Abstract
Thioredoxin reductase 1 (TrxR) is a homodimeric selenoenzyme catalyzing thioredoxin (Trx) in an NADPH-dependent manner. With regard to carcinogenesis, these redox proteins have been implicated in cell proliferation, transformation and anti-apoptosis. In the present study, using a hamster cholangiocarcinoma (ChC) model, we evaluated the immunohistochemical expression pattern of TrxR in precancerous lesions and ChCs as well as in normal bile ducts. The goal of this study was to determine the potential role and importance of TrxR in cholangiocarcinogenesis. For the ChC model, we obtained liver tissue specimens with dysplastic bile ducts prior to the development of ChC 8 weeks after initiation of the experiment and ChC samples at 27 weeks. The immunohistochemical analysis showed diffuse cytoplasmic overexpression of TrxR in the dysplastic bile duct epithelial cells as well as in cholangiocarcinoma; this was comparable to the negative or weakly positive in normal and type 1 hyperplastic bile ducts. However, TrxR appeared to be considerably down-regulated in the ChCs when compared to the higher expression observed in the dysplastic bile ducts. Therefore, these results suggest that TrxR overexpression followed by down-regulation might be an important event in cholangiocarcinogenesis, especially at early stages including the cellular transformation of candidate bile ducts. Further studies are however required to determine whether TrxR may be a potential target molecule for chemoprevention against cholangiocarcinogenesis. In addition, the molecular mechanism as well as the importance of the loss of TrxR in the development of cholangiocarcinoma, following dysplastic transformation of bile duct cells, also remains to be clarified.
Cholangiocarcinoma (ChC) is a highly malignant epithelial cancer of the biliary tract. [22,23] In humans, primary sclerosing cholangitis, hepatolithiasis, fibropolycystic diseases of the biliary tract, Caroli's disease and liver-fluke infection have been considered as conditions that increase risk for the development of ChC. [9,23] These risk conditions share, as a common feature, long-standing inflammation as well as chronic injury to the biliary tract, which may result in producing harmful reactive oxygen species (ROS) inducing DNA damage and chronically stimulating biliary cell proliferation. [9,17,23] Therefore, it is possible that dysfunction of the intracellular reduction-oxidation (redox) regulatory system may be involved in cholangiocarcinogenesis.
Thioredoxin reductase-1 (TrxR) is a homodimeric selenoenzyme belonging to the flavoprotein family of pyridine nucleotide-disulphide oxidoreductases with mechanistic and sequence identity, including a conserved -Cys-Val-Asn-Val-Gly-Cys- redox catalytic site, to human glutathione reductases. [16,20] TrxR catalyzes its physiological substrate, thioredoxin (Trx), in an NADPH-dependent reaction. [16] The TrxR/Trx couple constitutes a ubiquitous redox system in prokaryotic and eukaryotic cells. [18] Trx has been implicated in a variety of intra- and extracellular processes including regulation of cell proliferation, apoptosis and regulation of transcription factors such as nuclear factor-kappa B (NF-κB), activator protein-1 (AP-1) and p53. [7,18-21] Therefore, TrxR has been considered to play an important role in regulating the cell growth and death and its dysregulation has been closely linked to tumorigenesis. In fact, overexpression of TrxR has been reported in a variety of human primary cancers including: breast cancer, thyroid, prostate, non-small cell lung carcinoma, malignant melanomas and mesothelioma as well as in human cancer cell lines including Jurkat and A549 cells. [2,4,8,13,25] Therefore, TrxR has been considered a potential molecular target for anticancer drugs. [1,3,5,6,10,24] The anti-cancer effects of doxorubicin and anti-tumor effects of quinine, that are commercially available, are achieved by inhibiting TrxR. [14,15] However, the role and importance of TrxR in cholangiocarcinogenesis has not yet been studied.
In the present study, we evaluated the immunohistochemical expression pattern of TrxR in precancerous lesions represented by dysplastic transformation of hyperplastic bile ducts and biliary cancers, as well as in non-tumorigenic bile ducts, type 1 hyperplastic and normal bile ducts, to determine the potential role and importance of TrxR in cholangiocarcinogenesis using a liver-fluke infected hamster ChC model.
Young Syrian golden hamsters, 130 to 150 g in body weight, were purchased from Japan SLC (Japan). They were housed five per polycarbonate cage in a clean rack to which pre-filtered air was supplied and maintained at room temperature (22~26℃) under a 12 h light/dark illumination cycle in an approved animal facility at Kangwon National University in Korea. Animals were given a normal diet (Samyang, Korea) and drinking tap water ad libitum throughout the entire experimental period, except during the (4 weeks) administration of the carcinogen in drinking water. All of the hamsters were included in the experiment after one-week acclimatization.
The hamster ChC model was modified according to a previous study. [12] Twenty-nine Syrian golden hamsters were randomly divided into a control and a ChC model group. On the first day of the experiment, the hamsters in the experimental group were infected with 15 metacercariae of liver fluke, Clonorchis sinensis. One day after the parasite infection, they received 15 ppm of dimethylnitrosamine (DMN; Kasei, Japan) in the drinking water for four weeks with a normal diet. Thereafter, the hamsters were given drinking tap water with a normal diet throughout the rest of the study.
To obtain the precancerous liver tissues with dysplastic bile ducts, we sacrificed five control and seven ChC model hamsters at an interim stage of cholangiocarcinogenesis (eight weeks after initiation of the experiment). Thereafter, the remaining five control and 10 ChC model hamsters were maintained for 19 weeks longer (total 27 weeks) to develop cholangiocarcinoma. Necropsy of the hamsters was then performed under ether anesthesia. We grossly examined the liver of each sacrificed hamster and confirmed the liver-fluke infection by identifying adult parasites in the biliary tracts. The experimental design is depicted in Fig. 1.
Hamster liver tissues obtained at eight weeks and 27 weeks after the initiation of the experiment were fixed in 10% buffered neutral formalin for 48 h. After routine tissue processing, the tissues were embedded in a low-melting-point paraffin. Then 3 µm tissue sections were prepared for hematoxylin and eosin (H&E) staining for histological examination and for immunohistochemistry for the TrxR, respectively.
For immunostaining of TrxR, the avidin biotin complex (ABC) method was used. After deparaffinization and hydration, the liver tissue sections were immersed for 30 min in 100% methanol containing 0.3% hydrogen peroxide (Showa, Japan) to block endogenous peroxidase activity. After washing in distilled water and phosphate buffered saline (PBS, pH 7.2), the sections were microwaved in preheated DAKO antigen retrieval solution (pH 6.0) for 15 min at high power, followed by 30 min detergent treatment at room temperature with 0.05% tween 20 in PBS (pH 7.2). After washing in PBS, the tissue sections were incubated in normal blocking serum provided in the Vectastatin Elite avidin-biotin peroxidase complex immunostaining kits. Avidin D and biotin blocking reagents supplied in the Vector Avidin/Biotin blocking kit were applied to the tissue sections according to the manufacturer's instruction to minimize background staining due to endogenous biotin or biotin-binding proteins, lectins, or nonspecific binding substances present within the tissue sections. The sections were then incubated overnight at 4℃ with a primary antibody for TrxR (1 : 500; Upstate Biologicals, USA). For the negative controls, PBS was applied to the sections instead of a primary antibody. The tissue sections were then incubated for 40 min at room temperature with a biotinylated secondary antibody, followed by a 30 min incubation with Vectastatin Elite ABC reagent (Vector, USA) at room temperature. The specific bindings of antibodies within the tissue sections were visualized with 3,3-diaminobenzidine tetrahydrochloride (Dako, Denmark) solution diluted in PBS, and the sections were then counterstained with Mayer hematoxylin. A selected tissue slide, demonstrating a typical positive reaction, was used as a positive control for every batch of TrxR immunostaining.
Two hamsters died at three weeks after initiation of the experiment during DMN treatment. The livers of the hamsters sacrificed during the interim stage of cholangiocarcinoma (eight weeks after initiation of the experiment) were grossly enlarged and yellowish brown in color with multiple small nodules (1~5 mm) on the surface. The common bile ducts and hepatic bile ducts were severely dilated with the adult liver flukes. Histologically, intrahepatic hyperplastic and dysplastic bile ducts of various sizes and shapes were commonly evident in the periportal areas with prominent fibrosis. The hyperplastic bile ducts had a normal structure lined by simple cuboidal bile duct epithelial cells (Fig. 2B), while dysplastic bile ducts were determined based on their atypical duct structures characterized by irregular lumina, multi-layers of epithelial cells, sometimes showing papillary projection, and transformed individual cellular morphology exemplified by taller and larger epithelial cells with larger and heterochromatic nuclei (Fig. 2C). Mitotic figures were quite often evident in the dysplastic bile ducts. Desmoplastic connective tissue with inflammatory cell infiltration surrounded the proliferating hyperplastic and dysplastic bile ducts. Large intrahepatic and hepatic ducts, severely dilated by the presence of adult parasites, showed epithelial cell proliferation with surrounding desmoplastic reaction of connective tissue in which eosinophils, lymphocytes and plasma cells had massively infiltrated. These histopathological findings were consistently noted in all hamster livers sacrificed at the interim stage of cholangiocarcinogenesis. Only one case out of the seven hamsters sacrificed at the interim stage of cholangiocarcinogenesis developed cholangiocarcinoma.
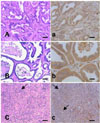
All the hamsters survived up to 27 weeks after initiation of the experiment had developed cholangiocarcinoma in the livers. Histologically, the cholangiocarcinomas that developed were tubular or tubulopapillary cystic types composed of various shapes of tubules or cyst-like structures lined by single to multi-layered tall columnar to cuboidal neoplastic cells and intermittent mucus-producing goblet cells (Fig. 3A-C). The tubules and cysts in general contained relatively large amounts of mucus in their lumina. The tumor cells were aggressively invading adjacent liver parenchyma forming irregular tubular structures or solid sheets of undifferentiated neoplastic cells accompanied by surrounding desmoplastic reaction of connective tissue (Fig. 3A-C). The neoplastic cells had large round nuclei, resulting in a high nucleus/cytoplasm ratio. The supporting connective tissue consisted of bundles of abundant collagen fibers containing interspersed inflammatory cells (Fig. 3A-C).
In the liver tissues at the interim stage of the ChC model, diffuse cytoplasmic overexpression of TrxR was noted in the dysplastic bile duct epithelial cells (Fig. 2c), but the TrxR observed was in general negative or weakly positive in the hyperplastic bile ducts (Fig. 2b). However, at times the immunoreactivity of TrxR in the hyperplastic bile duct epithelial cells was heterogenous and could be negative to strong positive in the same bile duct. In some dysplastic bile ducts, a stronger positive staining was noted at the base of the bile duct cells. The hyperplastic large hepatic bile ducts, containing adult parasites, showed the strongest immunoreactivity for TrxR. The normal bile ducts from the control hamsters were almost negative or very weakly positive for immunoreactivity of TrxR (Fig. 2a).
For all of the tubular types of ChCs that developed, TrxR was diffusely expressed in the cytoplasm of the neoplastic cells (Fig. 3a-c). Compared with the expression level in the dysplastic bile duct cells, TrxR immunoreactivity seemed to be less pronounced in the biliary cancer cells; however, it was still much higher than that observed in normal and type 1 hyperplastic bile ducts. The undifferentiated neoplastic cells forming solid sheets and invading the liver parenchyma totally lost TrxR immunoreactivity in their cytoplasm (Fig. 3c). Type 1 hyperplastic bile ducts showed negative or weakly positive for TrxR, but they were at times strongly positive. Sinusoidal cells showed a strong immunoreactivity for TrxR, while hepatocytes were negative using our immunostaining protocol.
Immunoreactivity of the liver cell components for TrxR is summarized in Table 1.
The critical properties of neoplastic cells include maintaining their ability to proliferate, evading autonomous cell death programs and host-derived immune attacks, and protecting themselves against the harmful subcellular by-products including free radicals and ROS which are excessively generated during hyperactive cellular proliferation. One of the strategies that neoplastic cells use to survive is to utilize the intracellular redox system such as the TrxR/Trx couple, glutathione (GSH)/oxidized glutathione (GSSG) couple and other related enzymes. Trx has been shown to have a variety of intra- and extracellular functions. [18-21] In addition to an antioxidant function, Trx has been implicated in cell proliferation and survival by increasing DNA synthesis in synergy with a number of cytokines and by regulating various transcription factors. [7,18-21] TrxR catalyzes oxidized Trx into reduced Trx in a NADPH-dependent manner, enabling active functioning of Trx. [18-21] Since TrxR has been shown to be highly expressed in some tumors including gastric cancer, [2,4,8,13,25] TrxR has attracted attention for further understanding of its role in carcinogenesis of a variety of cancers as a potential target molecule for treatment. [1,3,5,6,10,24] In the present study, we investigated the expression pattern of TrxR in ChCs and precancerous dysplastic bile ducts in a hamster ChC model.
According to previous studies using a hamster ChC model, ChC usually develops at 16 weeks after initiation of the experiment. [12] Therefore, we considered that at eight weeks an early stage ChC would be present. In the hamster livers sacrificed at eight weeks, proliferating dysplastic bile duct cells were characterized by multi-layered tall cuboidal epithelia forming irregular lumina, frequently with interspersed mucus-secreting cells. The dysplastic bile duct epithelial cells showed strong cytoplasmic expression of TrxR, which was comparable to the negative or weakly positive immunoreactivity of normal and type 1 hyperplastic bile ducts. This unique expression of TrxR in the dysplastic bile ducts may suggest a role of TrxR in the early stages of celluar transformation prior to ChC development. Improved activation of TrxR may increase the opportunity for the transformed bile duct cells to survive cellular apoptosis signaling and continue to proliferate in the microenvironment of the injured biliary cell, which is an inevitable event that progresses to cholangiocarcinogenesis.
Highly expressed TrxR was also evident in the cholangiocarcinomas that developed in the hamster livers sacrificed at 27 weeks in this study. However, the expression level was considerably down-regulated compared with the highly expressed dysplastic bile ducts at the interim stage of cholangiocarcinogenesis. These findings suggest that once a tumor has become established and/or as the tumor advances, highly expressed TrxR appears to diminish. It could not be determined in this study what significant effect such down-regulation of TrxR immunoreactivity could have in association with carcinogenesis; however, it might be associated with the alleviation of inflammation followed by fibrosis surrounding the neoplastic bile ducts.
The expression pattern of TrxR may be one of the characteristics of specific cancers. Depending on the tumor involved, a large difference in TrxR mRNA and protein expression levels has been reported. [11] Loss of TrxR had been demonstrated in colon cancers and the transplanted HT-29 colon cancer cell line, while fibrosarcoma and gastric cancers usually show high expression levels. [11] The TrxR expression level has also been shown to be associated with tumor cell differentiation in specific tumors, as Soini et al. [25] reported a lower expression level of TrxR in high grade non-small cell lung carcinomas. This concept could also be applied to the cholangiocarcinoma, since undifferentiated neoplastic cells forming solid sheets and invading into adjacent liver parenchyma totally lost TrxR immunoreactivity, compared with the mild to moderate overexpression of TrxR in the well-differentiated neoplastic cells. Further study using human cholangiocarcinomas should verify a possible association between TrxR immunoreactivity and tumor cell differentiation grade.
In summary, TrxR was overexpressed in dysplastic bile ducts and cholangiocarcinoma, suggesting a significant role of TrxR in cholangiocarcinogenesis, especially in the early stages including the transformation of candidate bile duct cells. However, down-regulation of TrxR in the biliary cancer cells requires further study to determine whether TrxR may be a potential target molecule for chemoprevention against cholangiocarcinogenesis. In addition, the molecular mechanism and importance of loss of TrxR in the process of the development of cholangiocarcinoma, following dysplastic transformation of bile duct cells, also remains to be clarified.
Figures and Tables
Fig. 1
Experimental protocol. For the hamster cholangiocarcinoma model, 15 metacercariae of Clonorchis sinensis were used for infection one day before initiating DMN treatment. ChC: Cholangiocarcinoma.

Fig. 2
Microphotographs of the liver. A and a, normal bile duct; B and b, type 1 hyperplastic bile ducts; C and c, dysplastic bile ducts. Dysplastic bile ducts and large hyperplastic bile ducts showed strong immunoreactivity for TrxR, while normal (arrow in a) and type 1 hyperplastic bile duct cells (arrow in b) were negative or weakly positive. Dysplastic bile ducts were composed of transformed multi-layered bile duct cells forming irregular lumina. A, B, C; H&E stain, a, b, c; immunohistochemistry for the TrxR. bars = 25 µm.
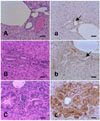
Fig. 3
Microphotographs of cholangiocarcinomas that developed in the hamster model. A and a, tubular type; B and b, tubulocystic type; C and c, undifferentiated type. Immunoreactivity of cholangiocarcinomas for TrxR was down-regulated compared to dysplastic bile ducts (c in Fig. 2); however, it was still much higher than in the normal and type 1 hyperplastic bile duct cells. Expression of TrxR was independent of the classified ChC types except for the undifferentiated type. The neoplastic cells of the undifferentiated type (arrow in C) totally lost TrxR immunoreactivity (arrows in c). A, B, C; H&E stain, bars = 50 µm; a, b, c; immunohistochemistry for TrxR, bars = 25 µm.
Acknowledgments
This study was supported by Kangwon National University and Korea Research Foundation Grant (KRF-2004-041-E00324). The authors wish to thank Dr. Min-Ho Choi, College of Medicine, Seoul National University, for providing Clonorchis sinensis.
References
1. Becker K, Gromer S, Schirmer RH, Müller S. Thioredoxin reductase as a pathophysiological factor and drug target. Eur J Biochem. 2000. 267:6118–6125.

2. Berggren M, Gallegos A, Gasdaska JR, Gasdaska PY, Warneke J, Powis G. Thioredoxin and thioredoxin reductase gene expression in human tumors and cell lines, and the effects of serum stimulation and hypoxia. Anticancer Res. 1996. 16:3459–3466.
3. Biaglow JE, Miller RA. The thioredoxin reductase/thioredoxin system: Novel redox targets for cancer therapy. Cancer Biol Ther. 2005. 4:6–13.

4. Choi JH, Kim TN, Kim S, Baek SH, Kim JH, Lee SR, Kim JR. Overexpression of mitochondrial thioredoxin reductase and peroxiredoxin III in hepatocellular carcinomas. Anticancer Res. 2002. 22:3331–3335.
5. Engman L, Al-Maharik N, McNaughton M, Birmingham A, Powis G. Thioredoxin reductase and cancer cell growth inhibition by organotellurium compounds that could be selectively incorporated into tumor cells. Bioorg Med Chem. 2003. 11:5091–5100.

6. Engman L, Al-Maharik N, McNaughton M, Birmingham A, Powis G. Thioredoxin reductase and cancer cell growth inhibition by organotellurium antioxidants. Anticancer Drugs. 2003. 14:153–161.

7. Gius D, Botero A, Shah S, Curry HA. Intracellular oxidation/reduction status in the regulation of transcription factors NF-kappa B and AP-1. Toxicol Lett. 1999. 106:93–106.

8. Kahlos K, Soini Y, Saily M, Koistinen P, Kakko S, Paakko P, Holmgren A, Kinnula VL. Up-regulation of thioredoxin and thioredoxin reductase in human malignant pleural mesothelioma. Int J Cancer. 2001. 95:198–204.

9. Khan SA, Thomas HC, Davidson BR, Taylor-Robison SD. Cholangiocarcinoma. Lancet. 2005. 366:1303–1314.

10. Kunkel MW, Kirkpatrick DL, Johnson JI, Powis G. Cell line-directed screening assay for inhibitors of thioredoxin reductase signaling as potential anti-cancer drugs. Anticancer Drug Des. 1997. 12:659–670.
11. Lechner S, Müller-Ladner U, Neumann E, Spöttl T, Schlottmann K, Rüschoff J, Schölmerich J, Kullmann F. Thioredoxin reductase 1 expression in colon cancer: discrepancy between in vitro and in vivo findings. Lab Invest. 2003. 83:1321–1331.

12. Lee JH, Rim HJ, Sell S. Heterogeneity of the "oval-cell" response in the hamster liver during cholangiocarcinogenesis following Clonorchis sinensis infection and dimethylnitrosamine treatment. J Hepatol. 1997. 26:1313–1323.

13. Lincoln DT, Ali Emadi EM, Tonissen KF, Clarke FM. The thioredoxin-thioredoxin reductase system: over-expression in human cancer. Anticancer Res. 2003. 23:2425–2433.
14. Mau BL, Powis G. Inhibition of cellular thioredoxin reductase by diaziquone and doxorubicin. Biochem Pharmacol. 1992. 43:1621–1627.

15. Mau BL, Powis G. Mechanism-based inhibition of thioredoxin reductase by antitumor quinoid compounds. Biochem Pharmacol. 1992. 43:1613–1620.

17. Pinlaor S, Hiraku Y, Ma N, Yongvanit P, Semba R, Oikawa S, Murata M, Sripa B, Sithithaworn P, Kawanishi S. Mechanism of NO-mediated oxidative and nitrative DNA damage in hamsters infected with Opisthorchis viverrini: a model of inflammation-mediated carcinogenesis. Nitric Oxide. 2004. 11:175–183.

18. Powis G, Briehl M, Oblong J. Redox signalling and the control of cell growth and death. Pharmacol Ther. 1995. 68:149–173.

19. Powis G, Kirkpatrick DL, Angulo M, Baker A. Thioredoxin redox control of cell growth and death and the effects of inhibitors. Chem Biol Interact. 1998. 111-112:23–34.

20. Powis G, Montfort WR. Properties and biological activities of thioredoxins. Annu Rev Biophys Biomol Struct. 2001. 30:421–455.

21. Powis G, Oblong JE, Gasdaska PY, Berggren M, Hill SR, Kirkpatrick DL. The thioredoxin/thioredoxin reductase redox system and control of cell growth. Oncol Res. 1994. 6:539–544.
22. Sirica AE. Cholangiocarcinoma: Molecular targeting strategies for chemoprevention and therapy. Hepatology. 2005. 41:5–15.

23. Sirica AE, Lai GH, Endo K, Zhang Z, Yoon BI. Cyclooxygenase-2 and ERBB-2 in cholangiocarcinoma: potential therapeutic targets. Semin Liver Dis. 2002. 22:303–313.

24. Smart DK, Ortiz KL, Mattson D, Bradbury CM, Bisht KS, Sieck LK, Brechbiel MW, Gius D. Thioredoxin reductase as a potential molecular target for anticancer agents that induce oxidative stress. Cancer Res. 2004. 64:6716–6724.

25. Soini Y, Kahlos K, Näpänkangas U, Kaarteenaho-Wiik R, Säily M, Koistinen P, Pääkkö P, Holmgren A, Kinnula VL. Widespread expression of thioredoxin and thioredoxin reductase in non-small cell lung carcinoma. Clin Cancer Res. 2001. 7:1750–1757.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download