Abstract
The increase in medical research has led to a large body of related studies. The huge volume of research brings about a problem of how to organize and summarize the findings of studies. Meta-analysis is a statistical technique for combining the results from two or more studies, which addresses a similar hypothesis in a similar way. Meta-analysis includes the complete coverage of all relevant studies, and describes the results of each study via a quantitative index of effect size. Meta-analysis presents the precise estimate of treatment effect via combining these estimates across studies. Further, meta-analysis looks for the presence, degree and cause of heterogeneity, and explores the robustness of the main findings using statistical techniques. The author dealt with the some statistical issues and considerations which should be considered in conducting and presenting meta-analysis with explanation (ie. Effect size, Fixed and Random effect model, Heterogeneity, Reporting bias, and Meta-analysis Packages). This article may remind readers to conduct and evaluate the meta-analysis systematically and comprehensively.
Figures and Tables
References
1. Greenhalgh T. How to read a paper: papers that summarise other papers (systematic reviews and meta-analyses). BMJ. 1997; 315:672–675.

2. Higgins JP, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. The Cochrane Collaboration;2011. updated March 2011. Available from: http://www.cochrane-handbook.org.
3. Wolf FM. Meta-Analysis: Quantitative Method for Research Synthesis. Beverly Hills, CA: SAGE Publications, Inc;1986.
4. Pillemer DB, Light RJ. Synthesizing outcomes: how to use research evidence from many studies. Harvard Educ Rev. 1980; 50:176–195.

5. Egger M, Smith GD. Bias in location and selection of studies. BMJ. 1998; 316:61–66.
6. Egger M, Smith GD, Altman DG. Systematic reviews in health care: meta-analysis in context. London: BMJ Publishing Group;2001.
7. Furberg CD, Morgan TM. Lessons from overviews of cardiovascular trials. Stat Med. 1987; 6:295–306.

8. Hedges LV, Olkin I. Statistical methods for meta-analysis. Academic Press: 1985.
9. Rosenthal R. Parametric measures of effect size. In : Cooper H, Hedges LV, editors. The handbook of research synthesis. New York: Russell Sage Foundation;1994. p. 231–244.
10. Baumeister RF. Self-regulation, ego depletion, and inhibition. Neuropsychologia. 2014; 65:313–319.

11. Fisher RA. Frequency distribution of the values of the correlation coefficient in samples from an indefinitely large population. Biometrika. 1915; 10:507–521.

13. DerSimonian R, Kacker R. Random-effects model for meta-analysis of clinical trials: an update. Contemp Clin Trials. 2007; 28:105–114.

15. Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003; 327:557–560.

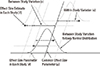
16. Song F, Sheldon TA, Sutton AJ, Abrams KR, Jones DR. Methods for exploring heterogeneity in meta-analysis. Eval Health Prof. 2001; 24:126–151.

17. Thompson SG, Higgins JP. How should meta-regression analyses be undertaken and interpreted? Stat Med. 2002; 21:1559–1573.

18. Simes RJ. Confronting publication bias: a cohort design for meta-analysis. Stat Med. 1987; 6:11–29.

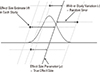
19. Peters JL, Sutton AJ, Jones DR, Abrams KR, Rushton L. Contour-enhanced meta-analysis funnel plots help distinguish publication bias from other causes of asymmetry. J Clin Epidemiol. 2008; 61:991–996.

20. Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994; 50:1088–1101.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download