Abstract
Background
Toll-like receptors (TLRs) are expressed by human epidermal keratinocytes and are involved in immune responses.
Objective
The goal of this was to investigate the expression of TLR2 in response to bacterial antigens, cytokines, and different calcium concentrations.
Methods
The expression of TLR2 was assessed after stimulation by lipoteichoic acid (LTA) and streptolysin O (SLO). In addition, TLR2 expression was evaluated after treatment with IFN-γ and TNF-α, and different concentrations of calcium. The expression levels of TLR2 mRNA and protein were studied using RT-PCR and Western blot analysis.
The immediate response by the host to microbial invasion is mediated by the innate immune system which, in turn, alerts the adaptive immune response via cytokines/chemokines or co-stimulatory signals expressed by antigen-presenting cells. The innate immune system senses invading organisms via germ-line encoded, non-clonal pattern recognition receptors that recognize molecular patterns shared by large groups of pathogens. One group of such receptors is the Toll-like receptors (TLRs); these receptors are the mammalian homologue of Toll, a type 1 trans-membrane receptor first described in Drosophila where, in adult flies, is involved in antifungal defence1. The TLR structure includes leucine-rich repeats in the extracellular domain and a cytoplasm domain that shares significant homology with the interleukin 1 receptor (IL-1R) signaling domain, termed the Toll/IL-1R (TIR) region2,3. Signaling pathways triggered via the TIR domain induce the activation of the transcription factor NF-κB4, which translocates from the cytoplasm to the nucleus where it binds to the promoter regions of a wide variety of immune and inflammatory genes.
The TLRs play a crucial role in the induction of antimicrobial responses in different cells. In the past few years, 10 different human TLRs have been identified. TLR2 has broad specificity and is involved in the recognition of yeast5, a wide variety of microbial compounds from Gram-positive bacteria such as peptidoglycan6 and lipoproteins7,8, as well as mycobacterial cell wall components9. TLR2 is required for pro-inflammatory signaling to lipoteichoic acid, lipoproteins and lipoarabinomannan. By contrast, TLR4, together with CD14, recognizes lipopolysaccharides (LPS) on Gram-negative bacteria10,11.
The epidermis, the outermost skin layer, provides the first line of defense against the external environment. The major cell type of the epidermis is the keratinocyte. In addition to forming a physical barrier, keratinocytes have been shown to play an important regulatory role in the cutaneous inflammatory and immune response by producing a variety of cytokines. Keratinocyte-derived cytokines are critical to mobilizing leukocytes from the blood and in signaling other cutaneous cells. In addition to regulating immunological and inflammatory responses, the epidermal keratinocytes contribute to the protective barrier of the epithelium and participate in the host defense by destroying invading microorganisms12,13. Human keratinocytes express TLRs, which enable them to initiate the innate immune response to environmental microbiological challenges. Several reports have demonstrated that human keratinocytes express TLR1, 2, 3, 4, 5, 6, 9 and 102-4,6-8,13,14; however, the expression of some of these compounds was not detected in all studies15.
Among them, TLR2 has been thought to have a more important role in the keratinocytes than the others. This is because the main pathogens in the skin are predominantly gram positive bacteria. The molecules known to affect TLR2 activation include: TNF-α, IL-1β, IL-6, IL-8, IL-10, IL-12, NO, IL-4, IL-5, IL-6, and IL-1316-18. Baker et al.19 reported on the difference in TLR2 expression in psoriatic skin compared to normal skin and suggested that the difference might be due to altered epidermal differentiation or the effects of proinflammatory cytokines. Therefore, TLR2 might be affected by TNF-α and IFN-γ, key cytokines in psoriasis, and the calcium level, which is associated with keratinocyte differentiation.
The aim of this study was to confirm the TLR-inducing effects of bacterial antigens on keratinocytes, and to determine whether the expression of TLR2 is modulated by cytokines and calcium levels.
Normal human keratinocytes were isolated from neonatal foreskin, and were cultured in 154 medium (Cascade Biologics, Portland, OR, USA) with human keratinocyte growth supplement (Cascade Biologics) and 1% penicillin-streptomycin-amphotericin B (10,000 U/ml, 10,000 µg/ml, and 25 µg/ml, respectively; GIBCO BRL., Grand Island, NY, USA) in a humidified atmosphere containing 5% CO2 at 37℃. Isolated keratinocytes were cultivated at 37℃ and 5% CO2 in Epilife (Cascade Biologics). Cells passed three times were used for the experiments and the media was renewed every second day.
Lipoteichoic acid (LTA), derived from Staphylococcus aureus, and streptolysin O (SLO), derived from Streptococcus pyogens, were purchased from sigma. LTA was used at a concentration of 0.1 µg/ml, 1 µg/ml, and 10 µg/ml, and SLO was used at a concentration of 0.1 U/ml, 1 U/ml, and 10 U/ml. The duration of treatment with LTA and SLO was 3 and 24 hrs. Both IFN-γ and TNF-α (R&D System, Minneapolis, MN, USA) were used at a concentration of 10 ng/ml, 50 ng/ml, and 100 ng/ml. Culture medium with and without calcium (Medium 154CF) was purchased from Cascade Biologics. The concentrations of calcium used were: 0.05 mM, 0.1 mM, and 0.2 mM. The duration of treatment with IFN-γ, TNF-α and calcium was 6 and 24 hrs.
Total cellular RNA was purified from cultured cells by the RNA-Bee solution (Tel-test Inc., Friendswood, TX, USA). The cells were lysed with 1.0 ml RNA-Bee solution and extracted by adding 0.1 volume chloroform to the reaction tube. After the mixture was centrifuged at 12,000 g (4℃) for 15 min, the supernatant was transferred to a new 1.5 ml tube. After centrifugation, the cells were removed and an equal volume of isopropanol was added. The samples were precipitated for 15 min at 4℃. After the mixture was centrifuged, the pellet was washed with 800 µl of 75% ethanol and stored in diethylpyrocarbonate (DEPC) treated water. The total RNAs were measured at 260 nm with a spectrophotometer (Ultraspec 2000, Parmacia Biotech, Cambridge, England).
First-strand cDNA synthesis was performed by reverse transcription in a total volume of 20 µl of reaction mixture containing 1 µg of RNA, 1× reaction buffer, 1 mM each dNTP, 5 µM random primers, 20 units RNase inhibitor, and 20 units AMV reverse transcriptase (Promega, Madison, WI, USA). The reaction mixture was incubated at 25℃ for 10 min, 42℃ for 1 hr, and terminated by heating at 95℃ for 5 min. The polymerase chain reaction (PCR) was performed with 2 µl of cDNA in a 50 µl reaction mixture of 1× PCR buffer (10 mM Tris-HCl, pH 9.0, 50 mM KCl, 1.5 mM MgCl2), 200 µM of each dNTP, 20 pmol of sense and antisense primer, and 1 unit of Ampli-Taq Gold DNA polymerase (Roche, Applied Biosystems, Foster City, CA, USA). The primer sequences are shown in Table 1. The amplification conditions were as follows: first denaturation at 95℃ for 14 min, then denaturation at 95℃ for 1 min, annealing at 53℃ to 64℃ for 1 min, and extension at 72℃ for 1 min for 21 to 30 cycles, and final extension at 72℃ for 5 min. Ten microliters of the PCR products were separated by electrophoresis on a 2% agarose gel containing ethidium bromide and visualized by image analysis (Gel Doc 1000 gel documentation system, Bio-Rad, Hercules, CA, USA). The results are reported as the ratio of relative absorbance of TLR2/β-actin analyzed by densitometry. All experiments were performed in triplicate.
The cells were washed in PBS and solubilized in lysis buffer (20 mM Tris (pH 7.5), 150 mM NaCl, and 1 mM phenylmethylsulfonyl) for 15 min on ice and lysates were centrifuged at 13,000×g for 20 min. The protein concentration was determined by the BCA protein assay (Pierce, Rockford, IL, USA). Fifty micrograms of protein was fractionated on 12.5% SDS-PAGE and transferred by electrophoresis to a nitrocellulose membrane. The membranes were blocked with 5% non-fat dry milk in TBS-T buffer (20 mM Tris base, 137 mM NaCl, pH 7.6, 0.1% Tween-20) at room temperature for 1 hr and incubated with primary antibodies (TLR2 and β-actin; Santa Cruz Biotechnology, CA, USA) at a 1:200 dilution with 5% non-fat dry milk in TBS-T at room temperature for 1 hr. After washing with TBS-T for 1 hr, the membranes were reacted with horseradish peroxidase-conjugated anti-rabbit IgG antibody diluted 1: 2,000 with TBS-T at room temperature for 1 hr. After washing the membranes with TBS-T for 1 hr, the membrane was reacted with ECL detection reagents (ECL Western blotting detection reagents and analysis system, Amersham, Aylesbury, UK) according to the manufacture's protocol and autoradiograms were exposed with Konica X-ray film (Konica corporation, Tokyo, Japan). All experiments were performed in triplicate.
After treatment with LTA for 3 hrs, the expression of TLR2 mRNA showed a tendency to increase as the LTA concentration increased. The increase of expression was statistically significant compared to the control, only at the concentration of 10 µg/ml. After 24 hrs, the increase of expression was more significant than at 3 hrs. At all concentrations of LTA, the difference was statistically significant compared to the control (Fig. 1A). The results of the TLR2 protein expression were similar to those of the TLR2 mRNA. After treatment with LTA for 3 hrs and 24 hrs, the TLR2 protein expression levels increased. The difference was statistically significant at 10 µg/ml, after 3 hrs, and 0.1 µg/ml, 1 µg/ml and 10 µg/ml, after 24 hrs (Fig. 1B).
The expression of TLR2 mRNA and protein after treatment with SLO was also analyzed in the same manner. After treatment with SLO for 3 hrs, no significant change was detected. However, after treatment for 24 hrs, the TLR2 mRNA expression showed a significant increase associated with SLO stimulation. At all concentrations of SLO, the TLR2 mRNA expression increased significantly compared to the control (Fig. 2A). The results of the TLR2 protein expression were the same as for the TLR2 mRNA. After 24 hrs of treatment, it was observed that SLO increased the expression of TLR2 protein and the difference was more significant at greater SLO concentrations (Fig. 2B).
These findings provide evidence that both LTA and SLO stimulate the expression of TLR2 in human epidermal keratinocytes. The stimulative effects of both on TLR2 were distinct after 24 hrs of treatment and were proportional to the concentration levels.
The RT-PCR showed that IFN-γ was associated with an increase in the level of TLR2 mRNA after stimulation for 6 hrs. After treatment for 24 hrs, the increase in TLR2 mRNA expression was more significant. These effects were detected even at the lowest concentration of IFN-γ, 10 ng/ml; no difference in the concentrations was observed (Fig. 3A). The results of TLR2 protein expression after IFN-γ treatment were identical to the TLR2 mRNA results (Fig. 3B).
When TNF-α was added to the culture of epidermal keratinocytes, the TLR2 expression increased. This effect was confirmed in both the mRNA and protein levels (Fig. 4). The TLR2-stimulating effect of TNF-α was more pronounced after treatment for 24 hrs compared to 6 hrs; however, the differences were not statistically significant.
These results showed that IFN-γ and TNF-α significantly upregulated the expression of TLR2, regardless of their concentration.
Six hours after the addition of calcium, there was no significant change observed in the mRNA or protein, at all concentrations studied. Only the treatment with 0.2 mM of calcium for 24 hrs, was associated with an increased expression of TLR2 mRNA and protein; however, the difference was not statistically significant (Fig. 5).
The results of this study showed that human keratinocytes constitutively express TLR2 mRNA and protein. Treatment of keratinocytes with LTA and SLO stimulated TLR2 expression, especially after 24 hrs of treatment. IFN-γ and TNF-α both strongly induced TLR2 expression in keratinocytes, regardless of the concentration used. However, there were no significant changes in the expression of TLR2 associated with calcium concentrations.
Several prior studies reported that the expression of TLRs, including TLR2, in human keratinocytes was stimulated by yeast and bacteria. Most of these prior studies investigated the changes of TLR2 and/or TLR4 expression by the LPS of gram negative bacteria20-22; however, some investigated the effects of Staphylococcus aureus itself15 or, the LTA of Staphylococcus aureus23. In this study we demonstrated that both SLO, a pathogenic toxin of Streptococcus pyogenes, and LTA stimulated the expression of TLR2 in human keratinocytes. Streptococcus pyogenes and Staphylococcus aureus are pathogens known to cause cutaneous infectious diseases. In addition, both are the main cause of secondary infection in atopic dermatitis, and contribute to the aggravation of atopic dermatitis. Streptococcus pyogenes plays an important role in the development of psoriasis and SLO has been associated with guttate psoriasis. Staphylococcus aureus has been demonstrated in 20~50% of patients with psoriasis24,25. The results of this study showed that TLR2 expression was upregulated in keratinocytes as the concentration of LTA and SLO increased, although the increase was not directly proportional. These effects were more significant after 24 hrs of stimulation, which is consistent with the results of previous reports21-23. The increase of TLR2 expression by LTA was previously reported to be statistically significant only at concentrations of 10 µg/ml23; however, in the present study, a significant increase of TLR2 expression was observed starting at concentrations as low as 0.1 µg/ml.
TLRs, pathogen-recognition receptors, transduce signals leading to the activation of NF-κB that subsequently drive the immune reaction by the transcriptional induction of several genes coding for cytokines, chemokines, and adhesion molecules26-28. Many investigators have focused mainly on the downstream pathway after activation of the TLRs, such as the induction of cytokines or chemokines; several molecules have been shown to affect the activation of TLRs16-18,29-31. However, in the present study the goal was to identify the factors that regulate or influence the expression of TLR. In this study, it was confirmed that both IFN-γ and TNF-α stimulate TLR2 expression; the effects were strong enough to increase TLR2 at 6 hrs, a relatively short duration of treatment. The stimulation by the two cytokines appears to occur regardless of their concentration. Pivarcsi et al.21 investigated co-treatment with LPS+IFN-γ, and found that 10 ng/ml increased the expression of TLR2 and the stimulation increased as the duration of treatment increased.
Therefore, induction of TLR2 by bacterial pathogens such as LTA and SLO might require a certain time interval for stimulation and this might have some correlation to their concentration. In the case of IFN-γ and TNF-α, both the duration of treatment and the concentration appeared to have little importance.
In addition, we studied whether TLR2 expression was influenced by the calcium concentration. Prior to this study, we hypothesized that TLR2 activation would be influenced by the differentiation of epidermal keratinocytes, and expected that the expression of TLR2 would change with the calcium concentration. However, there was no significant effect of the calcium concentration on TLR2 observed in this study. It is well known that a culture medium with a high calcium concentration induces keratinocytes to differentiate and proliferate. In addition, it has been reported that keratinocytes maintain a proliferative basal cell phenotype when they are cultured in media with a low calcium concentration (0.03 mM), and they are induced to differentiate by increasing the calcium concentration to above 0.1 mM32. A change of TLR2 expression was not detected at the low calcium concentration of 0.05 mM, in which, keratinocytes have a tendency to proliferate, or at the relatively high concentration of 0.2 mM, where keratinocytes tend to differentiate. Kawai et al.33 reported that TLR2 was expressed throughout the entire epidermis. By contrast, another study reported that TLR2 was highly expressed in proliferating basal keratinocytes19. Although it is difficult to interpret the present results, the calcium concentration itself might have little effect on TLR2 expression.
Baker et al.19 demonstrated a difference in TLR2 expression in psoriatic epidermis compared to normal epidermis; TLR2 was more highly expressed in the upper epidermis of psoriatic skin, while, in normal skin, TLR2 was expressed throughout the epidermis, with higher staining in the basal keratinocytes. These findings might explain the different TLR2 expression patterns in psoriatic epidermis. This study confirmed that LTA and SLO induce TLR2 expression, and that proinflammatory cytokines such as IFN-γ and TNF-α stimulate the expression of TLR2. In the presence of psoriasis, epidermal differentiation is altered, and the proliferation and differentiation balance is distorted34. In addition, IFN-γ and TNF-α are the main cytokines involved in the pathogenesis of psoriasis35. These results suggest that TLR2 is upregulated in response to the presence of Gram positive bacteria in the keratin layer24,25. In addition, TLR2 upregulation might not be secondary to altered keratinocyte differentiation but result from the effects of proinflammatory cytokines such as TNF-α and IFN-γ present in the psoriatic lesions.
In conclusion, TLR2 plays a crucial role in the induction of antimicrobial responses in specific immune cells; it was found to be expressed in human keratinocytes even without stimulation. The expression of TLR2 increased with the concentration and duration of treatment with bacterial pathogens, and the increase was amplified by several cytokines, from activated keratinocytes and other cells. Therefore, these results help us understand the expression of TLR2 in cutaneous infectious diseases as well as inflammatory or immune-mediated skin diseases such as atopic dermatitis and psoriasis.
Figures and Tables
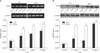 | Fig. 1(A) TLR2 mRNA expression levels at 0.1 µg/ml, 1 µg/ml and 10 µg/ml of LTA compared to the control group for 3 hrs and 24 hrs (n=3, *p<0.05 when compared to the control). (B) TLR2 protein expression levels at 0.1 µg/ml, 1 µg/ml and 10 µg/ml of LTA for 3 hrs and 24 hrs compared to the control group (n=3, *p<0.05 when compared to the control). |
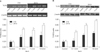 | Fig. 2(A) TLR2 mRNA expression levels at 0.1 U/ml, 1 U/ml and 10 U/ml of SLO compared to the control group at 3 hrs and 24 hrs (n=3, *p<0.05 when compared to the control). (B) TLR2 protein expression levels at 0.1 U/ml, 1 U/ml and 10 U/ml of SLO for 3 hrs and 24 hrs (n=3, *p<0.05 when compared to the control). |
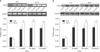 | Fig. 3(A) TLR2 mRNA expression levels after treatment with IFN-γ (10 ng/ml, 50 ng/ml and 100 ng/ml) for 6 hrs and 24 hrs compared to the control group (n=3, *p<0.05 when compared to the control). (B) TLR2 protein expression levels at 10 ng/ml, 50 ng/ml and 100 ng/ml of IFN-γ for 6 hrs and 24 hrs (n=3, *p<0.05 when compared to the control). |
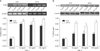 | Fig. 4(A) TLR2 mRNA expression levels after treatment with TNF-α (10 ng/ml, 50 ng/ml and 100 ng/ml) compared to the control group for 6 hrs and 24 hrs (n=3, *p<0.05 when compared to the control). (B) TLR2 protein expression levels at 10 ng/ml, 50 ng/ml and 100 ng/ml of TNF-α for 6 hrs and 24 hrs (n=3, *p<0.05 when compared to the control). |
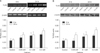 | Fig. 5(A) TLR2 mRNA expression levels according to the calcium concentration (0.05 mM, 0.1 mM, and 0.2 mM) in cultured human keratinocytes at 6 hrs and 24 hrs (n=3). (B) TLR2 protein expression levels according to the calcium concentration (0.05 mM, 0.1 mM, and 0.2 mM) in cultured human keratinocytes at 6 hrs and 24 hrs (n=3). |
References
1. Lemaitre B, Nicolas E, Michaut L, Reichhart JM, Hoffmann JA. The dorsoventral regulatory gene cassette spatzle/Toll/cactus controls the potent antifungal response in Drosophila adults. Cell. 1996. 86:973–983.

2. Rock FL, Hardiman G, Timans JC, Kastelein RA, Bazan JF. A family of human receptors structurally related to Drosophila Toll. Proc Natl Acad Sci U S A. 1998. 95:588–593.
3. Kopp EB, Medzhitov R. The Toll-receptor family and control of innate immunity. Curr Opin Immunol. 1999. 11:13–18.

4. O'Neill LA, Greene C. Signal transduction pathways activated by the IL-1 receptor family: ancient signaling machinery in mammals, insects, and plants. J Leukoc Biol. 1998. 63:650–657.
5. Underhill DM, Ozinsky A, Hajjar AM, Stevens A, Wilson CB, Bassetti M, et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature. 1999. 401:811–815.

6. Yoshimura A, Lien E, Ingalls RR, Tuomanen E, Dziarski R, Golenbock D. Cutting edge: recognition of Gram-positive bacterial cell wall components by the innate immune system occurs via Toll-like receptor 2. J Immunol. 1999. 163:1–5.
7. Lien E, Sellati TJ, Yoshimura A, Flo TH, Rawadi G, Finberg RW, et al. Toll-like receptor 2 functions as a pattern recognition receptor for diverse bacterial products. J Biol Chem. 1999. 274:33419–33425.

8. Brightbill HD, Libraty DH, Krutzik SR, Yang RB, Belisle JT, Bleharski JR, et al. Host defense mechanisms triggered by microbial lipoproteins through toll-like receptors. Science. 1999. 285:732–736.

9. Underhill DM, Ozinsky A, Smith KD, Aderem A. Toll-like receptor-2 mediates mycobacteria-induced proinflammatory signaling in macrophages. Proc Natl Acad Sci U S A. 1999. 96:14459–14463.

10. Poltorak A, He X, Smirnova I, Liu MY, Van Huffel C, Du X, et al. Defective LPS signaling in C3H/HeJ and C57BL/10ScCr mice: mutations in Tlr4 gene. Science. 1998. 282:2085–2088.

11. Qureshi ST, Lariviere L, Leveque G, Clermont S, Moore KJ, Gros P, et al. Endotoxin-tolerant mice have mutations in Toll-like receptor 4 (Tlr4). J Exp Med. 1999. 189:615–625.

12. Jung HC, Eckmann L, Yang SK, Panja A, Fierer J, Morzycka-Wroblewska E, et al. A distinct array of proinflammatory cytokines is expressed in human colon epithelial cells in response to bacterial invasion. J Clin Invest. 1995. 95:55–65.

13. Wang B, Ruiz N, Pentland A, Caparon M. Keratinocyte proinflammatory responses to adherent and nonadherent group A streptococci. Infect Immun. 1997. 65:2119–2126.

14. Lebre MC, van der Aar AM, van Baarsen L, van Capel TM, Schuitemaker JH, Kapsenberg ML, et al. Human keratinocytes express functional Toll-like receptor 3, 4, 5, and 9. J Invest Dermatol. 2007. 127:331–341.

15. Mempel M, Voelcker V, Kollisch G, Plank C, Rad R, Gerhard M, et al. Toll-like receptor expression in human keratinocytes: nuclear factor kappaB controlled gene activation by Staphylococcus aureus is toll-like receptor 2 but not toll-like receptor 4 or platelet activating factor receptor dependent. J Invest Dermatol. 2003. 121:1389–1396.

16. Kang SS, Kauls LS, Gaspari AA. Toll-like receptors: applications to dermatologic disease. J Am Acad Dermatol. 2006. 54:951–983.

17. Thoma-Uszynski S, Stenger S, Takeuchi O, Ochoa MT, Engele M, Sieling PA, et al. Induction of direct antimicrobial activity through mammalian toll-like receptors. Science. 2001. 291:1544–1547.

18. Supajatura V, Ushio H, Nakao A, Akira S, Okumura K, Ra C, et al. Differential responses of mast cell Toll-like receptors 2 and 4 in allergy and innate immunity. J Clin Invest. 2002. 109:1351–1359.

19. Baker BS, Ovigne JM, Powles AV, Corcoran S, Fry L. Normal keratinocytes express Toll-like receptors (TLRs) 1, 2 and 5: modulation of TLR expression in chronic plaque psoriasis. Br J Dermatol. 2003. 148:670–679.

20. Song PI, Park YM, Abraham T, Harten B, Zivony A, Neparidze N, et al. Human keratinocytes express functional CD14 and toll-like receptor 4. J Invest Dermatol. 2002. 119:424–432.

21. Pivarcsi A, Bodai L, Rethi B, Kenderessy-Szabo A, Koreck A, Szell M, et al. Expression and function of Toll-like receptors 2 and 4 in human keratinocytes. Int Immunol. 2003. 15:721–730.

22. Lebre MC, Antons JC, Kalinski P, Schuitemaker JH, van Capel TM, Kapsenberg ML, et al. Double-stranded RNA-exposed human keratinocytes promote Th1 responses by inducing a Type-1 polarized phenotype in dendritic cells: role of keratinocyte-derived tumor necrosis factor alpha, type I interferons, and interleukin-18. J Invest Dermatol. 2003. 120:990–997.
23. Park YM, Kwon HJ, Kang YS, Koo JK, Kim MY, Kim HO, et al. Expression of Toll-like receptor 4 on human keratinocytes by lipoteichoic acid. Korean J Dermatol. 2006. 44:15–21.
25. Leung DY, Harbeck R, Bina P, Reiser RF, Yang E, Norris DA, et al. Presence of IgE antibodies to staphylococcal exotoxins on the skin of patients with atopic dermatitis. Evidence for a new group of allergens. J Clin Invest. 1993. 92:1374–1380.

26. McInturff JE, Modlin RL, Kim J. The role of toll-like receptors in the pathogenesis and treatment of dermatological disease. J Invest Dermatol. 2005. 125:1–8.

27. Akashi-Takamura S, Miyake K. Toll-like receptors (TLRs) and immune disorders. J Infect Chemother. 2006. 12:233–240.

28. Pandey S, Agrawal DK. Immunobiology of Toll-like receptors: emerging trends. Immunol Cell Biol. 2006. 84:333–341.

29. Mizel SB, Honko AN, Moors MA, Smith PS, West AP. Induction of macrophage nitric oxide production by Gram-negative flagellin involves signaling via heteromeric Toll-like receptor 5/Toll-like receptor 4 complexes. J Immunol. 2003. 170:6217–6223.

31. Nagase H, Okugawa S, Ota Y, Yamaguchi M, Tomizawa H, Matsushima K, et al. Expression and function of Toll-like receptors in eosinophils: activation by Toll-like receptor 7 ligand. J Immunol. 2003. 171:3977–3982.

32. Watt FM, Green H. Stratification and terminal differentiation of cultured epidermal cells. Nature. 1982. 295:434–436.

33. Kawai K, Shimura H, Minagawa M, Ito A, Tomiyama K, Ito M. Expression of functional Toll-like receptor 2 on human epidermal keratinocytes. J Dermatol Sci. 2002. 30:185–194.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download