Abstract
The severe shortage of human organ donors is one of the biggest problems with organ transplantation. The solution for this problem would be development of artificial organs or mechanical devices, stem cell derived organs, and xenogeneic organs. Artificial organs may provide a short term life or functional support, but they cannot be considered as a life-long curative therapeutic modality in the near future. Although considerable efforts have been invested in the production of lab-grown organs using stem cells, clinical application of these organs will demand many years of research and investment. Currently, stem cells are clinically applied in cell replacement therapy. Therefore, xenotransplantation would be the most imminent solution for the organ shortage. Recent advances in our understanding of the mechanisms of xenograft rejection, zoonotic infections including PERV (porcine endogenous retrovirus), and production of α-1,3-galactosyltransferase-deficient pigs, put xenotransplantation closer to the clinical reality. At this stage, pancreatic islet xenotransplantation would be the first target for clinical application, the efficacy of which has been proven in non-human primate study and is waiting for the development of relatively non-toxic or clinically applicable immunosuppressive or tolerance-inducing regimens.
Figures and Tables
Figure 1
SNU miniature swine is breeding to meet the PHS guidelines for xenograft donor in Center for Animal Resource Development at Seoul National University College of Medicine under the qualified specific pathogen free condition. Gnotobiotic condition is established through hysterectomy followed by breeding in germ-free isolator.

Figure 2
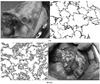
Histophathologic finding of AHXR in the pig lung 1 hour post transplantation to to beagle dog. Massive hemorrhage, thrombosis, edema and infiltration of inflammatory cells were observed. As both pig and dog express α1,3-Gal antigen, the antibody mediated rejection could be caused by anti-non-Gal antibodies.
References
1. Yang YG, Sykes M. Xenotransplantation: current status and a perspective on the future. Nature reviews. 2007. 7:519–531.

2. Pratschke J, Wilhelm MJ, Kusaka M, Basker M, Cooper DK, Hancock WW, Tilney NL. Brain death and its influence on donor organ quality and outcome after transplantation. Transplantation. 1999. 67:343–348.

3. Laskowski I, Pratschke J, Wilhelm MJ, Gasser M, Tilney NL. Molecular and cellular events associated with ischemia/reperfusion injury. Ann Transplant. 2000. 5:29–35.
4. Salama AD, Delikouras A, Pusey CD, Cook HT, Bhangal G, Lechler RI, Dorling A. Transplant accommodation in highly sensitized patients: a potential role for Bcl-xL and alloantibody. Am J Transplant. 2001. 1:260–269.

5. Cooper DK, Gollackner B, Sachs DH. Will the pig solve the transplantation backlog? Annual review of medicine. 2002. 53:133–147.

7. Fishman JA. Infection and xenotransplantation. Developing strategies to minimize risk. Annals of the New York Academy of Sciences. 1998. 862:52–66.

8. Setcavage TM, Kim YB. Variability of the immunological state of germfree colostrum-deprived Minnesota miniature piglets. Infection and immunity. 1976. 13:600–607.

9. Galili U, Rachmilewitz EA, Peleg A, Flechner I. A unique natural human IgG antibody with anti-alpha-galactosyl specificity. The Journal of experimental medicine. 1984. 160:1519–1531.

10. Good AH, Cooper DK, Malcolm AJ, Ippolito RM, Koren E, Neethling FA, Ye Y, Zuhdi N, Lamontagne LR. Identification of carbohydrate structures that bind human antiporcine antibodies: implications for discordant xenografting in humans. Transplantation proceedings. 1992. 24:559–562.
11. Galili U. Interaction of the natural anti-Gal antibody with alpha-galactosyl epitopes: a major obstacle for xenotransplantation in humans. Immunology today. 1993. 14:480–482.

12. Shimizu I, Smith NR, Zhao G, Medof E, Sykes M. Decay-accelerating factor prevents acute humoral rejection induced by low levels of anti-alphaGal natural antibodies. Transplantation. 2006. 81:95–100.

13. Gollackner B, Goh SK, Qawi I, Buhler L, Knosalla C, Daniel S, Kaczmarek E, Awwad M, Cooper DK, Robson SC. Acute vascular rejection of xenografts: roles of natural and elicited xenoreactive antibodies in activation of vascular endothelial cells and induction of procoagulant activity. Transplantation. 2004. 77:1735–1741.

14. Ramírez P, Montoya MJ, Ríos A, García Palenciano C, Majado M, Chávez R, Muñoz A, Fernández OM, Sánchez A, Segura B, Sansano T, Acosta F, Robles R, Sánchez F, Fuente T, Cascales P, González F, Ruiz D, Martínez L, Pons JA, Rodríguez JI, Yélamos J, Cowan P, d'Apice A, Parrilla P. Prevention of hyperacute rejection in a model of orthotopic liver xenotransplantation from pig to baboon using polytransgenic pig livers (CD55, CD59, and H-transferase). Transplantation proceedings. 2005. 37:4103–4106.

15. Schulte am Esch J 2nd, Rogiers X, Robson SC. Molecular incompatibilities in hemostasis between swine and men-impact on xenografting. Ann Transplant. 2001. 6:12–16.
16. Lee KF, Salvaris EJ, Roussel JC, Robson SC, d'Apice AJ, Cowan PJ. Recombinant pig TFPI efficiently regulates human tissue factor pathways. Xenotransplantation. 2008. 15:191–197.

17. Davila E, Byrne GW, LaBreche PT, McGregor HC, Schwab AK, Davies WR, Rao VP, Oi K, Tazelaar HD, Logan JS, McGregor CG. T-cell responses during pig-to-primate xenotransplantation. Xenotransplantation. 2006. 13:31–40.

18. Kuwaki K, Tseng YL, Dor FJ, Shimizu A, Houser SL, Sanderson TM, Lancos CJ, Prabharasuth DD, Cheng J, Moran K, Hisashi Y, Mueller N, Yamada K, Greenstein JL, Hawley RJ, Patience C, Awwad M, Fishman JA, Robson SC, Schuurman HJ, Sachs DH, Cooper DK. Heart transplantation in baboons using alpha1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nature medicine. 2005. 11:29–31.

19. Forte P, Lilienfeld BG, Baumann BC, Seebach JD. Human NK cytotoxicity against porcine cells is triggered by NKp44 and NKG2D. J Immunol. 2005. 175:5463–5470.

20. Yi S, Hawthorne WJ, Lehnert AM, Ha H, Wong JK, van Rooijen N, Davey K, Patel AT, Walters SN, Chandra A, O'Connell PJ. T cell-activated macrophages are capable of both recognition and rejection of pancreatic islet xenografts. J Immunol. 2003. 170:2750–2758.

21. Wang H, VerHalen J, Madariaga ML, Xiang S, Wang S, Lan P, Oldenborg PA, Sykes M, Yang YG. Attenuation of phagocytosis of xenogeneic cells by manipulating CD47. Blood. 2007. 109:836–842.

22. Taniguchi S, Neethling FA, Korchagina EY, Bovin N, Ye Y, Kobayashi T, Niekrasz M, Li S, Koren E, Oriol R, Cooper DK. In vivo immunoadsorption of antipig antibodies in baboons using a specific Gal (alpha)1-3Gal column. Transplantation. 1996. 62:1379–1384.

23. Ye Y, Neethling FA, Niekrasz M, Koren E, Richards SV, Martin M, Kosanke S, Oriol R, Cooper DK. Evidence that intravenously administered alpha-galactosyl carbohydrates reduce baboon serum cytotoxicity to pig kidney cells (PK15) and transplanted pig hearts. Transplantation. 1994. 58:330–337.

24. Alwayn IP, Xu Y, Basker M, Wu C, Buhler L, Lambrigts D, Treter S, Harper D, Kitamura H, Vitetta ES, Abraham S, Awwad M, White-Scharf ME, Sachs DH, Thall A, Cooper DK. Effects of specific anti-B and/or anti-plasma cell immunotherapy on antibody production in baboons: depletion of CD20- and CD22-positive B cells does not result in significantly decreased production of anti-alphaGal antibody. Xenotransplantation. 2001. 8:157–171.

25. Leventhal JR, Dalmasso AP, Cromwell JW, Platt JL, Manivel CJ, Bolman RM 3rd, Matas AJ. Prolongation of cardiac xenograft survival by depletion of complement. Transplantation. 1993. 55:857–865. discussion 65-66.

26. Kang HJ, Lee G, Kim JY, Lee SH, Wi HC, Hwang PG, Chung DH, Kim YT. Pre-treatment of donor with 1-deamino-8-darginine vasopressin could alleviate early failure of porcine xenograft in a cobra venom factor treated canine recipient. Eur J Cardiothorac Surg. 2005. 28:149–156.

27. Lambrigts D, Sachs DH, Cooper DK. Discordant organ xenotransplantation in primates: world experience and current status. Transplantation. 1998. 66:547–561.
28. Cowan PJ, Aminian A, Barlow H, Brown AA, Dwyer K, Filshie RJ, Fisicaro N, Francis DM, Gock H, Goodman DJ, Katsoulis J, Robson SC, Salvaris E, Shinkel TA, Stewart AB, d'Apice AJ. Protective effects of recombinant human antithrombin III in pig-to-primate renal xenotransplantation. Am J Transplant. 2002. 2:520–525.

29. Byrne GW, Schirmer JM, Fass DN, Teotia SS, Kremers WK, Xu H, Naziruddin B, Tazelaar HD, Logan JS, McGregor CG. Warfarin or low-molecular-weight heparin therapy does not prolong pig-to-primate cardiac xenograft function. Am J Transplant. 2005. 5:1011–1020.

30. Dwyer KM, Robson SC, Nandurkar HH, Campbell DJ, Gock H, Murray-Segal LJ, Fisicaro N, Mysore TB, Kaczmarek E, Cowan PJ, d'Apice AJ. Thromboregulatory manifestations in human CD39 transgenic mice and the implications for thrombotic disease and transplantation. The Journal of clinical investigation. 2004. 113:1440–1446.

31. Lai L, Kolber-Simonds D, Park KW, Cheong HT, Greenstein JL, Im GS, Samuel M, Bonk A, Rieke A, Day BN, Murphy CN, Carter DB, Hawley RJ, Prather RS. Production of alpha-1,3-galactosyltransferase knockout pigs by nuclear transfer cloning. Science. 2002. 295:1089–1092.

32. Yamada K, Yazawa K, Shimizu A, Iwanaga T, Hisashi Y, Nuhn M, O'Malley P, Nobori S, Vagefi PA, Patience C, Fishman J, Cooper DK, Hawley RJ, Greenstein J, Schuurman HJ, Awwad M, Sykes M, Sachs DH. Marked prolongation of porcine renal xenograft survival in baboons through the use of alpha1,3-galactosyltransferase gene-knockout donors and the cotransplantation of vascularized thymic tissue. Nature medicine. 2005. 11:32–34.

33. Crikis S, Cowan PJ, d'Apice AJ. Intravascular thrombosis in discordant xenotransplantation. Transplantation. 2006. 82:1119–1123.

34. Groth CG, Korsgren O, Tibell A, Tollemar J, Möller E, Bolinder J, Ostman J, Reinholt FP, Hellerström C, Andersson A, Simeonovic CJ. Xenogeneic islet transplantation. Xenotransplantation. 1999. 6:1–5.

35. Rood PP, Buhler LH, Bottino R, Trucco M, Cooper DK. Pig-to-nonhuman primate islet xenotransplantation: a review of current problems. Cell transplantation. 2006. 15:89–104.

36. Groth CG, Korsgren O, Tibell A, Tollemar J, Möller E, Bolinder J, Ostman J, Reinholt FP, Hellerström C, Andersson A. Transplantation of porcine fetal pancreas to diabetic patients. Lancet. 1994. 344:1402–1404.

37. Bennet W, Groth CG, Larsson R, Nilsson B, Korsgren O. Isolated human islets trigger an instant blood mediated inflammatory reaction: implications for intraportal islet transplantation as a treatment for patients with type 1 diabetes. Upsala journal of medical sciences. 2000. 105:125–133.

38. Bennet W, Sundberg B, Lundgren T, Tibell A, Groth CG, Richards A, White DJ, Elgue G, Larsson R, Nilsson B, Korsgren O. Damage to porcine islets of Langerhans after exposure to human blood in vitro, or after intraportal transplantation to cynomologus monkeys: protective effects of sCR1 and heparin. Transplantation. 2000. 69:711–719.
39. Goto M, Johansson H, Maeda A, Elgue G, Korsgren O, Nilsson B. Low molecular weight dextran sulfate prevents the instant blood-mediated inflammatory reaction induced by adult porcine islets. Transplantation. 2004. 77:741–747.

40. Cardona K, Korbutt GS, Milas Z, Lyon J, Cano J, Jiang W, Bello-Laborn H, Hacquoil B, Strobert E, Gangappa S, Weber CJ, Pearson TC, Rajotte RV, Larsen CP. Long-term survival of neonatal porcine islets in nonhuman primates by targeting costimulation pathways. Nature medicine. 2006. 12:304–306.

41. Hering BJ, Wijkstrom M, Graham ML, Hårdstedt M, Aasheim TC, Jie T, Ansite JD, Nakano M, Cheng J, Li W, Moran K, Christians U, Finnegan C, Mills CD, Sutherland DE, Bansal-Pakala P, Murtaugh MP, Kirchhof N, Schuurman HJ. Prolonged diabetes reversal after intraportal xenotransplantation of wild-type porcine islets in immunosuppressed nonhuman primates. Nature medicine. 2006. 12:301–303.

42. Kim JH, Kim HI, Lee KW, Yu JE, Kim SH, Park HS, Park CG, Ihm SH, Ha J, Kim SJ, Lee HK, Ahn C, Park KS. Influence of strain and age differences on the yields of porcine islet isolation: extremely high islet yields from SPF CMS miniature pigs. Xenotransplantation. 2007. 14:60–66.

43. Fishman JA, Patience C. Xenotransplantation: infectious risk revisited. Am J Transplant. 2004. 4:1383–1390.

44. Garkavenko O, Muzina M, Muzina Z, Powels K, Elliott RB, Croxson MC. Monitoring for potentially xenozoonotic viruses in New Zealand pigs. Journal of medical virology. 2004. 72:338–344.

45. Rowinski W. Future of transplantation medicine. Ann Transplant. 2007. 12:5–10.
46. Ott HC, Matthiesen TS, Goh SK, Black LD, Kren SM, Netoff TI, Taylor DA. Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart. Nature medicine. 2008. 14:213–221.

47. Biotech Policy Research Center. Report for patent map. Trend in industry. 2007. 45.
48. Yamanaka S. Pluripotency and nuclear reprogramming. Philos Trans R Soc Lond B Biol Sci. 2008. 363:2079–2087.

49. Rowinski W. Future of transplantation medicine. Ann Transplant. 2007. 12:5–10.
50. Mimeault Murielle, Batra Surinder. K.. Recent Progress on Tissue-Resident Adult Stem Cell Biology and Their Therapeutic Implications. Stem Cell Rev. 2008. 4:27–49.

51. Bryder D, Rossi DJ, Weissman IL. Hemato-poietic stem cells: the paradigmatic tissue-specific stem cell. American Journal of Pathology. 2006. 169:338–346.
52. Schaffler A, Buchler C. Concise review: adipose tissue-derived stromal cells-basic and clinical implications for novel cell-based therapies. Stem Cells. 2007. 25:818–827.

53. Dhawan J, Rando TA. Stem cells in postnatal myogenesis: molecular mechanisms of satellite cell quie-scence, activation and replenishment. Trends in Cell Biology. 2005. 15:666–673.

54. Watts C, McConkey H, Anderson L, Caldwell M. Anatomical perspectives on adult neural stem cells. Journal of Anatomy. 2005. 207:197–208.

55. Lim DA, Huang YC, Alvarez-Buylla A. The adult neural stem cell niche: lessons for future neural cell replacement strategies. Neurosurgery Clinics of North America. 2007. 18:81–92.

56. Koblas T, Zacharovová K, Berková Z, Mindlová M, Girman P, Dovolilová E, Karasová L, Saudek F. Isolation and characterization of human CXCR4- positive pancreatic cells. Folia Biologica (Praha). 2007. 53:13–22.
57. Bonner-Weir S, Weir GC. New sources of pan-creatic beta-cells. Nature Biotechnology. 2005. 23:857–861.
58. Hall B, Andreeff M, Marini F. The participation of mesenchymal stem cells in tumor stroma formation and their application as targeted gene delivery vehicles. Handb Exp Pharmacol. 2007. 180:263–283.
59. Tang J, Xie Q, Pan G, Wang J, Wang M. Mesenchymal stem cells participate in angiogenesis and improve heart function in rat model of myocardial ischemia with reperfusion. Eur J Cardiothorac Surg. 2006. 30:353–361.

60. Studeny M, Marini FC, Champlin RE, Zompetta C, Fidler IJ, Andreeff M. Bone marrow derived mesenchymal stem cells as vehicles for interferon-b delivery into tumors. Cancer Res. 2002. 62:3603–3608.
61. Ringdén O, Uzunel M, Rasmusson I, Remberger M, Sundberg B, Lönnies H, Marschall HU, Dlugosz A, Szakos A, Hassan Z, Omazic B, Aschan J, Barkholt L, Le Blanc K. Mesenchymal stem cells for treatment of therapy-resistant graft-versus-host disease. Transplantation. 2006. 81:1390–1397.

62. Choi SC, Kim GH, Lee SJ, Park E, Yeo CY, Han JK. Regulation of activin/nodal signaling by Rap2-directed receptor trafficking. Dev Cell. 2008. 15:49–61.

63. Yang YJ, Lee HJ, Choi DH, Huang HS, Lim SC, Lee MK. Effect of scoparone on neurite outgrowth in PC12 cells. Neurosci Lett. 2008. 440:14–18. Epub 2008 May 18.

64. SIRT1 regulates apoptosis and Nanog expression in mouse embryonic stem cells by controlling p53 subcellular localization. Cell Stem Cell. 2008. 2:241–251.




 PDF
PDF ePub
ePub Citation
Citation Print
Print





 XML Download
XML Download