Abstract
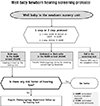
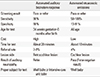
The incidence of bilateral profound hearing loss of newborns is 1 to 2 per 1,000 newborns. It is higher in infants with risk factors for hearing loss. Congenital hearing loss can cause many problems in language, learning, speech development and educational and occupational performance. Most developed countries have conducted the Universal Newborn Hearing Screening (UNHS) with automated otoacoustic emissions (AOAE) or automated auditory brainstem response (AABR). UNHS reduced the average age of identification of permanent hearing loss in infants 6 months or less after birth. This early identification and intervention of hearing loss with amplification and speech therapy optimizes communication during the early critical period of language acquisition and can improve language outcomes in children between 2 and 5 years of age. The aims of this paper are to explain the incidence of newborn hearing loss, the importance of early detection of hearing loss and intervention and newborn hearing screening methods.
Figures and Tables
References
1. Thompson DC, McPhillips H, Davis RL, Lieu TL, Homer CJ, Helfand M. Universal newborn hearing screening: summary of evidence. JAMA. 2001; 286:2000–2010.
2. Norton SJ, Gorga MP, Widen JE, Folsom RC, Sininger Y, Cone-Wesson B, et al. Identification of neonatal hearing impairment: evaluation of transient evoked otoacoustic emission, distortion product otoacoustic emission, and auditory brain stem response test performance. Ear Hear. 2000; 21:508–528.

3. American Academy of Pediatrics. Joint Committee on Infant Hearing. Year 2007 position statement: principles and guidelines for early hearing detection and intervention programs. Pediatrics. 2007; 120:898–921.
4. Yoshinaga-Itano C, Sedey AL, Coulter DK, Mehl AL. Language of early- and later-identified children with hearing loss. Pediatrics. 1998; 102:1161–1171.

5. Yoshinaga-Itano C, Gravel JS. The evidence for universal newborn hearing screening. Am J Audiol. 2001; 10:62–64.

6. Universal screening. US Preventive Services Task Force recommendation statement. Pediatrics. 2008; 122:143–148.
7. Nelson HD, Bougatsos C, Nygren P. Universal newborn hearing screening: systematic review to update the 2001 US Preventive Services Task Force Recommendation. Pediatrics. 2008; 122:e266–e276.

8. Yoshinaga-Itano C. Levels of evidence: universal newborn hearing screening (UNHS) and early hearing detection and intervention systems (EHDI). J Commun Disord. 2004; 37:451–465.

9. Pimperton H, Kennedy CR. The impact of early identification of permanent childhood hearing impairment on speech and language outcomes. Arch Dis Child. 2012; 97:648–653.

10. The Korean Audiological Society and the Korean Otologic Society. Korean Clinical Practice Guildline: newborn Hearing Screening 2010. Seoul: ML Communications;2011. p. 1–80.
11. Watkin PM. Outcomes of neonatal screening for hearing loss by otoacoustic emission. Arch Dis Child Fetal Neonatal Ed. 1996; 75:F158–F168.

12. Mason JA, Herrmann KR. Universal infant hearing screening by automated auditory brainstem response measurement. Pediatrics. 1998; 101:221–228.

13. Oh SH. Newborn hearing screening. The Korean Audiological Society. Practical manual of hearing tests. 1st ed. 2008. p. 247–268.
14. Finitzo T, Albright K, O'Neal J. The newborn with hearing loss: detection in the nursery. Pediatrics. 1998; 102:1452–1460.

15. Johnson JL, White KR, Widen JE, Gravel JS, James M, Kennalley T, et al. A multicenter evaluation of how many infants with permanent hearing loss pass a two-stage otoacoustic emissions/automated auditory brainstem response newborn hearing screening protocol. Pediatrics. 2005; 116:663–672.

16. Norton SJ, Gorga MP, Widen JE, Folsom RC, Sininger Y, Cone-Wesson B, et al. Identification of neonatal hearing impairment: a multicenter investigation. Ear Hear. 2000; 21:348–356.

17. White KR, Vohr BR, Meyer S, Widen JE, Johnson JL, Gravel JS, et al. A multisite study to examine the efficacy of the otoacoustic emission/auto mated auditory brainstem response newborn hearing screening protocol: research design and results of the study. Am J Audiol. 2005; 14:S186–S199.
18. Widen JE, Johnson JL, White KR, Gravel JS, Vohr BR, James M, et al. A multisite study to examine the efficacy of the otoacoustic emission/automated auditory brainstem response newborn hearing screening protocol: results of visual reinforcement audiometry. Am J Audiol. 2005; 14:S200–S216.
19. Gravel JS, White KR, Johnson JL, Widen JE, Vohr BR, James M, et al. A multisite study to examine the efficacy of the otoacoustic emission/automated auditory brainstem response newborn hearing screening protocol: recommendations for policy, practice, and research. Am J Audiol. 2005; 14:S217–S228.
20. UK Screening Portal [Internet]. UK: UK National Screening Committee. c2015. cited 2015 April 10. Available from: http://www.screening.nhs.uk/about/.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download