Abstract
The present study was conducted to investigate the effect of dietary supplementation of grape pomace on lipid peroxidation and related enzyme activities of rats fed high fat diet. Male Sprague-Dawley rats weighing about 90 g were assigned to 4 experimental groups of 8 rats on the basis of their body weight. The high fat diet contained additional 15% lard to AIN 93-based diet. Rats were fed experimental diets containing 5% grape pomace for 4 weeks. Dietary supplementation of grape pomace reduced serum concentration of lipid peroxide in rats fed high fat diet. Hepatic concentration of lipid peroxide tended to be lower by feeding grape pomace. Hepatic total glutathione content and GSH/GSSG ratio were increased by grape pomace feeding in normal or high fat diet groups. Hepatic superoxide dismutase activity of grape pomace group with high fat diet was induced significantly compared with high fat diet group without grape pomace. Hepatic catalase activity of high fat fed rats was induced by feeding grape pomace. Grape pomace diet increased glutathione-S-transferase and glutathione peroxidase activities in rat liver fed high fat. Hepatic glucose-6-phosphatase activity was not affected by dietary supplementation of grape pomace in rats fed high fat. These results suggest that dietary supplementation of grape pomace may alleviate lipid peroxidation through antioxidant effect in rats fed high fat.
Figures and Tables
Fig. 1
Effect of grape pomace diet on serum concentration of lipid peroxide in rats fed normal or high fat diet. Means ± S.D. Values with the same superscript letter are not significantly different (p < 0.05).
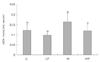
Fig. 2
Effect of grape pomace diet on hepatic homogenate (A) and microsomal (B) concentration of lipid peroxide in rats fed normal or high fat diet. Means ± S.D. Values with the same superscript letter are not significantly different (p < 0.05).

Fig. 3
Effect of grape pomace diet on hepatic level of total glutathione (A) and liver GSH/GSSG ratio (B) in rats fed normal or high fat diet. Means ± S.D. Values with the same superscript letter are not significantly different (p < 0.05).

Table 2
Effect of grape pomace diet on food intake, weight gain and feed efficiency ratio in rate fed normal or high fat diet

1) Groups are the same as the Table 1
2) mean ± SD. Values with the same superscript letter within the column are not significantly different (p < 0.05)
FER: Feed Efficiency Ratio, NS: not siginificant
References
1. 2006 Annual report on the cause of death statistics. 2007. Korea National statistical Office.
2. Agricultural Products Consumption Actual Condition. 2006. Korea Agro-Fisheries Trande Corp.
3. Soleas GJ, Diamandis EP, Goldberg DM. Wine as a biological fluid: history, production, and role in disease prevention. J Clin Lab Anal. 1997. 11(5):287–313.

4. Waddington E, Puddey IB, Croft KD. Red wine polyphenolic compounds inhibit atherosclerosis in apolipoprotein E-deficient mice indepently of effect on lipid peroxidation. Am J Clin Nutr. 2004. 79:54–61.

5. Renaud S, de Lorgeril M. Wine, alcohol, platelets, and the French paradox for coronary heart disease. Lancet. 1992. 339(8808):1523–1526.

6. Ana MGP, Sara ER, Celestino SB, Sonia DPT, Julian CRG. Flavanol content and antioxidant actinity in winery byproducts. J Agric Food Chem. 2004. 52(2):234–238.
7. Amico V, Napoli EM, Renda A, Ruberto G, Spatafora C, Tringali C. Constituents of grape pomace from the sicilian cultivar Nerello Mascales. Food Chem. 2004. 88:599–607.

8. Careri M, Coeeadini C, Elviri L, Nicoletti I, Zagnon I. Lipuid chromatography-electrospray tandem mass spectrometry of cisresveratrol and trans-resveratrol: Development, validation, and application of the method to red wine, grape, and wine making byproducts. J Agric Food Chem. 2004. 52:6868–6874.

9. Baumgartel T, Kluth H, Epperlein K, Rodehutscord M. A note on digestibility and energy value for sheep of different grape pomace. Small Ruminant Res. 2007. 67:302–306.

10. Kammerer D, Claus A, Carle R, Schieber A. Polyphenol screening of pomace from red and white grape varieties by HPLC-DAD-MS/MS. J Agric Food Chem. 2004. 52(14):4360–4367.

11. Jeon SH. Factors affecting resveratrol content in 'Campbell Early' Grape. 2003. Jechon: Chungbuk National University;[Ph.D. dissertation].
12. Harper CE, Patel BB, Wang Jun, Arabshahi A, Eltoum IA, Lamartiniere CA. Resveratrol suppresses prostate cancer progression in transgenic mice. Carcinogenesis. 2007. 28(9):1946–1953.

13. Yilmaz Y, Toledo RT. Health aspects of functional grape seed constituents. Trends Food Sci Technol. 2004. 15:422–433.

14. Olas B, Wachowicz B. Resveratrol, a phenolic antioxidant with effects on blood platelet functions. Platelets. 2005. 16(5):251–260.

15. Marhinez J, Moreno JJ. Effect of resvertrol a natural polyphenoic compound, on reactive oxygen species and prostaglandin production. Biochem Pharmaco. 2000. 59(7):865–870.
16. Leonard S, Chang X, Jiang BH, Stinefelt B, Klandorf H, Harris GK, Shi X. Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responese. Biochem Biophys Re Commun. 2003. 309:1017–1026.

17. Vitseva O, Varghese S, Chakrabarti S, Folts JD, Freedman JE. Grape seed and skin extracts inhibit platelet function and release of reactive oxygen intermediates. J Cardiovasc Pharmacol. 2005. 46(4):445–451.

18. Rho Kyoung-A. Effect of grape intake on antioxidative and antithrombogenic capacity of Cd-administered rats during aging. 2002. Seoul: Ewha Womans University;[Ph.D. dissertation].
19. Yoo MA, Chung HK, Kang MH. Optimal extract methods of antioxidant compounds from coat of grape dreg. Korean J Food Sci Technol. 2004. 36(1):134–140.
20. Philip GR, Forrest NH, George CF. AIN-93 purified diets for laboratory rodents: Final report of the american institute of nutrition AdHoc Writing committee on the reformulation of the AIN76 A rodent diet. J Nutr. 1993. 123(11):1939–1951.
21. Yagi K. A simple fluorometric assay for lipoperoxide in blood plasma. Biochem Med. 1976. 15(2):212–216.

22. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 1979. 95(2):351–358.

23. Akerboom TPM, Sies H. Assay of glutathione, glutathione disulfide, and glutathione mixed disulfides in biological samples. Method in Enzymology. 1981. 77:373–382.
25. Paglia DE, Valentine WN. Studies on quantitative an qualitative charaterization of erythrocyte glutathione peroxidase. Lab Clin Med. 1967. 70(1):158–166.
26. Marklund S, Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem. 1974. 47(3):469–474.

27. Habig WH, Pabst MJ, Jakoby WB. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem. 1974. 249(22):7130–7139.
28. Baginski ES, Foa PP, Zak B. Glucose-6-phosphatase. Methods of Enzymematic Analysis 2. 1983. New York: Academic Press;876–880.

29. Lowry OH, Resebrough NJ, Farr AL, Randall TJ. Protein measurement with folin reagent. J Bio Chem. 1951. 193(1):265–272.
30. Um MY, Kim MK. Effect of grape intakes on lipid metabolism of rats during aging. Korean J Nutr. 2002. 35(7):713–728.
31. Fuhrman B, Volkova N, Coleman R, Aviram M. Grape powder polyphenols attenuate atherosclerosis development in apolipoprotein E deficient (Eo) mice and reduce macrophage atherogenicity. J Nutr. 2005. 135:722–728.

32. Skottova N, Vecera R, Urbanek K, Vana P, Walterova D, Cvak L. Effects of polyphenolic fraction of silymarin on lipoprotein profile in rats fed cholesterol-rich diets. Pharma Res. 2003. 47(1):17–36.

33. Alia M, Horcajo C, Bravo L, Goya L. Effect of grape antioxidant dietary fiber on the total antioxidant capacity and the activity of liver antioxidant enzymes in rats. Nutr Res. 2003. 23:1251–1267.

34. Demrow HS, Slane PR, Folts JD. Administration of wine and grape juice inhibits in vivo platelet activity and thrombosis in stenosed canine coronary arteries. Circulation. 1995. 91(4):1182–1188.

35. Schieber A, Stintzing FC, Carle R. By-products of plant food processing as a source of functional compounds-recent developments. Trends Food Sci Technol. 2001. 12:401–413.

36. Bobek P. Dietary tomato and grape pomace in rat: effect on lipid in serum and liver, and on antioxidant status. Brit J Biomed Sci. 1999. 56(2):109–113.
37. Johan JP, Joenje H. Vigo-Pelfrey C, editor. Biological significance of oxygen toxicity: An introduction. Membrane lipid oxidation Volume III. 1993. Florida: CRC Press;7–8.
38. Llobera A, Canellas J. Dietary fibre content and antioxidant activity of manto negro red grape (Vitis vinifera): pomace and stem. Food Chem. 2007. 101:659–666.

39. Reiter RJ, Tan DX, Gitto E, Sainz RM, Mayo JC, Leon J, Manchester LC, Vijaylaxmi , Kilic E, Kilic U. Pharmacological utility of melatonin in reducing oxidative cellular and molecular damage. Pol J Pharmacol. 2004. 56:159–170.




 PDF
PDF ePub
ePub Citation
Citation Print
Print




 XML Download
XML Download