinase activity and TRP-122. When comparing biopsies of lesional and perilesional normal skin in melasma, lesional skin showed a statistically significant increase in expression of alpha-MSH23. Fibroblasts in the dermis may also play a role in the development of melasma. Kang et al.24 reported that the expression of both stem cell factor from fibroblasts and c-kit was significantly increased in the melasma lesional skin. The fibroblast-derived cytokines stimulate the proliferation and melanogenesis of melanocytes in culture25. Therefore, there is a possibility that the dermal inflammation induced by accumulation of UV irradiation may be associated with activation of fibroblasts, which may result in upregulation of SCF in the dermis of melasma patients leading to increased melanogenesis.
Inflammation during UV irradiation may function to stimuli for pigmentation. The results of our transcriptomic study suggested the possible role of prostaglandins in the pathogenesis of melasma (unpublished data). We found that prostaglandin metabolic process was significantly upregulated in melasma compared to perilesional normal skin. Prostaglandins and COX2 are synthesized or upregulated in the skin during UV irradiation and affect melanocytes and pigmentation26. It is clinically well documented that prostaglandins and their analogues can induce epidermal hyperpigmentation27. It can be speculated that the role of steroids in treating melasma may include blocking this process in melasma.
Although melasma is not always associated with pregnancy, melasma occurs in 10~15% of pregnant women and 10~25% of women taking oral contraceptives28,29. It was suggested that melasma which first appears during a pregnancy may be more likely to resolve without treatment afterwards1. Elevated levels of estrogen, progesterone and MSH, especially in the third trimester, have been found in association with melasma30. In vitro studies have shown that cultured human melanocytes express estrogen receptors31. Estradiol increases the level of melanogenic enzyme especially TRP-2 in normal human melanocytes32. Additional supporting evidences showed increased expression of estrogen receptors in the melasma lesional skin33,34. It is speculated that melanocytes in melasma patients may be inherently more sensitive to the increased concentration of estrogens and possibly to the other sex hormones. The effects induced by sex hormones in patients may require additional synergistic events to prime melanocytes, for example, UV light. Very recently, Kim et al.35 detected downregulation of the H19 gene on a microarray analysis of hyperpigmented and normally pigmented skin from melasma patients. These findings suggested that downregulation of H19 and a sufficient dose of estrogen might be involved in the development of melasma.
Melasma is characterized by epidermal hyperpigmentation through increased melanogenesis in melanocytes. Some patients have dermal melanin but its amount is not significant and its distribution is very heterogeneous in the whole melasma lesional skin. It is likely that melasma is not a homogeneous disease and there are personal characteristics of patients with melasma. Melanocytes require the effective modulators of melanin synthesis produced by their neighboring keratinocytes or fibroblasts. The above discussion suggests that endogeneous or exogeneous stimuli may stimulate the microenvironment leading to release of various mediators that cause activation of melanocytes in the development of melasma. It is also possible that these stimuli directly stimulate the melanocytes. Melasma patients may have specialized melanocytes with intrinsic sensitivity to these stimuli.
Melasma continues to be a challenge in treatment and several issues remain to be solved. The significance of melanophages in treating melasma is questionable. It needs to be clearly addressed whether the therapeutic outcome is really dependent on melanophages. It is still unknown whether the changes in dermis of melasma patients are simple epiphenomenon of chronic sun exposure or a primary event stimulating melanocytes. The role of vascularization in the pigmentation processes definitely needs to be studied further. This field of research may provide new therapeutic options such as anti-angiogenic agents in pigmentary disorders. Finally, it may be very difficult to address the intrinsic genetic alteration in melanocytes of melasma patients. Future work is needed to identify the candidate genes that are responsible for the development of melasma.
Figures and Tables
References
1. Ortonne JP, Arellano I, Berneburg M, Cestari T, Chan H, Grimes P, et al. A global survey of the role of ultraviolet radiation and hormonal influences in the development of melasma. J Eur Acad Dermatol Venereol. 2009. 23:1254–1262.

2. Kang HY, Valerio L, Bahadoran P, Ortonne JP. The role of topical retinoids in the treatment of pigmentary disorders: an evidence-based review. Am J Clin Dermatol. 2009. 10:251–260.
3. Ortonne JP, Passeron T. Melanin pigmentary disorders: treatment update. Dermatol Clin. 2005. 23:209–226.

4. Negishi K, Kushikata N, Tezuka Y, Takeuchi K, Miyamoto E, Wakamatsu S. Study of the incidence and nature of "very subtle epidermal melasma" in relation to intense pulsed light treatment. Dermatol Surg. 2004. 30:881–886.

5. Lee HS, Won CH, Lee DH, An JS, Chang HW, Lee JH, et al. Treatment of melasma in Asian skin using a fractional 1,550-nm laser: an open clinical study. Dermatol Surg. 2009. 35:1499–1504.
6. Jeong SY, Chang SE, Bak H, Choi JH, Kim IH. New melasma treatment by collimated low fluence Q-switched Nd:YAG laser. Korean J Dermatol. 2008. 46:1163–1170.
7. Polnikorn N. Treatment of refractory dermal melasma with the MedLite C6 Q-switched Nd:YAG laser: two case reports. J Cosmet Laser Ther. 2008. 10:167–173.

8. Cho SB, Kim JS, Kim MJ. Melasma treatment in Korean women using a 1064-nm Q-switched Nd:YAG laser with low pulse energy. Clin Exp Dermatol. 2009. 34:e847–e850.

9. Kim MJ, Kim JS, Cho SB. Punctate leucoderma after melasma treatment using 1064-nm Q-switched Nd:YAG laser with low pulse energy. J Eur Acad Dermatol Venereol. 2009. 23:960–962.

10. Sanchez NP, Pathak MA, Sato S, Fitzpatrick TB, Sanchez JL, Mihm MC Jr. Melasma: a clinical, light microscopic, ultrastructural, and immunofluorescence study. J Am Acad Dermatol. 1981. 4:698–710.

11. Grimes PE, Yamada N, Bhawan J. Light microscopic, immunohistochemical, and ultrastructural alterations in patients with melasma. Am J Dermatopathol. 2005. 27:96–101.

12. Sarvjot V, Sharma S, Mishra S, Singh A. Melasma: a clinicopathological study of 43 cases. Indian J Pathol Microbiol. 2009. 52:357–359.

13. Lawrence N, Cox SE, Brody HJ. Treatment of melasma with Jessner's solution versus glycolic acid: a comparison of clinical efficacy and evaluation of the predictive ability of Wood's light examination. J Am Acad Dermatol. 1997. 36:589–593.

14. Kang HY, Bahadoran P, Suzuki I, Zugaj D, Khemis A, Passeron T, et al. In vivo reflectance confocal microscopy detects pigmentary changes in melasma at a cellular level resolution. Exp Dermatol. 2010. 19:e228–e233.

15. Kang WH, Yoon KH, Lee ES, Kim J, Lee KB, Yim H, et al. Melasma: histopathological characteristics in 56 Korean patients. Br J Dermatol. 2002. 146:228–237.

16. Moncada B, Sahagún-Sánchez LK, Torres-Alvarez B, Castanedo-Cázares JP, Martínez-Ramírez JD, González FJ. Molecular structure and concentration of melanin in the stratum corneum of patients with melasma. Photodermatol Photoimmunol Photomed. 2009. 25:159–160.

17. Kim EH, Kim YC, Lee ES, Kang HY. The vascular characteristics of melasma. J Dermatol Sci. 2007. 46:111–116.

18. Kim EJ, Park HY, Yaar M, Gilchrest BA. Modulation of vascular endothelial growth factor receptors in melanocytes. Exp Dermatol. 2005. 14:625–633.

19. Jo HY, Kim CK, Suh IB, Ryu SW, Ha KS, Kwon YG, et al. Co-localization of inducible nitric oxide synthase and phosphorylated Akt in the lesional skins of patients with melasma. J Dermatol. 2009. 36:10–16.

20. Mahmoud BH, Ruvolo E, Hexsel CL, Liu Y, Owen MR, Kollias N, et al. Impact of long-wavelength UVA and visible light on melanocompetent skin. J Invest Dermatol. 2010. 130:2092–2097.

21. Hernández-Barrera R, Torres-Alvarez B, Castanedo-Cazares JP, Oros-Ovalle C, Moncada B. Solar elastosis and presence of mast cells as key features in the pathogenesis of melasma. Clin Exp Dermatol. 2008. 33:305–308.

22. Suzuki I, Kato T, Motokawa T, Tomita Y, Nakamura E, Katagiri T. Increase of pro-opiomelanocortin mRNA prior to tyrosinase, tyrosinase-related protein 1, dopachrome tautomerase, Pmel-17/gp100, and P-protein mRNA in human skin after ultraviolet B irradiation. J Invest Dermatol. 2002. 118:73–78.

23. Im S, Kim J, On WY, Kang WH. Increased expression of alpha-melanocyte-stimulating hormone in the lesional skin of melasma. Br J Dermatol. 2002. 146:165–167.
24. Kang HY, Hwang JS, Lee JY, Ahn JH, Kim JY, Lee ES, et al. The dermal stem cell factor and c-kit are overexpressed in melasma. Br J Dermatol. 2006. 154:1094–1099.

25. Imokawa G. Autocrine and paracrine regulation of melanocytes in human skin and in pigmentary disorders. Pigment Cell Res. 2004. 17:96–110.

26. Rhodes LE, Gledhill K, Masoodi M, Haylett AK, Brownrigg M, Thody AJ, et al. The sunburn response in human skin is characterized by sequential eicosanoid profiles that may mediate its early and late phases. FASEB J. 2009. 23:3947–3956.

27. Kapoor R, Phiske MM, Jerajani HR. Evaluation of safety and efficacy of topical prostaglandin E2 in treatment of vitiligo. Br J Dermatol. 2009. 160:861–863.

28. Hexsel D, Rodrigues TC, Dal'Forno T, Zechmeister-Prado D, Lima MM. Melasma and pregnancy in southern Brazil. J Eur Acad Dermatol Venereol. 2009. 23:367–368.

29. Moin A, Jabery Z, Fallah N. Prevalence and awareness of melasma during pregnancy. Int J Dermatol. 2006. 45:285–288.

30. Pérez M, Sánchez JL, Aguiló F. Endocrinologic profile of patients with idiopathic melasma. J Invest Dermatol. 1983. 81:543–545.

31. Jee SH, Lee SY, Chiu HC, Chang CC, Chen TJ. Effects of estrogen and estrogen receptor in normal human melanocytes. Biochem Biophys Res Commun. 1994. 199:1407–1412.

32. Kippenberger S, Loitsch S, Solano F, Bernd A, Kaufmann R. Quantification of tyrosinase, TRP-1, and Trp-2 transcripts in human melanocytes by reverse transcriptase-competitive multiplex PCR--regulation by steroid hormones. J Invest Dermatol. 1998. 110:364–367.

33. Lieberman R, Moy L. Estrogen receptor expression in melasma: results from facial skin of affected patients. J Drugs Dermatol. 2008. 7:463–465.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download