Abstract
Moyamoya disease is characterized by bilateral stenosis or occlusion of distal internal carotid artery (ICA) bifurcation including its proximal branches and abnormal vascular network (moyamoya vessel, MMV) in the vicinity of the arterial occlusions. It is the most common pediatric cerebrovascular disease in Eastern Asia, particularly in Korea and Japan. The etiology is still unknown, but much about the pathology from autopsies, factors involved in its pathogenesis, and its genetics have been studied and reported. It may cause ischemic attacks or cerebral infarctions in children and cerebral hemorrhage in adults. Because of its aggressive clinical course in very young children, the need for early detection and treatment has been recognized. Magnetic resonance imaging (MRI)/MR angiography (MRA), cerebral hemodynamic studies, and cerebral angiography are used for the diagnosis. The treatment basically focuses on prevention of further ischemia and infarction through revascularization. Technically, direct and indirect bypass methods are used. The treatment strategy needs to be individualized in each patient. Outcomes of revascularization procedures are excellent in preventing transient ischemic attacks (TIAs) in most patients.
Figures and Tables
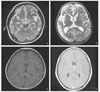 | Figure 1An axial T2-weighted MR image shows diminished flow voids in the internal carotid and middle cerebral arteries (A) and huge cortical infarction in the right hemisphere and left frontal lobe (B). An axial T1-weighted MR image demonstrates punctuate and curvilinear flow voids of the hypertrophied moyamoya collateral in the basal ganglia (C). An axial T1-weighted MR image with gadolinium enhancement reveals prominent leptomeningeal enhancement in the right hemisphere (D). |
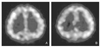 | Figure 2Rest (A) and acetazolamide (B) SPECT images show decreased perfusion with disturbed vasoreactivity to the acetazolamide injection in the right hemisphere. |
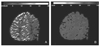 | Figure 3A perfusion MRI demonstrates an increased rCBV (A) and delayed TTP (B) in the right hemisphere. |
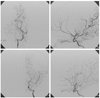 | Figure 4A right carotid angiography (A: AP view, B: lateral view) shows stenotic change in the terminal portion of the internal carotid artery, total occlusion in the middle cerebral artery and stenotic change in the proximal anterior cerebral artery. A left carotid angiography (C: AP view, D: lateral view) reveals stenotic change in the terminal portion of the internal carotid artery and the middle cerebral artery, and total occlusion in the anterior cerebral artery. |
 | Figure 5Operative illustrations of EDAS operation.
A) The skin incision was made along the course of the right STA.
B) The STA-galeal flap laid on the exposed cortex was sutured to the incised edge of the dura mater after dissection of the arachnoid membrane (Modified from (33) with the permission from Ilchokak).
|
 | Figure 6Operative illustrations of bifrontal EG(P)S.
A) S-shaped scalp incision was made 2 cm anterior to the coronal suture.
B) The prepared galeo (-periosteal) flap was inserted into the cerebral cortex and sutured to the dura (Modified from (35) with the permission from Ilchokak).
|
References
1. Suzuki J, Takaku A. Cerebrovascular "moyamoya" disease. Disease showing abnormal net-like vessels in base of brain. Arch Neurol. 1969. 20:288–299.
2. Natori Y, Ikezaki K, Matsushima T, Fukui M. 'Angiographic moyamoya' its definition, classification, and therapy. Clin Neurol Neurosurg. 1997. 99:S2. S168–S172.

3. Ikezaki K, Han DH, Kawano T, Kinukawa N, Fukui M. A clinical comparison of definite moyamoya disease between South Korea and Japan. Stroke. 1997. 28:2513–2517.

5. Nakashima H, Meguro T, Kawada S, Hirotsune N, Ohmoto T. Long-term results of surgically treated moyamoya disease. Clin Neurol Neurosurg. 1997. 99:S2. S156–S161.

6. Ikezaki K. Ikezaki K, Loftus C, editors. Clinical manifestations: epidemiology, symptoms and signs, laboratory findings. Moyamoya Disease. 2001. Rolling Meadows: American Association of Neurological Surgeons;43–54.
7. Wakai K, Tamakoshi A, Ikezaki K, Fukui M, Kawamura T, Aoki R, Kojima M, Lin Y, Ohno Y. Epidemiological features of moyamoya disease in Japan: findings from a nationwide survey. Clin Neurol Neurosurg. 1997. 99:S2. S1–S5.

8. Kim SK, Wang KC, Kim DG, Paek SH, Chung HT, Han MH, Ahn Y, Cho BK. Clinical feature and outcome of pediatric cerebrovascular disease: a neurosurgical series. Childs Nerv Syst. 2000. 16:421–428.

10. Malek AM, Connors S, Robertson RL, Folkman J, Scott RM. Elevation of cerebrospinal fluid levels of basic fibroblast growth factor in moyamoya and central nervous system disorders. Pediatr Neurosurg. 1997. 27:182–189.

11. Hojo M, Hoshimaru M, Miyamoto S, Taki W, Nagata I, Asahi M, Matsuura N, Ishizaki R, Kikuchi H, Hashimoto N. Role of transforming growth factor-beta1 in the pathogenesis of moyamoya disease. J Neurosurg. 1998. 89:623–629.

12. Kim SK, Yoo JI, Cho BK, Hong SJ, Kim YK, Moon JA, Kim JH, Chung YN, Wang KC. Elevation of CRABP-I in the cerebrospinal fluid of patients with Moyamoya disease. Stroke. 2003. 34:2835–2841.

13. Yamada H, Deguchi K, Tanigawara T, Takenaka K, Nishimura Y, Shinoda J, Hattori T, Andoh T, Sakai N. The relationship between moyamoya disease and bacterial infection. Clin Neurol Neurosurg. 1997. 99:S2. S221–S224.

14. Ikezaki K, Kono S, Fukui M. Ikezaki K, Loftus C, editors. Etiology of moyamoya disease: pathology, pathophysiology, and genetics. Moyamoya Disease. 2001. Rolling Meadows: American Association of Neurological Surgeons;55–64.
15. Kang HS, Kim SK, Cho BK, Kim YY, Hwang YS, Wang KC. Single nucleotide polymorphisms of tissue inhibitor of metalloproteinase genes in familial moyamoya disease. Neurosurgery. 2006. 58:1074–1080.

16. Kim SK, Seol HJ, Cho BK, Hwang YS, Lee DS, Wang KC. Moyamoya disease among young patients: its aggressive clinical course and the role of active surgical treatment. Neurosurgery. 2004. 54:840–844. discussion 4-6.

17. Ogawa A, Nakamura N, Yoshimoto T, Suzuki J. Cerebral blood flow in moyamoya disease. Part 2: Autoregulation and CO2 response. Acta Neurochir (Wien). 1990. 105:107–111.

18. Seol HJ, Wang KC, Kim SK, Hwang YS, Kim KJ, Cho BK. Headache in pediatric moyamoya disease: review of 204 consecutive cases. J Neurosurg. 2005. 103:439–442.

19. Ikezaki K, Fukui M, Inamura T, Kinukawa N, Wakai K, Ono Y. The current status of the treatment for hemorrhagic type moyamoya disease based on a 1995 nationwide survey in Japan. Clin Neurol Neurosurg. 1997. 99:S2. S183–S186.

20. Saeki N, Nakazaki S, Kubota M, Yamaura A, Hoshi S, Sunada S, Sunami K. Hemorrhagic type moyamoya disease. Clin Neurol Neurosurg. 1997. 99:S2. S196–S201.

21. Iwama T, Morimoto M, Hashimoto N, Goto Y, Todaka T, Sawada M. Mechanism of intracranial rebleeding in moyamoya disease. Clin Neurol Neurosurg. 1997. 99:S2. S187–S190.

22. Houkin K, Yoshimoto T, Kuroda S, Ishikawa T, Takahashi A, Abe H. Angiographic analysis of moyamoya disease-how does moyamoya disease progress? Neurol Med Chir (Tokyo). 1996. 36:783–787. discussion 8.
23. Kuroda S, Kamiyama H, Isobe M, Houkin K, Abe H, Mitsumori K. Cerebral hemodynamics and "re-build-up" phenomenon on electroencephalogram in children with moyamoya disease. Childs Nerv Syst. 1995. 11:214–219.

24. Ikezaki K, Matsushima T, Kuwabara Y, Suzuki SO, Nomura T, Fukui M. Cerebral circulation and oxygen metabolism in childhood moyamoya disease: a perioperative positron emission tomography study. J Neurosurg. 1994. 81:843–850.

25. Kim SK, Wang KC, Oh CW, Kim IO, Lee DS, Song IC, Cho BK. Evaluation of cerebral hemodynamics with perfusion MRI in childhood moyamoya disease. Pediatr Neurosurg. 2003. 38:68–75.

26. Kuwabara Y, Ichiya Y, Sasaki M, Yoshida T, Masuda K, Matsushima T, Fukui M. Response to hypercapnia in moyamoya disease. Cerebrovascular response to hypercapnia in pediatric and adult patients with moyamoya disease. Stroke. 1997. 28:701–707.
27. Seol HJ, Wang KC, Kim SK, Lee CS, Lee DS, Kim IO, Cho BK. Unilateral (probable) moyamoya disease: long-term follow-up of seven cases. Childs Nerv Syst. 2006. 22:145–150.

28. Okada Y, Shima T, Matsumura S, Nishida M, Yamada T, Okita S. [Pathophysiological studies in moyamoya disease by rCBF and cortical artery pressure measurements in comparison to those in ICA or MCA occlusion]. No To Shinkei. 1988. 40:899–903.
29. Matsushima Y, Inaba Y. Moyamoya disease in children and its surgical treatment. Introduction of a new surgical procedure and its follow-up angiograms. Childs Brain. 1984. 11:155–170.
30. Kim SK, Wang KC, Kim IO, Lee DS, Cho BK. Combined encephaloduroarteriosynangiosis and bifrontal encephalogaleo (periosteal)synangiosis in pediatric moyamoya disease. Neurosurgery. 2002. 50:88–96.

31. Kim CY, Wang KC, Kim SK, Chung YN, Kim HS, Cho BK. Encephaloduroarteriosynangiosis with bifrontal encephalogaleo (periosteal) synangiosis in the pediatric moyamoya disease: the surgical technique and its outcomes. Childs Nerv Syst. 2003. 19:316–324.

32. Chung YN, Wang KC, Cho BK. Kim DG, editor. Moyamoya disease, Encephaloduroarteriosynangiosis. Practical Points in Neurosurgery. 2007. Seoul: Ilchokak;385–389.
33. Kim SK, Wang KC, Cho BK. Kim DG, editor. Moyamoya disease, Bifrontal Encephalogaleo (periosteal) synangiosis. Practical Points in Neurosurgery. 2007. Seoul: Ilchokak;390–394.
34. Choi IJ, Hong SH, Cho BK, Wang KC, Kim SK. Encephalo-duro-arterio-synangiosis (EDAS) using occipital artery in children with moyamoya disease. J Korean Neurosurg Soc. 2005. 38:413–418.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download