Abstract
Dedifferentiated liposarcoma is a variant of liposarcoma, and this is characterized by the coexistence of well-differentiated liposarcoma with areas of poorly differentiated, non-lipogenic tumor and this is also known to be associated with more aggressive behavior. Dedifferentiated liposarcoma occurs principally in the retroperitoneum or the deep soft tissue of limbs, but it can also occur in subcutaneous locations. We report here on a peculiar case of dedifferentiated liposarcoma that developed in the dermis, which is an exceedingly rare location for this type of tumor. The occurrence of this tumor in the dermis made it easy to surgically remove and monitor for recurrence, and we expect this patient to have a better prognosis than that of a patient with dedifferentiated liposarcoma located in the retroperitoneum or deep soft tissue.
Liposarcoma is the most common soft tissue sarcoma, accounting for 20% of all the soft tissue malignancies in adults1. The anatomic distribution of liposarcoma depends largely on the histologic subtype, but the deep soft tissue of the extremities and the retroperitoneum are the most frequently affected sites. Whereas it is accepted that liposarcoma rarely occurs in the subcutaneous soft tissue, the dermis seems to represent an exceedingly rare site of occurrence2. Five major types of liposarcomas are generally recognized: well-differentiated, myxoid, round cell, pleomorphic and dedifferentiated variants3. Dedifferentiated liposarcoma is defined as a neoplasm with well-differentiated liposarcoma juxtaposed to high-grade, pleomorphic sarcoma, and this known to be associated with more aggressive behavior.
Reports of liposarcoma are rare in the dermatologic field, so only two cases of liposarcoma have been described in the Korean dermatologic literature: one case of myxoid liposarcoma4 and the other of well-differentiated liposarcoma5 and both of these occurred in the deep soft tissue. We report here on a case of dedifferentiated liposarcoma affecting the dermis of the thigh, and we also review the relevant medical literature.
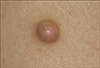
A 40-year-old Korean woman presented to our dermatology clinic with a skin lesion on the right thigh, and this lesion had developed 5 years previously. She noticed a recent increase of its size and the development of tenderness. Her personal and family histories were not remarkable. The physical examination revealed a 9 mm-size dome-shape brownish nodule on the right thigh (Fig. 1).
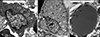
When the incisional biopsy was performed, a poorly demarcated lipogenic cellular proliferation was observed with an infiltrative growth pattern in the dermis (Fig. 2A). Well-differentiated elements were comingled with dedifferentiated elements. The well-differentiated areas contained lipoblasts with several lipid vacuoles of varying size, signet ring cells and mature fat cells (Fig. 2B), whereas the dedifferentiated areas consisted of poorly differentiated, slightly lipogenic or nonlipogenic round cells (Fig. 2C). The nuclei of some of the adipocytes and lipoblasts showed slight pleomorphism and hyperchromatism. Spindle cells with enlarged hyperchromatic atypical nuclei were interspersed in the surrounding myxoid stroma. Immunohistochemically, the round cells were negative for CD34, CD68 and S-100 protein. Alcian blue staining revealed mucin deposits in the surrounding stroma. In the ultrastructural study, some of round cells, which were nonlipogenic under light microscopic examination, contained non-membrane-bound lipid inclusions (Fig. 3A), indicating an adipose origin for the undifferentiated round cells. There were also lipoblasts of varying degrees of differentiation that also showed lipid inclusions of varying sizes and numbers (Fig. 3B, C).
On the basis of the histological findings, a diagnosis of dedifferentiated liposarcoma was made and an additional wide excision was performed. There has been no evidence of recurrence for 18 months on the patient's follow-up.
Liposarcoma is a heterogenous group of malignant mesenchymal neoplasms that exhibit varying degrees of adipocytic differentiation. The anatomic distribution of liposarcoma appears to be partly related to the histologic type: well-differentiated liposarcoma tends to occur in the deep soft tissues of both the limbs and the retroperitoneum; myxoid/round cell and pleomorphic liposarcomas exhibit a prominent predilection for the limbs; and dedifferentiated liposarcoma occurs predominantly in the retroperitoneum. Although any liposarcoma subtype occasionally arises in the subcutaneous tissue, the dermis seems to represent an exceedingly rare site for the occurrence of liposarcoma2. According to Dei Tos et al2, only seven cases (approximately 1%) out of 671 liposarcomas were identified to occur primarily in the dermis. Clinically, they presented as dome-shaped or polypoid lesions that showed exophytic growth.
The pathologic classification of liposarcomas has been undergoing change. The World Health Organization distinguishes the 4 variants as proposed by Enzinger and Winslow, that is, well-differentiated, myxoid, round cell and pleomorphic variants, and a fifth dedifferentiated varient3. On the other hand, the newer classification of liposarcoma into 3 groups (myxoid/round cell, well-differentiated/dedifferentiated and pleomorphic) has been increasingly accepted and this new classification is supported by the cytogenetic and molecular evidence6. Dedifferentiated liposarcomas display the same chromosomal abnormalities that are characteristic of well-differentiated liposarcoma, such as a supernumerary ring and/or giant chromosomes composed of the q13~q15 regions of chromosome 127. Myxoid and round cell tumors are associated with the unique translocation t(12;16)(q13;p11)8 that results in the fusion of 2 genes known as TLS (translocated in liposarcoma) and CHOP91011 or a t(12;13)(q13;q12) that fuses CHOP with a different gene, EWS12.
Dedifferentiation or histologic progression to a high-grade, less-differentiated neoplasm is a recognized complication of several low-grade mesenchymal neoplasms, including well-differentiated chondrosarcoma, chordoma and parosteal osteosarcoma1314. Dedifferentiated liposarcoma is defined as a neoplasm with well-differentiated liposarcoma juxtaposed to high-grade, pleomorphic sarcoma, and the latter usually resembles malignant fibrous histiocytoma or fibrosarcoma. In most cases, dedifferentiation is seen in the original biopsy or excision (de novo or primary dedifferentiation), whereas in a minority of cases it develops late in the course of a previous well-differentiated liposarcoma (secondary dedifferentiation).
The interface between the well-differentiated and dedifferentiated zones was reported to be of three types15. In most cases, an abrupt transition was noted such that the two zones were starkly juxtaposed to one another. Less frequently, there was a gradual transition between the two zones. The least common pattern was that of intricate comingling of the dedifferentiated elements among the well-differentiated elements, and this was the pattern for our case.
The appearance of dedifferentiated foci varies more than has been acknowledged in the past. In the report by Henricks et al15, the most common pattern was that of a high-grade malignant fibrous histiocytoma consisting of atypical spindle cells and pleomorphic cells arranged haphazardly or in short fascicles. In a few tumors, the dedifferentiated area was exclusively low grade, and the dedifferentiated area consisted of bland spindle cells arranged in long or short fascicles. In addition, a number of cases that showed unusual features have also been reported. In a few cases, the dedifferentiated foci consisted of sheets of large rounded cells, which resemble those of our case. Even rhadomyosarcomatous, leiomyosarcomatous, angiosarcomatous or osteosarcomatous elements have been observed in the dedifferentiated components16, which may suggest they originate from primitive mesenchymal cells.
Dedifferentiation generally confers to the tumor a more aggressive biologic potential, yet there is limited data on this. Moreover, some data has suggested variability in the course of dedifferentiated liposarcoma17181920. The prognosis of dedifferentiated liposarcoma is suspected to depend on several factors, such as the location, the extent of dedifferentiation and the histologic grade. Yet several authors have reported that neither the extent of dedifferentiation nor the histologic grade affect the prognosis (i.e., tumors with less dedifferentiation or low-grade tumors did not have a better prognosis than their counterparts)1520. On the other hand, the location has been identified to be an important determinant for the prognosis in that tumors located in the retroperitoneum had a worse prognosis than those located in the accessible soft tissue sites15. In addition, seven cases of liposarcoma that occurred primarily in the dermis, among which four cases of pleomorphic liposarcoma were included, although dedifferentiated liposarcoma was not included, exhibited relatively indolent clinical behavior with no disease-related deaths after performing wide excision of the tumor2.
In conclusion, this is a very peculiar case of dedifferentiated liposarcoma that developed in the dermis, an exceedingly rare location, and the tumor histologically showed intricate comingling of dedifferentiated elements among the well-differentiated elements, which is an uncommon histologic pattern. After performing wide excision, no recurrence was observed for 18 months on the follow-up. We expect a favorable prognosis because the tumor was located in the dermis, and this made it easy to surgically remove the tumor and to monitor for recurrence.
Figures and Tables
Fig. 2
(A) Well-differentiated elements consisting of mature adipocytes and lipoblasts were comingled with dedifferentiated elements composed of poorly differentiated round cells in the myxoid dermis (H&E, ×40). (B) Mature adipocytes and well-differentiated lipoblasts were observed in the well-differentiated areas. Some of these cells showed enlarged hyperchromatic atypical nuclei (H&E, ×400). (C) The dedifferentiated areas consisted of poorly differentiated, slightly lipogenic or nonlipogenic round cells (H&E, ×400).

Fig. 3
(A) A nonlipogenic lipoblast exhibited a small number of non-membrane bound lipid inclusions (EM, ×12,000). (B) A slightly lipogenic lipoblast showed multiple lipid inclusions (EM, ×7,000). (C) The cell with a large lipid vacuole corresponded to a signet ring cell noted on the light microscopic examination (EM, ×2,000).
References
1. Fletcher CDM. Adipocytic tumors. In : Fletcher CDM, editor. Diagnostic histopathology of tumors. 2nd ed. London: Churchill Livingstone;2000. p. 1474–1485.
2. Dei Tos AP, Mentzel T, Fletcher CD. Primary liposarcoma of the skin: a rare neoplasm with unusual high grade features. Am J Dermatopathol. 1998; 20:332–338.

3. Weiss SW. WHO histological typing of soft tissue tumours. 2nd ed. Berlin: Springer-Verlag;1994.
5. Kim WH, Seung NR, Kim CW, Cho HJ, Kim KH, Kim KJ. A case of well-differentiated liposarcoma. Ann Dermatol. 2006; 18:105–108.

7. Fletcher CD, Akerman M, Dal Cin P, de Wever I, Mandahl N, Mertens F, et al. Correlation between clinicopathological features and karyotype in lipomatous tumors. A report of 178 cases from the Chromosomes and Morphology (CHAMP) Collaborative Study Group. Am J Pathol. 1996; 148:623–630.
8. Turc-Carel C, Limon J, Dal Cin P, Rao U, Karakousis C, Sandberg AA. Cytogenetic studies of adipose tissue tumors. II. Recurrent reciprocal translocation t(12;16)(q13;p11) in myxoid liposarcomas. Cancer Genet Cytogenet. 1986; 23:291–299.

9. Crozat A, Aman P, Mandahl N, Ron D. Fusion of CHOP to a novel RNA-binding protein in human myxoid liposarcoma. Nature. 1993; 363:640–644.

10. Rabbitts TH, Forster A, Larson R, Nathan P. Fusion of the dominant negative transcription regulator CHOP with a novel gene FUS by translocation t(12;16) in malignant liposarcoma. Nat Genet. 1993; 4:175–180.

11. Knight JC, Renwick PJ, Dal Cin P, Van den Berghe H, Fletcher CD. Translocation t(12;16)(q13;p11) in myxoid liposarcoma and round cell liposarcoma: molecular and cytogenetic analysis. Cancer Res. 1995; 55:24–27.
12. Dal Cin P, Sciot R, Panagopoulos I, Aman P, Samson I, Mandahl N, et al. Additional evidence of a variant translocation t(12;22) with EWS/CHOP fusion in myxoid liposarcoma: clinicopathological features. J Pathol. 1997; 182:437–441.

13. Enzinger FM, Weiss SW. Liposarcomas. In : Enzinger FM, Weiss SW, editors. Soft tissue tumors. 3rd ed. St. Louis: Mosby;1995. p. 431–466.
14. Meis JM. "Dedifferentiation" in bone and soft-tissue tumors. A histological indicator of tumor progression. Pathol Annu. 1991; 26:37–62.
15. Henricks WH, Chu YC, Goldblum JR, Weiss SW. Dedifferentiated liposarcoma: a clinicopathological analysis of 155 cases with a proposal for an expanded definition of dedifferentiation. Am J Surg Pathol. 1997; 21:271–281.
16. Evans HL, Khurana KK, Kemp BL, Ayala AG. Heterologous elements in the dedifferentiated component of dedifferentiated liposarcoma. Am J Surg Pathol. 1994; 18:1150–1157.

17. Brooks JJ, Connor AM. Atypical lipoma of the extremities and peripheral soft tissues with dedifferentiation: implications for management. Surg Pathol. 1990; 3:169–178.
18. Evans HL. Liposarcomas and atypical lipomatous tumors: a study of 66 cases followed for a minimum of 10 years. Surg Pathol. 1988; 1:41–54.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download