Abstract
Induced pluripotent stem cells (iPSC) are specially manipulated cells from somatic cells by the introduction of four factors that are reprogrammed. The properties of iPSC are similar to embryonic stem cells (ESC) characteristic of self-renewal and pluripotency. The technology of reprogramming somatic cells to iPSC enables the generation of patient-specific cells that can be used as powerful tools for drug screening, in vitro models for human disease and autologous transplantation. The iPSC technology provides a priceless resource for regenerative medicine but there are still changing obstacles over the safety of iPSC in avoiding induction of tumorigenicity and maintaining high purity of re-differentiated cells from iPSC to produce more functional cells for cell therapy. A variety of methods to overcome the limitation of iPSC application applied in the clinical setting have been developed. In this review, we summarize the recent progress in iPSC generation and differentiation techniques to facilitate clinical application of iPSC with future potential in regenerative medicine.
Regenerative medicine has been an emerging field that represents replacement of lost or damaged tissues in the human body through either cellular transplantation or endogenous repair. It has been considered that stem cells are the potential mediators that function as reservoir for repair and regeneration of damaged tissues, because of its ability of differentiation into many different lineages of specialized cells [123]. Recently, the discovery of induced pluripotent stem cells (iPSC) which have differentiation capacity into all kind of cells in the body, called pluripotency, encourages the synthesis of scientific principles and technology in applications in regenerative medicine.
iPS cells can be achieved by reprogramming somatic cells using reprogramming factors and can undergo self-renewal as well as differentiate into all three germ layers [45]. The in vitro generation of pluripotent cells without the use of embryonic material has been rendered a more suitable source for regenerative medicine compared with nuclear transfer-embryonic stem cells (NT-ESC) [67] or somatic cell nuclear transfer (SCNT) [89]. Moreover, the one of advantages of iPSC is to overcome immune rejection that occurs when one uses cells of an individual to generate pluripotent stem cells. These features make iPSC a useful tool for drug screening, the human disease model and cell therapy including autologous transplantation [31011]. Thereby, since the initial report of mouse and human iPSCs by Takahashi and Yamanaka group, other groups recapitulated and found the iPSC technology to be more reproducible, effective, safe and a therapeutically compatible iPSC generation for the clinical setting. More studies about recent trends will be further discussed.
The challenge of iPSC technology remains similar to those faced in the human embryonic stem cells (hESC) field, such as appropriate differentiation of the cells and the risk of tumor formation following cellular transplantation, although the latter concerns regarding tumor development have been overcome by selective drugs to induce cell death of remaining iPSC after differentiation [1213]. There are still many unresolved issues mainly associated with the maturation of cells to a fully functional state. To overcome these limitations, recent research focuses on using devices and materials to help make iPSC into miniature organs to produce more functional cells [1415]. This may allow production of more appropriate cells for transplantation, meanwhile other goals for higher reproducibility and efficient differentiation protocol currently remain homework. Discussed here are recent advancements in this field related to regenerative medicine, with an emphasis on iPSC generation applicable in the clinical setting and creating more functional iPSC-derivates.
The primary component of reprogramming factors consists of klf4, Oct4, Sox2, and c-Myc which were effective throughout several types of mouse and human cells. Another set including Oct4, Sox2, Nanog, and Lin28 were also enough to reprogram human fibroblasts [16]. Traditionally, iPS cells could be induced from mouse or human fibroblasts through transduction of reprogramming factors mediated by virus systems, which might bring about insertional mutagenesis. In addition, upon re-differentiation of iPSC into target cells, reactivation of reprogramming factors integrated in the donor genome have raised concerns regarding the clinical application due to induction of tumorigenicity [17]. Many studies have been developed to overcome this issue by approaching geno-me footprint free-/non-integrating iPSC for clinically applicable iPSC generation [1819]. Table 1 shows the classified reprogramming techniques based on efficiency, value of footprint, and number of donor types to be reprogrammed as previously reported.
Initial iPSC generation from mouse and human were mediated by retro- and lenti-virus containing four transcription factors. Those systems cause the integration of transgene into donor genome to allow for insertional mutagenesis, thus limiting use of iPSC for clinic. Moreover, iPSC generated by virus often maintains transgene expression, although silencing of transgene occurs gradually during iPSC induction, and then finally undergoes silencing in the pluripotent state. Importantly, the efficiency of iPSC generation was much lower than 0.01% [520]. After this first discovery, modifications of reprogramming methods were performed to eliminate the integration of transgene. Soldner et al. reported integration of free-human induced pluripotent stem cells (hiPSC) from patient cells with Parkinson's disease using the Cre-loxP mediated transgene excision system. The transgenes can be removed by treating Cre-recombinase protein once cells are fully reprogra mmed [2122]. We also used Cre-loxP mediated reprogramming methods to generate hiPSC from human fibroblast cells (Fig. 1).
To overcome the main obstacle of clinical implication of iPSC, researchers have been established integration-free iPSC using adenovirus [23], sendai-virus [24], proteins [25], mRNA [26], miRNA [27] and episomal vector [28] expressing reprogramming factors. However, these methods are still necessary for much work to be widely used for the following reasons: 1) Need for intensive labor, 2) Extremely low reprogramming efficiency, 3) Validation in other cell types other than fibroblasts, 4) Long term passage and culture at a higher temperature of reprogrammed iPSC to be genome footprint-free iPSC. Although more validation is required before non-integrating reprogramming methods can be adapted, episomal reprogramming might be considered to be an effective strategy to generate footprint-free iPSC.
Studies on the processes of reprogramming have shown that modulation of the epigenetic status in cells play an important role in generating iPSC during cellular reprogramming [2930]. Epigenetic modulators can modify chromatin structure and make it more permissive to transcriptional machinery during reprogramming. Various small molecules against epigenetic modification can improve the reprogramming efficiency, thus these reagents including inhibitor of histone deacetylatase (HDAC) and DNA methyltransferase (DNMT), are most generally used in reprogramming methods [313233]. One interesting thing is that these small molecules even substitute some of the roles of reprogramming factors by regulating gene expression of pluripotent transcription factors, leading to enhanced reprogramming of somatic cells into iPSC. Using this strategy, recently, chemically iPSC (CiPSC) are generated from mouse embryonic fibroblasts (MEF) by treating small molecules (VPA, Chir99021, Forskolin, DzNep, Tranylcypromine, TTNPB), but not from human cells yet [34]. A remarkable finding of small molecule based-iPSC would provide significant advantages to reduce the risk of genetic modification and improve the quality of established iPSC. Given that human iPSC were derived without the use of genetic factors are a promising resource in its clinical application as integration-free iPSC, the new optimization of sequential and combinatorial treatments with small molecules should be necessary to enable reprogramming in human cells.
After obtaining reprogrammed cells, identification of iPSC colonies is necessary to determine whether selected the colony is fully reprogrammed by investigating hESC-like morphological criteria, expression of hESC markers endogenously (Tra1-81, Nanog, Oct4, Sox2), loss of transgenes, and ability of differentiation potential as teratoma formation in the case of human iPSC.
The first generated iPSC was selected with ESC-specific gene, Fbx15, which induces partially reprogrammed cells, not yet fully reprogrammed. It was later found that selected colonies with nanog or Oct4, ESC-specific genes, were much more similar to properties of ESC [45]. The identification of human iPSC colony was often based upon morphological features such as a cobblestone appearance with prominent nucleoli and pronounced individual cell borders. Of note is that non-iPSC colonies similar morphologically also appeared during cell reprogramming. Additional methods to identify true hiPSC have been using hESC surface antigen expression and loss of transgene dependence as strategies to identify fully reprogrammed cells. In humans, live staining of cultures for Tra 1-81, hESC-specific surface marker has become a useful tool to distinguish between true or false iPSC [35]. The loss of transgene expression related to full reprogramming, can be evaluated by using reporter gene including green or red fluorescence protein (GFP or RFP) that are silenced in the pluripotent state [36]. Finally, whether a fully reprogrammed state has been achieved by assessing the functional properties of iPSC including expression of other hESC surface markers (SSEA4), alkalinposphatase activity, endogenous pluripotent markers (Nanog, Oct4, Sox2) on a molecular level, and the ability to differentiate into lineages cells derived three embryonic germ layer on a functional level. Fig. 2 shows, at molecular levels, the functional properties of iPSC generated from human fibroblasts using Cre-loxP mediated reprogramming methods.
One of the major challenges for cell therapy using stem cells is host immune rejection of the transplanted cells. To facilitate clinical application of stem cells, it is important to develop therapeutically valuable cells that do not induce an immune rejection. Currently, iPS cell lines can be generated from patients with several genetic disorders [35]. Theses patient-specific derived target cells can serve as easily accessible autologous source for cell replacement therapies as well as drug screening and in vitro disease models [10]. Thereby, iPSC technology providing potentially unlimited autologous cells is especially attractive for regenerative medicine. Despite the advantages of iPSC-derivates, there still remain many obstacles for transplantation including whether iPSC-derivates are mature functional cells that can be used clinically. Recent progress on iPSC technology including detailed differentiation required to fulfill the promises (or dreams) of stem cell-based therapeutics for human disease will be discussed.
For cell therapy in the clinic setting, clinically applicable hiPSC generation is the first step with the ultimate goal being reproducible differentiation and maximum enrichment to specific cell lineages. While multiple protocols have been developed to generate the clinically relevant cell types including cardiomyocytes [37], hematopoetic progenitors [38], neurons [39], hepatocytes [40] in vitro, there still are limitations in the use of iPSC-derivates for regenerative regeneration. The best differentiation seems to produce immature cells but they are not engraftable and mature functional cells are required for tissue regeneration. For example, in trials to produce mature blood cells from hiPSC, there has been little success in generating a cell type that will engraft into the bone marrow of irradiated mice, one of the functional features of mature blood cells [41] Considering that the clinical application of stem cells will depend on obtaining sufficient numbers of functional mature cells, the generation of enriched populations of various differentiated cell lineages from hiPSC and improved functionality of their differentiated derivatives has been addressed by using various devices and materials such as microfluidic bioreactors, suspension culture or co-culture with tissue-specific ECM coating.
Microfluidic bioreactors have shown their potential to establish highly controllable microenvironmental cues. The 3D niche mimics the in vivo microenvironments to contribute both autocrine and paracrine functions in individual hiPSC [42]. Also, stem cell cultured matrigel-coated polystyrene microcarriers also feasibly expand endodermal cells with high numbers/efficiency in 8 days to greater than 80% as determined by co-expression of Sox17 and Foxa2 [43]. ECM coatings by tissue materials enhanced the maturation of both skeletal progenitors and stem cell derived cardiomyocytes by mimicking the in vivo 3D ECM microenvironment [44]. Therefore, control of the dynamics of intercellular interactions in multidimensional environments represents powerful approaches to achieve functional manure cell types from pluripotent stem cells.
One of the most exciting developments of hiPSC differentiation is shown in the 3D based organogenesis from iPSC which regenerate brain [4546] intestinal [47] and liver [48] organs in a dish, called organoid. Among iPSC derived organoids, we introduced detailed organoid of the liver as we worked this field previously [36]. A co-culture system based on three dimensions has been employed in an approach of generating a liver organoid. Aggregation of three cell types including Human Umbilical Vein Endothelial Cells (HUVEC), hMSC and hepatic cells derived from hiPSC accelerated the exhibition of liver budding by allowing their self-organization in a 3D culture system. Indeed, transplantation of liver organoid was able to mature hepatocytes and resembled adult liver histologically in vivo as well as showing therapeutic effect by rescuing mice with liver injury [49]. The approach of self-organization into functional 3D liver buds would provide a developmental basis for establishing efficiently mature functional cells from hiPSC.
Recent progress of iPSC strategy for generation and differentiation has provided a great opportunity to accelerate translation of iPSC technology into clinical therapy. The clinically relevant reprogrammed cells such as integration free-, footprint free- and small molecule based-iPSC will contribute to the use of iPSC in regenerative medicine. To cell replacement therapies, patient-specific iPSC-derivates based on the 3D organogenesis system that produce functionally mature target cells by inducing the process of self-organization would have superior resources to applicative autologous transplantation. In the future, the challenges to enable/improve the generation of high survival and engraftable cells in vivo from iPSC are undoubtedly unveiled leading to answers in any unmet medical needs.
Figures and Tables
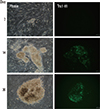 | Fig. 1Mophology and expression of hESC-specific surface antigen on human fibroblast transduced with cre-excisable reprogramming factors during the course of reprogramming. Transduced fibroblasts with cre-excisable polycistronic lentivirus containing reprogramming four factors (klf4, oct4, sox2, and c-myc) were cultured for indicated days on mouse embryonic fibroblast (MEF) in hESC medium. Picture shows the morphology (left) and livestaining of Tra 1-81 (right), surface marker of pluripotent stem cells on transduced fibroblasts during culture. Medium was changed daily. MEF used as feeder cells. Bars indicate 100 µm. hESC, human embryonic stem cells. |
 | Fig. 2Expression of hESC surface antigens and pluripotent markers in integration free-hiPSC derived from human fibroblast. iPSC coloies were picked up and sub-cultured. (A) Images of sub-cultured iPS and livestaining of surface antigen (SSEA4 and Tra1-60). Bars indicate 100 µm. (B) Immunostaining of pluripotent markers of hiPSC at passage 6. Bars indicate 100 µm. hESC, human embryonic stem cells; hiPSC, human induced pluripotent stem cells; iPSC, induced pluripotent stem cells; SSEA4, stage specific embryonic antigen 4. |
ACKNOWLEDGMENTS
This research was supported by the National R&D Program through the Korea Institute of Radiological and Medical Sciences funded by the Ministry of Science, ICT & Future Planning (No. 1711021870).
References
2. Svendsen CN. Back to the future: how human induced pluripotent stem cells will transform regenerative medicine. Hum Mol Genet. 2013; 22:R32–R38.

3. Chen KG, Mallon BS, McKay RD, Robey PG. Human pluripotent stem cell culture: considerations for maintenance, expansion, and therapeutics. Cell Stem Cell. 2014; 14:13–26.

4. Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006; 126:663–676.

5. Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007; 131:861–872.

6. Wakayama S, Jakt ML, Suzuki M, Araki R, Hikichi T, Kishigami S, et al. Equivalency of nuclear transfer-derived embryonic stem cells to those derived from fertilized mouse blastocysts. Stem Cells. 2006; 24:2023–2033.

7. Brambrink T, Hochedlinger K, Bell G, Jaenisch R. ES cells derived from cloned and fertilized blastocysts are transcriptionally and functionally indistinguishable. Proc Natl Acad Sci U S A. 2006; 103:933–938.

8. Cibelli JB, Stice SL, Golueke PJ, Kane JJ, Jerry J, Blackwell C, et al. Transgenic bovine chimeric offspring produced from somatic cell-derived stem-like cells. Nat Biotechnol. 1998; 16:642–646.

9. Wakayama T, Tabar V, Rodriguez I, Perry AC, Studer L, Mombaerts P. Differentiation of embryonic stem cell lines generated from adult somatic cells by nuclear transfer. Science. 2001; 292:740–743.

10. Rajamohan D, Matsa E, Kalra S, Crutchley J, Patel A, George V, et al. Current status of drug screening and disease modelling in human pluripotent stem cells. Bioessays. 2013; 35:281–298.

11. Halevy T, Urbach A. Comparing ESC and iPSC-Based Models for Human Genetic Disorders. J Clin Med. 2014; 3:1146–1162.

12. Ben-David U, Gan QF, Golan-Lev T, Arora P, Yanuka O, Oren YS, et al. Selective elimination of human pluripotent stem cells by an oleate synthesis inhibitor discovered in a high-throughput screen. Cell Stem Cell. 2013; 12:167–179.

13. Lee MO, Moon SH, Jeong HC, Yi JY, Lee TH, Shim SH, et al. Inhibition of pluripotent stem cell-derived teratoma formation by small molecules. Proc Natl Acad Sci U S A. 2013; 110:E3281–E3290.

14. Sasai Y. Next-generation regenerative medicine: organogenesis from stem cells in 3D culture. Cell Stem Cell. 2013; 12:520–530.

15. Ranga A, Gjorevski N, Lutolf MP. Drug discovery through stem cell-based organoid models. Adv Drug Deliv Rev. 2014; 69-70:19–28.

16. Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, et al. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007; 318:1917–1920.

17. Nakagawa M, Koyanagi M, Tanabe K, Takahashi K, Ichisaka T, Aoi T, et al. Generation of induced pluripotent stem cells without Myc from mouse and human fibroblasts. Nat Biotechnol. 2008; 26:101–106.

18. Malik N, Rao MS. A review of the methods for human iPSC derivation. Methods Mol Biol. 2013; 997:23–33.

19. Higuchi A, Ling QD, Kumar SS, Munusamy MA, Alarfaj AA, Chang Y, et al. Generation of pluripotent stem cells without the use of genetic material. Lab Invest. 2015; 95:26–42.

20. Stadtfeld M, Hochedlinger K. Induced pluripotency: history, mechanisms, and applications. Genes Dev. 2010; 24:2239–2263.

21. Soldner F, Hockemeyer D, Beard C, Gao Q, Bell GW, Cook EG, et al. Parkinson's disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell. 2009; 136:964–977.

22. Sommer CA, Sommer AG, Longmire TA, Christodoulou C, Thomas DD, Gostissa M, et al. Excision of reprogramming transgenes improves the differentiation potential of iPS cells generated with a single excisable vector. Stem Cells. 2010; 28:64–74.

23. Zhou W, Freed CR. Adenoviral gene delivery can reprogram human fibroblasts to induced pluripotent stem cells. Stem Cells. 2009; 27:2667–2674.

24. Nishimura K, Sano M, Ohtaka M, Furuta B, Umemura Y, Nakajima Y, et al. Development ofdefective and persistent Sendai virus vector: a unique gene delivery/expression system ideal for cell reprogramming. J Biol Chem. 2011; 286:4760–4771.
25. Zhou H, Wu S, Joo JY, Zhu S, Han DW, Lin T, et al. Generation of induced pluripotent stem cells using recombinant proteins. Cell Stem Cell. 2009; 4:381–384.

26. Warren L, Manos PD, Ahfeldt T, Loh YH, Li H, Lau F, et al. Highly efficient reprogrammingto pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell. 2010; 7:618–630.

27. Anokye-Danso F, Trivedi CM, Juhr D, Gupta M, Cui Z, Tian Y, et al. Highly efficient miRNA-mediated reprogramming of mouse and human somatic cells to pluripotency. Cell Stem Cell. 2011; 8:376–388.

28. Yu J, Hu K, Smuga-Otto K, Tian S, Stewart R, Slukvin II, et al. Human induced pluripotentstem cells free of vector and transgene sequences. Science. 2009; 324:797–801.

29. Orkin SH, Hochedlinger K. Chromatin connections to pluripotency and cellular reprogramming. Cell. 2011; 145:835–850.

30. Liang G, Zhang Y. Embryonic stem cell and induced pluripotent stem cell: an epigenetic perspective. Cell Res. 2013; 23:49–69.

31. Huangfu D, Maehr R, Guo W, Eijkelenboom A, Snitow M, Chen AE, et al. Induction of pluripotent stem cells by defined factors is greatly improved by small-molecule compounds. Nat Biotechnol. 2008; 26:795–797.

32. Huangfu D, Osafune K, Maehr R, Guo W, Eijkelenboom A, Chen S, et al. Induction of pluripotent stem cells from primary human fibroblasts with only Oct4 and Sox2. Nat Biotechnol. 2008; 26:1269–1275.

33. Mali P, Chou BK, Yen J, Ye Z, Zou J, Dowey S, et al. Butyrate greatly enhances derivation of human induced pluripotent stem cells by promoting epigenetic remodeling and the expression of pluripotency-associated genes. Stem Cells. 2010; 28:713–720.

34. Hou P, Li Y, Zhang X, Liu C, Guan J, Li H, et al. Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science. 2013; 341:651–654.

35. Lowry WE, Richter L, Yachechko R, Pyle AD, Tchieu J, Sridharan R, et al. Generation of human induced pluripotent stem cells from dermal fibroblasts. Proc Natl Acad Sci U S A. 2008; 105:2883–2888.

36. Lee SB, Seo D, Choi D, Park KY, Holczbauer A, Marquardt JU, et al. Contribution of hepatic lineage stage-specific donor memory to the differential potential of induced mouse pluripotent stem cells. Stem Cells. 2012; 30:997–1007.

37. Acimovic I, Vilotic A, Pesl M, Lacampagne A, Dvorak P, Rotrekl V, et al. Human pluripotent stem cell-derived cardiomyocytes as research and therapeutic tools. BioMed Res Int. 2014; 2014:512831.

38. Kim EM, Manzar G, Zavazava N. Human iPS cell-derived hematopoietic progenitor cells induce T-cell anergy in in vitro-generated alloreactive CD8(+) T cells. Blood. 2013; 121:5167–5175.

39. Schondorf DC, Aureli M, McAllister FE, Hindley CJ, Mayer F, Schmid B, et al. iPSC-derived neurons from GBA1-associated Parkinson's disease patients show autophagic defects and impaired calcium homeostasis. Nat Commun. 2014; 5:4028.

40. Sun C, Wilson GS, Fan JG, Qiao L. Potential applications of induced pluripotent stem cells (iPSCs) in hepatology research. Curr Stem Cell Res Ther. 2015; 10:208–215.

41. Ye Z, Chou BK, Cheng L. Promise and challenges of human iPSC-based hematologic disease modeling and treatment. Int J Hematol. 2012; 95:601–609.

42. Moledina F, Clarke G, Oskooei A, Onishi K, Gunther A, Zandstra PW. Predictive microfluidic control of regulatory ligand trajectories in individual pluripotent cells. Proc Natl Acad Sci U S A. 2012; 109:3264–3269.

43. Lock LT, Tzanakakis ES. Expansion and differentiation of human embryonic stem cells to endoderm progeny in a microcarrier stirred-suspension culture. Tissue Eng Part A. 2009; 15:2051–2063.

44. DeQuach JA, Mezzano V, Miglani A, Lange S, Keller GM, Sheikh F, et al. Simple and high yielding method for preparing tissue specific extracellular matrix coatings for cell culture. PLoS One. 2010; 5:e13039.

45. Eiraku M, Takata N, Ishibashi H, Kawada M, Sakakura E, Okuda S, et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature. 2011; 472:51–56.

46. Efthymiou A, Shaltouki A, Steiner JP, Jha B, Heman-Ackah SM, Swistowski A, et al. Functional screening assays with neurons generated from pluripotent stem cell-derived neural stem cells. J Biomol Screen. 2014; 19:32–43.

47. Spence JR, Mayhew CN, Rankin SA, Kuhar MF, Vallance JE, Tolle K, et al. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011; 470:105–109.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download