Abstract
In the 1970s, surgeons performed a variety of experimental procedures on animals. Experimental surgery has scientific merit and allows surgeons to carry out many studies to understand pathophysiology in several fields of medicine. However, the strong opposition to animal research and the surgeon's increasing demand of clinical activities has threatened the opportunities of performing experimental studies. Although large animals are not permitted, small animals are available to use. The rat is a frequently used species, since it is economical and easy to breed and anesthetize. As a hepatobiliary surgeon, I studied liver resection and liver transplantation in rats. The technique of transplantation is not easy to obtain since it requires skillful microsurgical techniques. However, these techniques help to undergo experimental research and can be used as practice operations before it is actually performed on humans. As the experimental research continues to be an important part of surgery, every possible method should be considered in order to increase support and opportunities for research. The first step will be training young surgeons to enhance research. The second step is redirecting priorities among surgeons. The scientific productivity should be set as first priority for surgeons. Final strategy is to collaborate with basic scientists such as immunologist, biochemist and so on. There is no doubt that major advancement in the field of medicine in the 21st century will continue to arise from research in surgery. Surgeons should seek for more opportunities in good research training, and provide some time for experimental research.
Figures and Tables
 | Fig. 1Outline of the anatomy of the liver of the rat. LLS, left lateral liver; LML, left middle liver; RML, right middle liver; SRL, superior right liver; IRL, inferior right liver; AC, anterior caudate lobe; PC, posterior caudate lobe. |
References
1. Di Cataldo A, Trombatore G, Ciraldo R, Rodolico M, Sorbello M, Li Destri G. Experimental research and surgery: why, how, and when? Microsurgery. 2001; 21:118–120.

2. Di Cataldo A, Li Destri G, Trombatore G, Papillo B, Racalbuto A, Puleo S. Usefulness of microsurgery in the training of the general surgeon. Microsurgery. 1998; 18:446–448.

3. Lee S. Manual of microsurgeryed. CRC Press;1985.
4. Lee S. Historical events on development of experimental microsurgical organ transplantation. Yonsei Med J. 2004; 45:1115–1120.

5. Kogure K, Ishizaki M, Nemoto M, Kuwano H, Makuuchi M. A comparative study of the anatomy of rat and human livers. J Hepatobiliary Pancreat Surg. 1999; 6:171–175.

6. Aller MA, Mendez M, Nava MP, Lopez L, Arias JL, Arias J. The value of microsurgery in liver research. Liver Int. 2009; 29:1132–1140.

7. Martins PN, Theruvath TP, Neuhaus P. Rodent models of partial hepatectomies. Liver Int. 2008; 28:3–11.

8. Higgins GM, Anderson RM. Experimental pathology of the liver: restoration of the white rat following partial surgical removal. Arch Pathol. 1931; 12:186–202.
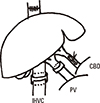
9. Lee S, Edgington TS. Liver transplantation in the rat. Surg Forum. 1966; 17:220–222.
10. Kamada N, Calne RY. Orthotopic liver transplantation in the rat. Technique using cuff for portal vein anastomosis and biliary drainage. Transplantation. 1979; 28:47–50.
12. Panis Y, Nordlinger B, Delelo R, Herve JP, Infante J, Kuhnle M, et al. Experimental colorectal liver metastases. Influence of sex, immunological status and liver regeneration. J Hepatol. 1990; 11:53–57.
13. Rahman TM, Hodgson HJ. Animal models of acute hepatic failure. Int J Exp Pathol. 2000; 81:145–157.

14. Gaub J, Iversen J. Rat liver regeneration after 90% partial hepatectomy. Hepatology. 1984; 4:902–904.

15. Emond J, Capron-Laudereau M, Meriggi F, Bernuau J, Reynes M, Houssin D. Extent of hepatectomy in the rat. Evaluation of basal conditions and effect of therapy. Eur Surg Res. 1989; 21:251–259.
16. Nikfarjam M, Malcontenti-Wilson C, Fanartzis M, Daruwalla J, Christophi C. A model of partial hepatectomy in mice. J Invest Surg. 2004; 17:291–294.

17. Madrahimov N, Dirsch O, Broelsch C, Dahmen U. Marginal hepatectomy in the rat: from anatomy to surgery. Ann Surg. 2006; 244:89–98.
18. Kubota T, Takabe K, Yang M, Sekido H, Endo I, Ichikawa Y, et al. Minimum sizes for remnant and transplanted livers in rats. J Hepatobiliary Pancreat Surg. 1997; 4:398–404.

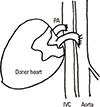
19. Ono K, Lindsey ES. Improved technique of heart transplantation in rats. J Thorac Cardiovasc Surg. 1969; 57:225–229.

20. Xiu D, Uchida H, To H, Sugimoto K, Kasahara K, Nagai H, et al. Simplified method of heterotopic rat heart transplantation using the cuff technique: application to sublethal dose protocol of methotrexate on allograft survival. Microsurgery. 2001; 21:16–21.

21. Kamada N, Calne RY. A surgical experience with five hundred thirty liver transplants in the rat. Surgery. 1983; 93:64–69.
22. Ishii E, Shimizu A, Kuwahara N, Kanzaki G, Higo S, Kajimoto Y, et al. Hepatic Artery Reconstruction Prevents Ischemic Graft Injury, Inhibits Graft Rejection, and Mediates Long-term Graft Acceptance in Rat Liver Transplantation. Transplant Proc. 2013; 45:1748–1753.





 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download