Abstract
Recently, regenerative medicine utilizing tissue manufacturing has been a creative topic of study, offering promise for resolving the gap between insufficient organ supply and transplantation needs. Moreover, 3D generation of functional organs is seen as the main hope to resolve these issues that will be a major advancement in the field over the next generation. Organ printing is the 3D construction of functional cellular tissue that can replace organs made by additive biofabrication with computational technology. Its advantages offer rapid prototyping (RP) methods for fabricating cells and adjunctive biomaterials layer by layer for manufacturing 3D tissue structures. There is growing interest in applying stem cell research to bio-printing. Recently several bio-printing methods have been developed that accumulate organized 3D structures of living cells by inkjet, extrusion, and laser based printing systems. By printing spatially organized gradients of biomolecules as an extracellular matrix, direct stem cell seeding can then be engineered to differentiate into different lineages forming multiple subpopulations that closely approximate the desired organ. Pliable implementation patches can Stem cells for tissue regeneration can be arranged or deposited onto pliable implementation patches with the purpose of generating functional tissue structures. In this review, current research and advancement of RP-based bio-printing methods to construct synthetic living organs will be discussed. Furthermore, recent accomplishments in bioprinting methods for stem cell study and upcoming endeavors relevant to tissue bioengineering, regenerative medicine and wound healing will be examined.
An insufficient supply of organs for transplant is an ever present obstacle for transplant medicine. A long term resolution of these problems is being sought by developing ways to engineer live organs from a person's own cells. Previous tissue manufacturing approaches involved seeding either adult or stem cells onto various 3D scaffolds, with subsequent cell proliferation and differentiation into spatially-arranged functional tissue. However, the approach has not been successful as organs are very complicated. Advances in our understanding of an organ's tissue components and progress in 3D printing technology have allowed a new era of bioprinting to unfold. Bioprinting is described as a 3D printing system that provides precise accumulation and arrangement of living cells, extracellular matrix elements, biochemical components, proteins, and bio-substances on solid or gel reservoirs that can closely mimic the living organ which is being artificially constructed [1,2,3,4]. Substantial achievements in understanding the intricate structure of organs have led researchers to conclude that artificial organ construction demands highly accurate placement of these components including complex vascular integration [5]. Recent advances in the development of 3D printers utilizing laser-based, inkjet-based and extrusion systems have facilitated the development of techniques that allow the construction of these intricate organ structures with precise arrangement of matrix, vasculature and cells [6]. Bio-printing is providing a greater ability in tissue engineering and researchers are hoping to be able to manufacture de novo living organs for transplant in the near future to alleviate the chronic shortage of organs for transplant. This review discusses the present state of bio-printing techniques, its benefits and limitations, and future trends towards the manufacturing of living synthetic organs both for research and clinical application.
3D printing can be considered as synonymous or analogous to the concept of additive manufacturing (AM), solid-freeform technology, or rapid prototyping (RP) as first developed by Charles Hull. In 1983, Hull first conceived of the idea 3D printing, calling it stereolithography. The format would continue evolving to connect computer aided design (CAD) software to produce designs that could be transmitted as files for 3D printing. Michael Cima and Emanuel Sachs, MIT professors, conceived term of "3D printer" in 1993 developing further techniques to manufacture plastic, ceramic, and metal products [7]. 3D printing has been applied in multiple industries such as the automotive and aerospace industries for the rapid manufacturing of new design prototypes for airplane and car parts [8]. Soon after, medical applications began to be developed in the early 2000s, especially for manufacturing individualized dental prosthetics and implants [9,10]. 3D printing has been expanded to numerous other medical applications including biomedical engineering.
AM applied developments in precision controlled laser and inkjet printing technology by expanding them to three dimensions (3D) creating solid objects, rather than just designs of ink on paper. The AM manufacturing technique reads CAD data to build 3D constructs layer by layer according to the virtual design [12]. Bio-fabrication is an emerging expansion of these AM technologies to the manufacture of complex 3D synthetic organs and tissues, using them to overcome limitations of conventional tissue engineering techniques [11]. Computer-aided bio-additive manufacturing of 3-D tissue and organs, involves the highly accurate deposition of living cells with an associated hydrogel scaffold of biomaterials mimicking vascular structure, basement membranes and associated extracellular matrix. This has allowed bio-printing to be extended to tissue engineering for the creation of de novo organs. Bio-additive fabricating techniques include laser-used [13], inkjet-used [14], extrusion-used [15] methods.
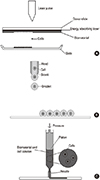
Various kinds of bio-printing machines have been developed from all over the world including Korea (Fig. 1). These printers are capable of using CAD plans to precision print cells in a matrix of biomaterials that can mimic the functional structure of an organ, such as liver or heart. Newly developed biomaterials having characteristics of biocompatibility and degradability that are used to fabricate cell-scaffold constructs allowing the printed cells, either adult differentiated cells or stem cells, to establish and maintain themselves and take on the roles they would perform in the naturally occurring organ. Since new advances in stem cell science suggest that it is entirely possible to use a patient's own cells in the manufacture of the organ, it is hoped that these will not only provide an alternative to replace donor organs, but that the transplanted de novo organ can also decrease chances of complications due to rejection, as it will be autologous.
Bio-printing technology commonly uses three common 3D printing methods, laser, inkjet, and extrusion techniques (Fig. 2). Recent laser systems used for bio-fabrication are capable of building high resolution patterns of viable cells layer by layer on the scaffold [16,17]. Laser energy transfers cells hanging on a material in donor slides to a collector substrate. A bubble formation created by a laser pulse pushes the cells to the collector substrate. This high degree of precision is the primary advantage of laser-based systems [18]. However, overheating of the cells and excessive exposure of the cellular bio-layer to laser energy has been a disadvantage and remains a challenge [19,20,21]. One possibility to surmount the challenges has been the use of lower energy infrared [17]. Although determining the optimal wavelength remains an issue, laser-based printing has not been shown to cause mutations or apoptosis [17].
In rapid development since the early 2000's, inkjet bio-printing will likely be the primary foundation for organ printing technologies in the future. Inkjet bio-printers eject suspension droplets onto a surface rasterizing a pattern from CAD software produced template instructions using reservoirs of suspended adult cells, stem cells, or bio-molecular substrate for scaffolding in printer cartridges. The CAD software digitally encodes the design data to fabricate tissue or organs [14]. Inkjet printers utilize currently available technology to provide flexible and reasonable solutions for constructing complicated encapsulated organs such as the heart [22] (Table 1). While also being capable of printing and patterning multi-layer cell matrix patterns for skin regeneration on injuries in situ in a process similar to spray painting [23]. In spite of these advantages, inkjet-based systems still have a number of significant limitations including sedimentation and aggregation of cells and biomaterials in reservoir cartridges causing frequent blockage of printhead nozzles and excessive cell damage due to passage through the small diameter, heat and electrical charge of the nozzle orifice, causing cell necrosis and apoptosis. These processes cell sedimentation, aggregation and damage compound over time, limiting the printing of cells in high densities [24,25,26]. Another challenge is the maintenance of structural integrity of the organ tissue during bio-printing that needs to be resolved by creating better suspensions that can allow better control of droplet form and more rapid fusing of the printed droplets to form the desired 3D structure.
Extrusion-based printing utilizes a continuous formation and accumulation of cylindrical supporting struts that provide better structural integrity during bio-printing. Extrusion printing presses dots or lines of material into the desired structural shape via computer-controlled actuators using mechanical and/or pneumatic power. Under robotic control, a pressure-assisted system dispenses either cellular or acellularbio-material precisely depositing the encapsulated cell in 3-D structures mimicking the desired [6]. Extrusion-based printing is currently the most convenient system for manufacturing 3-D cellular structures, but the system requires a rapid process of cell encapsulation,resulting in shear-induced cell distortion and there is a resulting restriction of material variety.
Organ printing is a process of self-assembly. Small units of engineered tissue consisting of bio-printed cellular aggregates are amassed layer by layer to form the 3D structure of a functional organ [27,28,29]. Development of clinically functional bio-printed organs that can be transplanted and then physiologically integrate with the recipient patient requires advancements in three related technologies [30]. First we need better understanding of cell biology and development of biotechnology to secure adequate populations of clinically functional cells. Then advancements in bio-printing processes are needed that allow creation of 3D conformations of cells and biomaterials that more closely mimic the natural organ function. Finally we need to be able to integrate the bio-synthesized organ to function and perform in vivo, i.e., in the patient. Challenges that we face in the final step of transplantation include ensuring immune tolerance of the organ in the patient, and the demonstration of safety and efficacy of the bio-printed de novo synthesized organ, and finally ability of the manufacturer to maintain adequate production and quality control.
Recent research trends in organ bio-printing have focused on the development of bio-gels that better provide structural templates for the genesis of adequate vascularization of the organ tissue. Bio-gels must also promote cellular aggregate formation and fusion in the appropriate conformation to mimic the structural and functional capacity of the target organ. Factors to balance in tissue printing involve creating cell-matrix composites that supply sufficient cohesion between aggregates while allowing cells to migrate and take position where they are needed. If bio-printing bio-gels are too fluid, there is insufficient cohesion to maintain desired structure, but if they are too rigid, then necessary cell migrations will be prohibited [29,31].
In 3-D bio-printing technologies, providing insufficient vascularization for 3-D organs is a fatal flaw. Without vascularization, cells cannot live, let alone function as normal tissues or organs [32,33]. Successful manufacture of 3-D tissues or organs absolutely requires vascularization, for without it the cells cannot exchange nutrients, remove wastes,or exchange gas, all of which are necessary for growth, maturation and long-term homeostatic maintenance of the bioengineered tissues. Current bio-printing methods are incapable of multiscale manufacture of branched vasculature and micro-vasculature capable of long term sustenance of a bio-mimetically synthesized organ. There is ongoing research using computer generated modeling of vascular structure formation in maturing organs, but there have been only a few partially successful endeavors toward the manufacturing of bifurcated and branched vascular trees in organ engineering [34,35]. Creaton of functionally integrated bifurcated vessels for organ and tissue engineering still remains a limiting obstacle challenging the maturation of bio-printing technologies.
Bio-printing refers to tissue regeneration by fabricating scaffolds and tissue constructs with functional 3D structures. Its potential has been demonstrated in a number of studies. In one study, extrusion- bio-printing was used to fabricate a trileaflet heart valve conduit, comprised of human aortic valve interstitial cells and hyaluronic acid and gelatin hydrogel showing the feasibility of bio-printing cellularized tissue valves for clinical use [36,37]. In another example demonstrating possible clinical application of current technologies, computer-controlled inkjet bio-printers printed layer by layer, functional human cartilage plugs using poly (ethylene glycol) dimenthacrylate (PEGDMA) plus human chondrocytes that were subsequently used to repair osteochondral defects in human patients [38]. The printed chondrocytes retained the initially deposited shape obtained by photopolymerization of biomaterial scaffold with precise cell distribution to allow anatomic cartilage engineering [38].
Cartilage 3D bio-printing has also been used in 2013 [39]. In this study, a bio-resorbable, customized tracheal splint was implanted to an infant with tracheobronchomalacia. The splint was designed with CAD based on imaging of the patient's airway and bio-fabricated with polycaprolactone using laser-based bio-printing. The implanted splint was circumferentially covered with the malacology left bronchus allowing functional expansion of the ariway. Subsequently, the bronchus revealed normal patency without collapse in bronchoscopy and normal level ventilation was recovered as measure by normal partial pressure of carbon dioxide in venous. This case suggests that CAD and 3D bio-printing can facilitate the manufacture of individualized, implantable bio-mimetic materials with structural features that are anatomically specific to the patient.The utilization of bio-printing for skeletal reconstruction has also been studied using extrusion bio-printing methods to fabricated bone using bone marrow stromal cells (BMSC) suspended and imprinted using hydrogels [40]. Bio-printing fabrication has been used to develop vascularized bone grafts [41]. Chondrocytes, from human multipotent stromal cells (MSC) have been engineered to organize construction of extracellular matrix [40,42]. These cells passing the nozzles retained viability and demonstrated osteogenic differentiation [43]. Furthermore, two different cell groups could be combined within single constructs by changing the nozzle in the course of deposition. It indicates that this 3D deposition technique is appropriate for bone grafts using multiple cell types [40].
Bio-printing has been used for repairing peripheral nerve injury by manufacturing biological nerve grafts. Like many other tissue, injured peripheral nerves do not fully regenerate motor and/or sensory function resulting in chronic disability or chronic pain or loss of sensation distal to the wound origin. In a rat model, nerve constructs comprised of stem cells and Schwann cells were successfully transplanted [44].
Another application utilizes bio-printing technology for repair and regeneration of the skin. Skin substitutes have been synthesized both in vitro and in situ by inkjet-used bio-printing systems [45]. In the study, human skin cell suspensions of fibroblasts and keratinocytes were imprinted using inkjet-based bio-printers capable of creating biomimetic multilayered skin tissue.
Bio-fabrication techniques are under development to construct bio-substance scaffolds consisting of microenvironment that can support and direct the differentiation and maturation of pluripotent or multipotent stem cells into adult tissue. Stem cells provide a high potential for creating complex constructs, and are expected to play a leading part in the engineering of 3D tissues or organs [27]. A challenge of stem cell applications in bio-fabrication will be to optimize the bio-hydrogels used to generae the microenvironment; integrating signals for bothcell proliferation and differentiation as well as the mechanical characteristics to imitate the in vivo conditions. Stem cell proliferation and differentiation to generate mature tissue that maintains the organ-specific phenotype are influenced by diverse biological components [46,47,48].
To optimize manufacturing design for production, computer simulations and mathematical modeling will play an important role in modern fabrication schemes [27,33]. These computer stimulations and mathematical studies will likely give greater understanding of bio-printing technologies and fabrication reproduce mechanical effects important in organ development as well as understanding encapsulating micro-environment of the tissue cells [49,50]. Deposition of 3D biomaterial is currently impeded by unsuitable manufacturing parameters such as deposition speed and overcoming the numerous challenges affecting printing rated will have many beneficial consequences. Computer simulations can predict and optimize the 3D constructs before printing. Nonetheless, this computational and mathematical methodology require further development and optimization to substantiate the promise for 3D bio-printing of engineered tissues and organs for use clinical use and research studies.
Another promising future for 3D bio-printing is in situ creation or repair of human tissues and organs in the clinic or in the surgical suite. In situ 3D bio-printing currently is investigated for repairing organs [23]. As noted above, for repair and regeneration of injured skin, the damaged area is filled in by successive layers of multiple cell types such as keratinocytes and fibroblasts. This innovative approach is at the cutting edge of 3D bio-printing, to therapeutically repair in situ injured tissues and damaged internal organs which do not have self-repair abilities.
Figures and Tables
 | Fig. 13D bio-printer available in Korea (given by KIMM). Hydrogel and cells can be injected by bio-printer with pre-determined CAD program and tissue with cells and biodegradable polymer can be made. |
References
1. Demirci U, Montesano G. Single cell epitaxy by acoustic picolitre droplets. Lab Chip. 2007; 7:1139–1145.

2. Moon S, Hasan SK, Song YS, Xu F, Keles HO, Manzur F, et al. Layer by layer three-dimensional tissue epitaxy by cell-laden hydrogel droplets. Tissue Eng Part C Methods. 2010; 16:157–166.

3. Mironov V, Kasyanov V, Drake C, Markwald RR. Organ printing: promises and challenges. Regen Med. 2008; 3:93–103.

4. Xu F, Moon SJ, Emre AE, Turali ES, Song YS, Hacking SA, et al. A droplet-based building block approach for bladder smooth muscle cell (SMC) proliferation. Biofabrication. 2010; 2:014105.

6. Melchels FP, Domingos MA, Klein TJ, Malda J, Bartolo PJ, Hutmacher DW. Additive manufacturing of tissues and organs. Prog Polym Sci. 2012; 37:1079–1104.

7. Cima MJ, Haggerty JS, Sachs EM, Williams PA. Three-dimensional printing techniques. Google Patents. 1993.
8. Bak D. Rapid prototyping or rapid production? 3D printing processes move industry towards the latter. Assem Autom. 2003; 23:340–345.

9. Leukers B, Gulkan H, Irsen SH, Milz S, Tille C, Schieker M, et al. Hydroxyapatite scaffolds for bone tissue engineering made by 3D printing. J Mater Sci Mater Med. 2005; 16:1121–1124.

10. Mironov V, Boland T, Trusk T, Forgacs G, Markwald RR. Organ printing: computer-aided jet-based 3D tissue engineering. Trends Biotechnol. 2003; 21:157–161.

11. Nakamura M, Iwanaga S, Henmi C, Arai K, Nishiyama Y. Biomatrices and biomaterials for future developments of bioprinting and biofabrication. Biofabrication. 2010; 2:014110.

12. Peltola SM, Melchels FP, Grijpma DW, Kellomaki M. A review of rapid prototyping techniques for tissue engineering purposes. Ann Med. 2008; 40:268–280.

13. Barron JA, Wu P, Ladouceur HD, Ringeisen BR. Biological laser printing: a novel technique for creating heterogeneous 3-dimensional cell patterns. Biomed Microdevices. 2004; 6:139–147.

14. Boland T, Xu T, Damon B, Cui X. Application of inkjet printing to tissue engineering. Biotechnol J. 2006; 1:910–917.

15. Mironov V. Printing technology to produce living tissue. Expert Opin Biol Ther. 2003; 3:701–704.

16. Odde DJ, Renn MJ. Laser-guided direct writing for applications in biotechnology. Trends Biotechnol. 1999; 17:385–389.

17. Nahmias Y, Odde DJ. Micropatterning of living cells by laser-guided direct writing: application to fabrication of hepatic-endothelial sinusoid-like structures. Nat Protoc. 2006; 1:2288–2296.

18. Schiele NR, Chrisey DB, Corr DT. Gelatin-based laser direct-write technique for the precise spatial patterning of cells. Tissue Eng Part C Methods. 2011; 17:289–298.

19. Xu F, Inci F, Mullick O, Gurkan UA, Sung Y, Kavaz D, et al. Release of magnetic nanoparticles from cell-encapsulating biodegradable nanobiomaterials. ACS Nano. 2012; 6:6640–6649.

20. Campbell PG, Miller ED, Fisher GW, Walker LM, Weiss LE. Engineered spatial patterns of FGF-2 immobilized on fibrin direct cell organization. Biomaterials. 2005; 26:6762–6770.

21. Gruene M, Pflaum M, Hess C, Diamantouros S, Schlie S, Deiwick A, et al. Laser printing of three-dimensional multicellular arrays for studies of cell-cell and cell-environment interactions. Tissue Eng Part C Methods. 2011; 17:973–982.

22. Xu T, Baicu C, Aho M, Zile M, Boland T. Fabrication and characterization of bio-engineered cardiac pseudo tissues. Biofabrication. 2009; 1:035001.

23. Binder KW, Zhao W, Aboushwareb T, Dice D, Atala A, Yoo JJ. In situ bioprinting of the skin for burns. J Am Coll Surg. 2010; 211:7.

24. Khalil S, Sun W. Biopolymer deposition for freeform fabrication of hydrogel tissue constructs. Mater Sci Eng: C. 2007; 27:469–478.

25. Cui X, Dean D, Ruggeri ZM, Boland T. Cell damage evaluation of thermal inkjet printed Chinese hamster ovary cells. Biotechnol Bioeng. 2010; 106:963–969.

26. Xu T, Jin J, Gregory C, Hickman JJ, Boland T. Inkjet printing of viable mammalian cells. Biomaterials. 2005; 26:93–99.

27. Mironov V, Trusk T, Kasyanov V, Little S, Swaja R, Markwald R. Biofabrication: a 21st century manufacturing paradigm. Biofabrication. 2009; 1:022001.

28. Muradoglu M, Tasoglu S. Impact and Spreading of a Microdroplet on a Solid Wall. In : ASME 2009 7th International Conference on Nanochannels, Microchannels, and Minichannels: American Society of Mechanical Engineers; 2009. p. 1095–1106.
29. Mironov V, Markwald RR, Forgacs G. ORGAN PRINTING: SELF-ASSEMBLING CELL AGGREGATES AS "BIOINK". Sci Med. 2003; 9:69–71.
30. Reddi AH. Morphogenesis and tissue engineering. In : Lanza RP, Langer R, Vacanti J, editors. Principles of tissue engineering. 2nd ed. San Diego: Academic press;2011. p. 81–92.
31. Mironov V, Prestwich G, Forgacs G. Bioprinting living structures. J Mater Chem. 2007; 17:2054–2060.

32. Leong KG, Wang BE, Johnson L, Gao WQ. Generation of a prostate from a single adult stem cell. Nature. 2008; 456:804–808.

33. Guillemot F, Mironov V, Nakamura M. Bioprinting is coming of age: Report from the International Conference on Bioprinting and Biofabrication in Bordeaux (3B'09). Biofabrication. 2010; 2:010201.

34. Mondy WL, Cameron D, Timmermans JP, De Clerck N, Sasov A, Casteleyn C, et al. Computer-aided design of microvasculature systems for use in vascular scaffold production. Biofabrication. 2009; 1:035002.

35. Norotte C, Marga FS, Niklason LE, Forgacs G. Scaffold-free vascular tissue engineering using bioprinting. Biomaterials. 2009; 30:5910–5917.

36. Duan B, Hockaday LA, Kang KH, Butcher JT. 3D bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. J Biomed Mater Res A. 2013; 101:1255–1264.

37. Duan B, Kapetanovic E, Hockaday LA, Butcher JT. Three-dimensional printed trileaflet valve conduits using biological hydrogels and human valve interstitial cells. Acta Biomater. 2014; 10:1836–1846.

38. Cui X, Breitenkamp K, Finn MG, Lotz M, D'Lima DD. Direct human cartilage repair using three-dimensional bioprinting technology. Tissue Eng Part A. 2012; 18:1304–1312.

39. Zopf DA, Hollister SJ, Nelson ME, Ohye RG, Green GE. Bioresorbable airway splint created with a three-dimensional printer. N Engl J Med. 2013; 368:2043–2045.

40. Fedorovich NE, De Wijn JR, Verbout AJ, Alblas J, Dhert WJ. Three-dimensional fiber deposition of cell-laden, viable, patterned constructs for bone tissue printing. Tissue Eng Part A. 2008; 14:127–133.

41. Zhao Y, Yao R, Ouyang L, Ding H, Zhang T, Zhang K, et al. Three-dimensional printing of Hela cells for cervical tumor model in vitro. Biofabrication. 2014; 6:035001.
42. Fedorovich NE, Schuurman W, Wijnberg HM, Prins HJ, van Weeren PR, Malda J, et al. Biofabrication of osteochondral tissue equivalents by printing topologically defined, cell-laden hydrogel scaffolds. Tissue Eng Part C Methods. 2012; 18:33–44.

43. Phillippi JA, Miller E, Weiss L, Huard J, Waggoner A, Campbell P. Microenvironments engineered by inkjet bioprinting spatially direct adult stem cells toward muscle- and bone-like subpopulations. Stem Cells. 2008; 26:127–134.

44. Baltich J, Hatch-Vallier L, Adams AM, Arruda EM, Larkin LM. Development of a scaffoldless three-dimensional engineered nerve using a nerve-fibroblast co-culture. In Vitro Cell Dev Biol Anim. 2010; 46:438–444.

45. Lee W, Debasitis JC, Lee VK, Lee JH, Fischer K, Edminster K, et al. Multi-layered culture of human skin fibroblasts and keratinocytes through three-dimensional freeform fabrication. Biomaterials. 2009; 30:1587–1595.

46. Reilly GC, Engler AJ. Intrinsic extracellular matrix properties regulate stem cell differentiation. J Biomech. 2010; 43:55–62.

47. Engler AJ, Sweeney HL, Discher DE, Schwarzbauer JE. Extracellular matrix elasticity directs stem cell differentiation. J Musculoskelet Neuronal Interact. 2007; 7:335.
48. Schuh E, Kramer J, Rohwedel J, Notbohm H, Muller R, Gutsmann T, et al. Effect of matrix elasticity on the maintenance of the chondrogenic phenotype. Tissue Eng Part A. 2010; 16:1281–1290.





 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download