Abstract
Figures and Tables
Fig. 1
Effect of dexamethasone (A) and lipopolysaccharide (B) on the viability of bovine aortic endothelial cells (BAECs). The cell viability was assessed by MTT assay. The cells were incubated with the treatments for 24 h. Data are presented as mean ± SD of three independent assays with six replicate samples in each assay. Significant differences compared with untreated cells indicated by **p < 0.01, ***p < 0.001 (Turkey's honest significant difference test, one-way ANOVA).

Fig. 2
Quantification of invasion efficiency of bacteria in bovine aortic endothelial cells using real-time polymerase chain reaction assay. The cells were incubated with or without treatments for 24 h and then incubated with bacteria at multiplicity of infection of 100:1 for 2 h. The control was untreated cells and the negative control was DNA of bovine aortic endothelial cells (BAECs). (A) A standard curve was constructed via 10-fold serial dilutions of the extracted genomic DNA of the bacteria from 2.7 × 102 to 2.7 × 10−3 ng. (B) Quantification of bacterial number per cell in different treatments of BAECs. Invasion efficiency is expressed as the bacterial number/BAEC. Data are presented as mean ± SD of two independent assays with at least three replicate samples each. Significant difference from untreated cells indicated by **p < 0.01, ***p < 0.001 (Turkey's honest significant difference test, one-way ANOVA). CFU, colony-forming unit; LPS, lipopolysaccharide; DEXA, dexamethasone.

Fig. 3
Micrographs of Pasteurella multocida B:2 infected dexamethasone-, lipopolysaccharide-treated, and untreated bovine aortic endothelial cells (BAECs). The cells were treated with 10 µM dexamethasone (B) or 100 ng/mL lipopolysaccharide (C) for 24 h, and the treated cells were incubated with P. multocida B:2 for 2 h. The untreated cells (A) were used as the control and (D) showed the P. multocida B:2. Micrographs (E) and (F) are enlargements of micrographs (B) and (C), respectively. The white arrows indicate a bacterium. The bacteria resided in vacuolar compartment of dexamethasone-treated and untreated cells, whereas the bacteria resided within the plasma membrane of lipopolysaccharide-treated cells. The samples were observed under transmission electron microscope (H-7100; Hitachi) operating at 100 kV. Scale bars = 1 µm (D-F); 2 µm (A and B); 5 µm (C).
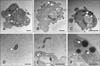
Fig. 4
Images of Pasteurella multocida B:2 infected bovine aortic endothelial cells. The cells were treated with 10 µM DEXA (B, E, H, and K) or 100 ng/mL LPS (C, F, I, and L) for 24 h. The untreated cells (A, D, G, and J) were used as the control. The samples were observed under 60× magnification using an Olympus IX81 microscope. (A-C) Images of cells labeled with DAPI (blue stained nuclei). (D-F) Images of the extracellular bacteria labeled with Alexa Fluor 488 (green stained bacteria). (G-I) Images of intracellular and extracellular bacteria labeled with Alexa Fluor 633 (red stained bacteria). (J-L) Overlapping of the three color images produced images that show the intracellular bacteria (indicated by arrows). Scale bars = 20 µm. DEXA, dexamethasone; LPS, lipopolysaccharide.
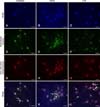
Fig. 5
Effect of dexamethasone (DEXA) and lipopolysaccharide (LPS) on the F- and G-actin pools of bovine aortic endothelial cells. The cells were treated with 10 µM of DEXA or 100 ng/mL of LPS for 24 h. Untreated cells were used as the control. Data are presented as mean ± SD of two independent assays with at least ten replicate samples. Significant differences from untreated cells indicated by ***p < 0.001 (Turkey's honest significant difference test, one-way ANOVA).

Fig. 6
Images of actin filaments of bovine aortic endothelial cells treated with dexamethasone (DEXA) and lipopolysaccharide (LPS). The cells were incubated with 10 µM DEXA or 100 ng/mL LPS for 24 h. The samples were observed under 60× magnification by using an Olympus IX81 microscope. (A-C) Images of F-actin of the cells labeled with Alexa Fluor 488 Phalloidin (green stained fibers). (D-F) Images of G-actin of the cells labeled with Alexa Fluor 594 DNase I (red fluorescence). All cells were labeled with DAPI (blue stained nuclei). Arrows show loss of the filamentous network of F-actin. Scale bars = 20 µm.
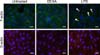
Table 1
Invasion efficiency of Pasteurella multocida B:2 in dexamethasone-, lipopolysaccharide-treated cells and untreated cells

Data are presented as mean ± SD of three independent assays with triplicate samples. Dexamethasone-treated cells showed no significantly different in the invasion efficiencies compared with untreated cells (p > 0.05), whereas the cells treated with 50 and 100 ng/mL of lipopolysaccharide showed significantly higher invasion efficiencies compared with untreated cells (***p < 0.001; Turkey's honest significant difference test, one-way ANOVA). *Invasion efficiency is expressed as the number of bacteria/bovine aortic endothelial cells after incubating the cells with bacteria at multiplicity of infection 100:1 for 2 h.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download