Abstract
As a medical doctor, the final goal of any type of research is focused on better treatment or contemporary care of the patient. As a surgeon, the research is focused on improving results in surgical procedure and improving the immediate to long term outcomes. In human beings, hepatobiliary tract and pancreas (HBP) play a major role in digestion, body metabolism and endocrine function. These organs are sophisticated in pathology as well as anatomy and physiology. The surgery of the hepatobiliary and pancreatic organ is very difficult. Therefore, the hepatobiliary surgeon's endeavor to treat patients who have hepatobiliary and pancreatic diseases should be scientific and based on evidence in order to get the best result.
Here I describe the topic of ischemia/reperfusion injury of the liver from my experience to establish safe surgical resection of the liver with limited bleeding and ischemia/reperfusion injury. Furthermore, I have added ischemia/reperfusion injury in the cirrhotic mouse model, methodology and brief result that I have achieved.
Figures and Tables
 | Fig. 1Protocol of hepatic ischemia/reperfusion of the experimental mouse. (A) Intermittent clamping, five cycles of repeated ischemia for 15 minutes and reperfusion for 5 minutes. (B) Ischemic preconditioning, ischemia for 10 minutes and reperfusion for 15 minutes, then prolonged ischemia for 75 minutes. (C) Control, continuous 75 minutes ischemia. |
 | Fig. 4TUNEL stain. (A) Negative for TUNEL stains, (B) Positive for TUNEL stain, multiple green colored fluorescent stained nuclei are shown, which reflects injured DNA of the hepatocyte nucleus. Ref. 4 with permission from John Wiley and Sons. |
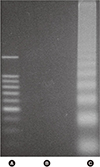 | Fig. 5DNA laddering. Agarose gel electrophoresis of DNA from livers after reperfusion for 3 hours. (A) Known markers are shown comparing the experimental material. (B) Varying ischemic insults showed no DNA laddering in both intermittent clamping. (C) By contrast, the livers subjected to continuous ischemia showed the typical laddering pattern indicating DNA fragmentation. Ref. 7 with permission from Elsevier. |
References
1. Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986; 74:1124–1136.

2. Gao W, Bentley RC, Madden JF, Clavien PA. Apoptosis of sinusoidal endothelial cells is a critical mechanism of preservation injury in rat liver transplantation. Hepatology. 1998; 27:1652–1660.

3. Yadav SS, Sindram D, Perry DK, Clavien PA. Ischemic preconditioning protects the mouse liver by inhibition of apoptosis through a caspase-dependent pathway. Hepatology. 1999; 30:1223–1231.

4. Kang KJ, Jang JH, Lim TJ, Kang Y, Park KK, Lee IS, et al. Optimal cycle of intermittent portal triad clamping during liver resection in the murine liver. Liver Transpl. 2004; 10:794–801.

5. Yadav SS, Gao W, Harland RC, Clavien PA. A new and simple technique of total hepatic ischemia in the mouse. Transplantation. 1998; 65:1433–1436.

6. Rudiger HA, Kang KJ, Sindram D, Riehle HM, Clavien PA. Comparison of ischemic preconditioning and intermittent and continuous inflow occlusion in the murine liver. Ann Surg. 2002; 235:400–407.

7. Jang JH, Kang KJ, Kim YH, Kang YN, Lee IS. Reevaluation of experimental model of hepatic fibrosis induced by hepatotoxic drugs: an easy, applicable, and reproducible model. Transplant Proc. 2008; 40:2700–2703.

8. Jang JH, Kang KJ, Kang Y, Lee IS, Graf R, Clavien PA. Ischemic preconditioning and intermittent clamping confer protection against ischemic injury in the cirrhotic mouse liver. Liver Transpl. 2008; 14:980–988.

9. Sindram D, Porte RJ, Hoffman MR, Bentley RC, Clavien PA. Platelets induce sinusoidal endothelial cell apoptosis upon reperfusion of the cold ischemic rat liver. Gastroenterology. 2000; 118:183–191.

10. Caban A, Oczkowicz G, Abdel-Samad O, Cierpka L. Influence of ischemic preconditioning and nitric oxide on microcirculation and the degree of rat liver injury in the model of ischemia and reperfusion. Transplant Proc. 2006; 38:196–198.

11. Alchera E, Tacchini L, Imarisio C, Dal Ponte C, De Ponti C, Gammella E, et al. Adenosine-dependent activation of hypoxia-inducible factor-1 induces late preconditioning in liver cells. Hepatology. 2008; 48:230–239.

12. Selzner M, Bielawska A, Morse MA, Rudiger HA, Sindram D, Hannun YA, et al. Induction of apoptotic cell death and prevention of tumor growth by ceramide analogues in metastatic human colon cancer. Cancer Res. 2001; 61:1233–1240.
13. Sakamoto Y, Makuuchi M, Takayama T, Minagawa M, Kita Y. Pringle's maneuver lasting 322 min. Hepatogastroenterology. 1999; 46:457–458.




 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download