Abstract
This study was conducted to evaluate the effects of induced endometritis on uterine blood flow in cows. Transrectal Doppler sonography was performed on uterine arteries of six cyclic cows before and for 4 days after inducing acute endometritis by intrauterine infusion of 720 mg of policresulen, and for 4 days of the following estrous cycle. Time-averaged maximum velocity (TAMV) increased (p < 0.001) and pulsatility index (PI) decreased (p < 0.0001) within 1 h of policresulen administration, and did not change (p > 0.05) in the next 4 days of the same cycle. TAMV and PI values in the subsequent cycle did not differ (p > 0.05) from the values measured before infusion and showed no changes (p > 0.05) within the cycle. Blood flow parameters were not related (p > 0.05) to plasma concentrations of progesterone and estrogen. All cows showed an acute endometritis determined by histopathological findings of biopsy samples taken 1 day after infusion and fibrotic endometrial alterations detected in the subsequent cycle. No relationships were observed between fibrotic changes of the endometrium and uterine blood flow during either cycle. In conclusion, acute inflammation is accompanied by a rise in uterine blood flow, but fibrotic alterations do not seem to be related to Doppler sonographic findings.
Uterine diseases affect half of all dairy cattle after parturition, causing infertility by disrupting uterine and ovarian function. Financial losses caused by uterine diseases are derived from infertility, increased culling because of failure to conceive, reduced milk production, and costs of treatment [33]. Policresulen (Lotagen Concentrate) is an antiseptic, astringent, hemostatic and tissue regeneration promoter that primarily acts via selective coagulation of the degenerated and necrotic tissue, which causes local reactive hyperemia [21213]. About 50 to 150 mL of a 2 to 4% solution (2–4 parts Lotagen Concentrate and 98–96 parts water), depending on the size of the uterus, can be infused into the uterus of cows affected by chronic purulent endometritis. If infused in healthy tissue, a strong degenerative and inflammatory reaction is observed in the first three days after infusion, and degenerative changes in the epithelia and subepithelial stromal tissues remain until day 10 after infusion [32].
Blood flow plays a key role in inflammation, and one of the most evident characteristics of all inflammatory processes is vasodilation [11]. Circulation of the reproductive tract in healthy and sick cows was initially investigated experimentally using invasive procedures [10]. However, color Doppler sonography has been increasingly used for blood flow studies in the past 15 years [18]. This technique allows a non-invasive investigation of blood flow of the entire reproductive tract. The most common approaches to this technique involve investigation of luteal or follicular blood flow in color mode [27], whereas spectral mode is indicated for the examination of uterine arteries [4]. In cattle, uterine perfusion was investigated by examining blood flow of uterine arteries during the estrous cycle [5], pregnancy [32029], superovulatory regimen [19] and the puerperium [161724].
Color Doppler studies in women have shown that Doppler indices of uterine perfusion are directly related to the state of pelvic inflammatory disease (PID), whether it is acute or cured [2537]. Transvaginal color Doppler ultrasonography of the uterine and ovarian arteries, as well as of arteries at the uterotubal junction in 20 cases of PID revealed decreased vascular resistance in cases of acute infections [1]. Moreover, this method accurately detected regression of infectious processes by an increasing vascular resistance before body temperature and acute phase reactants [1]. Furthermore, transvaginal color Doppler ultrasonography is a reliable method for follow-up of PID treatment, even in women with mild PID, since it can demonstrate a low resistance to blood flow to the uterus before treatment onset, and increasing resistance to blood flow and decreasing level of acute phase reactants after successful treatment [28].
To the best of our knowledge, no studies have investigated the effects of endometrial inflammations on uterine blood flow parameters in cows. Therefore, the present study was conducted to investigate blood flow in uterine arteries after an experimentally induced endometritis during one estrous cycle, and to analyze the consequences of the induced inflammation on the following estrous cycle to determine if there are changes related to acute and chronic histopathological alterations of the endometrium.
The study was conducted at the Clinic for Cattle, University of Veterinary Medicine Hannover, and at the Department of Veterinary Medical Sciences, University of Bologna. The experimental design and animal procedures were carried out in accordance with DL 116/92 and approved by the University of Bologna Ethical Review Committee and the Ministry of Health in Italy. A total of six non-pregnant, non-lactating healthy cows (5 Holstein and 1 Jersey cows), with an average Body Condition Score of 3 (range 2.5 to 3.5) were used. Their mean age was 4.5 years (range 3 to 7 years), and the mean body weight was 610 Kg (range 500 to 750 Kg). One cow was pluriparous (2 pregnancies) and five cows were primiparous. The average time from the last parturition was 8.5 months (range 4 to 24 months). Before the beginning of the experiment, the genital tracts of all cows were monitored to exclude ovarian and uterine pathologies as well as irregular cyclicity. The reproductive examination included manual palpation per rectum, transrectal ultrasonography and vaginoscopy.
The experimental design is summarized in Table 1. As a first step, each cow underwent ultrasonographic examination of the ovaries to establish the presence of a corpus luteum (CL) with a mean diameter of at least 20 mm (Table 1). If a functional CL was present, the experiment started. To induce luteolysis, a PGF2α analogue (Cloprostenol 500 µg/animal intramuscularly [i.m.], Estrumate; MSD Animal Health, Germany) was administered (day 0). A GnRH analogue (Buserelin acetate 10 µg/animal i.m., Receptal; MSD Animal Health) was injected 48 h later to induce ovulation (day 2), which was expected to take place between 24 and 32 h after GnRH analogue injection. On day 3, ovaries were ultrasonographically examined to confirm the presence of a preovulatory follicle, and on day 4 to confirm that ovulation had occurred. On day 6, the first Doppler examination was performed, and this was followed immediately by an intrauterine infusion of 720 mg of policresulen (50 mL of 4% Lotagen concentrate solution, policresulen : metacresolsulfonic acid and formaldehyde condensation product 36% w/v; MSD Animal Health). Doppler examinations were conducted at 1, 12, 24, 48, 72, and 96 h (t1, t12, t24, t48, t72, t96), and the last Doppler examination was followed by PGF2α administration to induce luteolysis. Two days later (day 12), GnRH analogue was injected to obtain a second ovulation (day 13). On day 16, a second sequence of Doppler examinations, without any uterine infusion, was started at the same time as the estrous cycle (day 2), as in the first cycle (at 0, 1, 12, 24, 48, 72, and 96 h, corresponding to t0, t1, t12, t24, t48, t72, t96, respectively).
On day 20, the PGF2α analogue was administered, followed by GnRH analogue on day 22. On the last day of the experiment (day 24), the ovaries were checked by ultrasound for the occurrence of the third induced ovulation. Additionally, blood samples were collected at the time of each Doppler examination, and endometrial biopsies were carried out on days 3, 7, 10, 13, 17, 20, and 23 of the experiment.
During Doppler examination and biopsy collection, animals were restrained in stocks, and an epidural anesthesia was performed by injecting a local anesthetic solution (Isocain 2%; Selectavet, Germany; Lidocain 2%; Fort Dodge Animal Health, Italy). The volume of the anesthetic ranged from 3 to 5 mL, depending on the size of the cow.
All sonographic examinations were performed by the same operator (LD) using a Toshiba SSA-370A ultrasonic device (Toshiba, Japan) equipped with a 7.0 MHz microconvextransducer (PVF-738-F; Toshiba). The examinations were digitalized and registered with a mobile video recorder (ARCHOS AV 740; ARCHOS, Germany) connected to the ultrasound machine. When only B-mode imaging was required, a portable LOGIQ Book XP ultrasound device (GE Healthcare, Germany) equipped with a linear probe (6 to 10 MHz, Model 1739-RS; GE Yokogawa Medical Systems, Japan) was used.
Each Doppler examination lasted about 45 min. Uterine blood flow was studied by examining both uterine arteries, which were found as described earlier [4]. Briefly, with the ultrasound transducer oriented transversely and faced dorsally, the aorta was found and followed caudally until its branching. At this level, the external iliac artery was identified, coursing caudo-ventrally next to the body of the ileum. Proceeding caudally, the arteria iliaca interna was observed. A common stem for the anulus umbilicalis and the arteria uterina was found about 4 cm caudal to the origin of the arteria iliaca interna from its ventral wall. Because the anulus umbilicalis is only canalized in its first 5 cm, the arteria uterina is the functional continuation of the anulus umbilicalis, and the umbilical artery gives rise only to a small vessel supplying the ureter. The uterine artery can be found in the mesometrium as a movable arterial vessel. Near its origin from the umbilical artery, the arteria uterina can be visualized by the color Doppler technique.
The device was then set in spectral mode and the size of the Doppler gate, which determines the location and the area from which Doppler information is processed and displayed, was adjusted to the diameter of the arteria uterina. To remove signals from slowly moving tissues and vessel movement in the path of the Doppler ultrasound pulse, the high-pass filter was set at 100 Hz. All blood flow velocity waveforms were obtained at an interrogation angle between the Doppler ultrasound beam and flow direction ranging from 20 to 60 degrees, and the observations were displayed on-line. Each Doppler examination consisted of one pair of consecutive, similar flow velocity waveforms, with maximum end-diastolic shift. After obtaining each pair of waves, the ultrasound transducer was only turned on its transverse axis, without moving it in any linear direction. This was done to display a transversal, circular cross section of the blood vessel without losing the point at which the waveforms were previously measured. From this position, a mean value of the transverse diameter was obtained by measuring the diameter of the vessel in frozen B-mode images three times for each pair of waves (Fig. 1).
The analysis of pairs of similar consecutive velocity waveforms is based on the envelope of the Doppler shift spectrum. To quantify the information obtained from the ultrasound device, a computer-assisted image analysis program (PixelFlux ver. 2.0; Chameleon Software, Germany) was used. This program operates on ultrasound screenshot images. Therefore, to evaluate the data, it was necessary to obtain suitable screenshot images of the elected waveforms from the digitalized video record of the Doppler examination using the optimal resolution for the PixelFlux software, which was accomplished using high-resolution video player software (PowerDVD 13 Pro; CyberLink, Taiwan). The following variables were determined: time-averaged maximum velocity over the cardiac cycle (TAMV) and pulsatility index (PI). TAMV was calculated from the time-averaged maximum frequency shift (TAMF) using the following equation: TAMV = (TAMF × c)/(2F × cosα), where c = ultrasound propagation speed, F = transmitted wave frequency and α = angle between the ultrasound beam and the blood flow direction. The PI index was calculated as the ratio of the difference between peak systolic frequency shift (PSF) and minimum-diastolic frequency shift (MDF) to the TAMF over the cardiac cycle: PI = (PSF - MDF)/TAMF.
On day 6, 720 mg of policresulen (50 mL of a 4% Lotagen Concentrate solution) were infused into the uterine lumen using a two-way Foley (18 G) catheter. The catheter was passed through the cervix and the tip was placed in the uterine body, caudal to the bifurcation of the uterine horns. The balloon was inflated with air (20 mL), policresulen was infused and the uterus massaged transrectally to distribute the solution homogeneously between uterine horns. Ten minutes later, the catheter was removed, by emptying the balloon and slightly pulling.
During the first estrous cycle, collection of endometrial biopsy samples was performed 3 days before, and 1, 4 and 7 days after policresulen infusion. The same schedule of sampling was carried out during the second cycle, without policresulen infusion (the last biopsy of the first cycle corresponded to the first one of the second cycle). Before each biopsy, the animals received an epidural anesthesia (Isocain 2%; Selectavet; Lidocain 2%; Fort Dodge Animal Health). Cows were kept in a stanchion for the whole time of the procedure. A sterile endometrial biopsy forceps (Hauptner, Germany) was introduced into the uterus through the cervix by transrectal manipulation. Endometrial tissue slices of about 1.5 cm in length were then cut from the mucosa of each horn and transferred to tubes pre-loaded with 4% neutral phosphate buffered formaldehyde solution. The formalin-conserved endometrial samples were sent to the Institute of Pathology of the Veterinary Medicine University of Leipzig. Tissues were embedded in Paraplast Plus (Fisher Scientific, USA) after a standard procedure in Hypercenter XP (a computer-controlled, fluid transfer tissue processor with automated fixation, dehydration, cleaning and paraffin infiltration features for tissue specimens) (Shandon, Germany), sectioned into 3 to 4 µm thick slices with a rotary microtome (Reichert-Jung; Reichert, Austria), and stained with hematoxylin and eosin. Pappenheim's panoptic staining (May-Grünwald-Giemsa staining) was used to identify neutrophilic and eosinophilic granulocytes and mast cells. Neutrophils showed a homogeneous pink cytoplasm, while eosinophils showed finely granular cytoplasm stained dark red, and mast cells of cytoplasm were characterized by a violet finely granular reaction product. Endometrial samples were considered suitable for examination when more than 50% of the tissue came from the glandular layer, and when myometrium, blood clots or squeezing artefacts due to the collection method were absent or only slightly present. All endometrial samples were investigated for the presence of histopathological alterations. The degree and character of histopathological alterations, especially endometritis and endometriosis, were documented. The histopathological criteria for diagnosis are listed in Table 2 to 4.
After each Doppler examination, a blood sample of 3 mL was collected from the coccygeal vein using a vacuum container (Vacutainer precision glide; Becton, Dickinson and Company, Switzerland). Samples were centrifuged at 750 × g for 15 min, after which plasma was transferred to an Eppendorf tube and stored at -20℃ until evaluation.
Steroid hormone concentrations were estimated by enzyme immunoassays as previously described [2630]. Briefly, the plasma progesterone concentration (P4) was measured directly in 20 µL plasma (enzyme-linked antigen: progesterone-3-carboxymethyloxim-horseradishperoxidase; antibody: clone 2H4; Sigma-Aldrich, Germany). Total estrogen (E) analysis was conducted after ether extraction (30% butyl-methyl ether and 70% petrol ether), using 17β-estradiol-hemisuccinatehorseradishperoxidase as an enzyme-linked antigen and E2/3 pool 1 (Professor HHD Meyer, Institute for Physiology, TU Munich, Freising-Weihenstephan, Germany) as antibody, which reacts with estradiol-17β (100%), estrone (100%) and estradiol-17α (66%). The sensitivity of the assays was 0.4 pg/mL for P4 and 0.12 pg/well for E. The intra- and inter-assay coefficients of variation of both assays were < 10%.
Statistical analyses were carried out using SPSS Statistics (ver. 21; IBM, Italy). All data were checked for normality using the Shapiro-Wilk test. TAMV and PI of the left and right uterine artery were compared using the correlation coefficient and a paired Student's t-test or the paired Wilcoxon's signed-rank-test depending on the distribution. Correlations between parameters were calculated using a Spearman's R test. All blood flow measurements were subjected to repeated measures GLM and to a multiple variable GLM taking into account the between-animal variance component over time. Bonferroni's test was used to determine differences in repeated measurements, and Tukey's HSD test was conducted to determine differences in measurements between cows and times of measurements in uterine blood flow. Hormone levels were analyzed using a repeated measures GLM, and P4 and E2 differences between cycles were compared using a Mann-Whitney U test. Significance was assessed at p < 0.05.
The values of all blood flow parameters of both sides were correlated with each other (TAMV: r = 0.73, p < 0.0001; PI: r = 0.70, p < 0.0001). Therefore, for all blood flow parameters the mean values of both sides were used for further calculations. TAMV was negatively correlated with PI (r = -0.67, p < 0.001).
There were differences in TAMV and PI between cows before and after the infusion of policresulen (p < 0.05). Both parameters were found to differ between the first and second cycle. TAMV was higher (p < 0.05) during the first cycle, while PI was higher (p < 0.05) during the second one (Table 5).
An increase (p < 0.05) of TAMV was observed soon after policresulen infusion (day 6, 1 h). TAMV stayed at a constant high level (p > 0.05) up to the end of the examinations (day 10, h96) in the same cycle. TAMV values were lower (p < 0.05) ten days later, at the beginning of the second cycle (day 16, 0 h), relative to the levels observed four days after infusion (day 10, h96), but similar (p > 0.05) to those detected before policresulen administration (day 6, 0 h) (panels A and B in Fig. 2). When compared to TAMV, PI values showed an inverse trend (panel C in Fig. 2), decreasing soon after infusion and increasing ten days later, at the beginning of the second cycle (p < 0.05), reaching levels similar to those observed before infusion (p > 0.05).
The mean progesterone concentration was lower (p < 0.01) in the first cycle than in the second cycle, while the mean estrogen concentration was higher (p < 0.05) during acute inflammation than in the following cycle. Comparison of the steroid hormone concentration at each time-point of both cycles revealed differences in progesterone (p < 0.01), but not in estrogen levels (p > 0.05) (panels A and B in Fig. 3). Progesterone did not increase significantly (p > 0.05) in the first cycle after the induction of acute endometritis, while the levels in the second cycle starting from t48 were higher (p < 0.05) than those at t0, t12 and t24 in the first cycle.
Histomorphological signs of endometritis were absent from all cows on day 3, with 2 cows showing completely healthy endometrial tissue (panel A in Fig. 4) and 4 cows showing very mild endometriosis (Table 5). One day after policresulen infusion, all animals showed acute purulent endometritis (panels B and C in Fig. 4) (mild to moderate). Four days after policresulen infusion, a diagnosis of acute purulent endometritis was found in 3 cows. Seven days after policresulen infusion, the endometrium returned to the same condition as before treatment in 4 cows (no endometritis and very mild endometriosis), mild endometriosis was present in one cow, and the other one showed mild chronic purulent endometritis. At 11, 14, and 17 days after the induction of inflammation, all cows showed mild endometriosis.
The results show an increase in TAMV and a decrease in PI values within 1 h of policresulen administration, indicating a rise in uterine blood flow in cows with acute endometritis. These changes mirror an evident increase in blood supply in the first four days after the induction of inflammation and a return to normal perfusion in a subsequent estrous cycle. This is the first study reporting a rise in uterine blood flow caused by an acute inflammation of the endometrium in cows. In the mare, within 1 h of intrauterine infusion of seminal plasma and raw sperm there was a significant increase in uterine perfusion, which was coupled with a simultaneous increase in intrauterine fluid accumulation and a positive cytology, suggesting that the increase in uterine perfusion was mainly due to an inflammatory response of the endometrium [6]. Experimental studies in ewes with induction of an acute uterine inflammation showed an increase in free PGE, which led to an increase in uterine perfusion [34]. Moreover, nitric oxide, a potent vasodilator, is released during endometrial inflammation [31].
In women affected by PID, Doppler ultrasonography revealed that a bacterial infection led to hyperemia in pelvic organs, and the resultant increase in uterine blood flow was reflected by low PI values in the uterine artery. As the infection ceased, PI values were found to increase rapidly [38]. In the present study, we also observed a decrease in uterine blood flow ten days after acute inflammation was induced. Moreover, in the second cycle, the uterine blood flow parameters were not different from those observed during the first cycle before Lotagen infusion, and were similar to those reported for normal estrous cycles [31940]. Furthermore, an inverse relationship between TAMV and PI was observed, as previously shown [3].
When a 2 to 4% Lotagen solution is infused in a healthy tissue, a strong degenerative and inflammatory reaction is observed in the first three days after infusion and degenerative changes in the epithelia and subepithelial stromal tissues are present until day 10 after infusion [32]. Intrauterine administration of disinfectants is a relatively common practice in the antimicrobial therapy of post partum uterine diseases in the cow and represents an alternative to local antibiotic therapy because of the absence of residues in treated animals [7]. Intrauterine infusion of irritating antiseptics transforms a chronic endometritis into an acute one, stimulating healing and recovery mechanisms [36]. However, treatment of bovine endometritis with intrauterine infusion of disinfectants or antibiotics is not devoid of negative side effects because of coagulation necrosis of the endometrium [1421]. Moreover, irritating the uterine mucosa can have negative effects on local defensive mechanisms and the ability for spontaneous healing through inhibition of neutrophil chemotaxis and phagocytosis [39].
Histopathological analysis of uterine biopsy samples confirmed the presence of an acute endometritis after policresulen infusion. All histopathological alterations of the endometrium showed regression of acute inflammation 10 days after infusion. It was noted that, in cows showing slight periglandular and perivascular fibrosis before Lotagen infusion, the induction of an acute inflammatory process did not influence it on recovery since the degree of fibrosis 2 weeks after treatment was the same before starting. Conversely, in the two cows in which the endometrium was completely healthy before induction of acute endometritis, slight endometrial fibrosis was present 2 weeks after treatment. Few studies have investigated endometriosis and angiosclerosis in cattle, but a relationship between the types of endometrial inflammatory cells and the degree of endometrial and perivascular fibrosis has been found [22]. Moreover, significant differences in uterine perfusion in mares with various degrees of endometriosis were found, and an age-related degeneration of the endometrial vasculature and higher vascular resistance have been reported [35]. When comparing blood flow parameters of cows with a healthy endometrium at the beginning of the experiment to those of cows that already had mild endometriosis, a higher BFV was observed for healthy endometrium after acute inflammation induction, as well as a persistence of higher BFV during the subsequent cycle (until 14 days after policresulen infusion). The number of cows with a healthy endometrium was very low; therefore, the results of the present study need to be verified, particularly the higher BFV in the second cycle, which was not expected based on the endometrial histology.
Blood concentrations of estrogens and progesterone play a role in the regulation of uterine blood flow and uterine arterial smooth muscle tone, with estrogens causing vasodilation and progesterone causing vasoconstriction of uterine vessels [89]. Blood flow parameters were positively correlated with systemic concentrations of estrogens and negatively correlated with progesterone in the bovine [31940]. In this study, we found alterations of steroid hormone profiles after acute endometritis induction, with a decrease in mean progesterone concentration and an increase in estrogen concentration. Cows with endometritis have been shown to form a corpus luteum; however, the peripheral plasma concentrations of progesterone are lower than those in normal fertile animals [41]. Nevertheless, the changes in progesterone and estrogen concentrations were moderate when compared to the alterations in blood flow that occurred immediately after policresulen infusion. Thus, it appears that the infusion had an effect on steroid hormones concentrations and therefore on blood flow. In conclusion, Doppler sonography is able to detect a marked increase in uterine blood flow during the course of induced endometritis in cows.
Figures and Tables
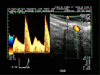 | Fig. 1Example of a spectral Doppler waveform representing one cardiac cycle. Left uterine artery in a cow. |
 | Fig. 2(A) Time-averaged maximum velocity (TAMV) of the uterine arteries before and after intrauterine infusion of policresulen. (B) Blood flow volume (BFV) of the uterine arteries before and after intrauterine infusion of policresulen. (C) Pulsatility Index values (PI) of the uterine arteries before and after intrauterine infusion of policresulen. Values are the means ± SEM of 6 cows. L, Lotagen infusion. |
 | Fig. 3(A) Progesterone (P4) concentrations before and after intrauterine infusion of policresulen. (B) Estrogen (E2) concentrations before and after intrauterine infusion of policresulen. Values are the means ± SEM of 6 cows. |
 | Fig. 4Histopathological examination of bovine endometrial biopsies. (A) Healthy endometrium (Hematoxylin-Eosin). (B) Acute purulent endometritis (Hematoxylin-Eosin). (C) Acute purulent endometritis (May-Grünwald-Giemsa). Scale bars = 100 µm (A), 50 µm (B), 20 µm (C). |
References
1. Alataş C, Aksoy E, Akarsu C, Yakin K, Bahçeci M. Hemodynamic assessment in pelvic inflammatory disease by transvaginal color Doppler ultrasonography. Eur J Obstet Gynecol Reprod Biol. 1996; 70:75–78.

2. Andresen P, Pohlenz J. Experimental studies on the treatment of bovine sterility with the acid derivative Lotagen. Tierarztl Umsch. 1971; 26:479–480.
3. Bollwein H, Baumgartner U, Stolla R. Transrectal Doppler sonography of uterine blood flow in cows during pregnancy. Theriogenology. 2002; 57:2053–2061.

4. Bollwein H, Maierl J, Mayer R, Stolla R. Transrectal color Doppler sonography of the A. uterina in cyclic mares. Theriogenology. 1998; 49:1483–1488.

5. Bollwein H, Meyer HHD, Maierl J, Weber F, Baumgartner U, Stolla R. Transrectal Doppler sonography of uterine blood flow in cows during the estrous cycle. Theriogenology. 2000; 53:1541–1552.

6. Bollwein H, Sowade C, Stolla R. The effect of semen extender, seminal plasma and raw semen on uterine and ovarian blood flow in mares. Theriogenology. 2003; 60:607–616.

7. Brander GC, Pugh DM, Bywater RJ, Jenkins WL. Veterinary Applied Pharmacology and Therapeutics. 5th ed. London: Bailliere Tindall;1991.
8. Ford SP. Control of uterine and ovarian blood flow throughout the estrous cycle and pregnancy in ewes, sows, and cows. J Anim Sci. 1982; 55:Suppl 2. 32–42.
9. Ford SP. Factors controlling uterine blood flow during estrus and early pregnancy. In : Rosenfield CR, editor. The Uterine Circulation. Ithaca: Perinatology Press;1989.
10. Ford SP, Chenault JR, Echternkamp SE. Uterine blood flow of cows during the oestrous cycle and early pregnancy: effect of the conceptus on the uterine blood supply. J Reprod Fertil. 1979; 56:53–62.

11. Foster RA. Female reproductive system. In : McGavin MD, Zachary JF, editors. Pathologic Basis of Veterinary Disease. 4th ed. Mosby Elsevier;2007. p. 1263–1316.
12. Frei A. Experience with Lotagen in the veterinary practice. Tierarztl Umsch. 1954; 9:343–346.
13. Frei A. The endometritis treatment of cattle with Lotagen. Schweiz Arch Tierheilkd. 1951; 93:553–564.
14. Gilbert RO, Schwark WS. Pharmacologic considerations in the management of peripartum conditions in the cow. Vet Clin North Am Food Anim Pract. 1992; 8:29–56.

15. Heppelmann M, Brömmling A, Ulbrich SE, Weinert M, Piechotta M, Wrenzycki C, Merbach S, Schoon HA, Hoedemaker M, Bollwein H. Effect of suppression of postpartum ovulation on endometrial inflammation in dairy cows. Theriogenology. 2015; 84:155–162.

16. Heppelmann M, Krüger L, Leidl S, Bollwein H. Transrectal Doppler sonography of uterine blood flow during the first two weeks after parturition in Simmenthal heifers. J Vet Sci. 2013; 14:323–327.

17. Heppelmann M, Weinert M, Brömmling A, Piechotta M, Hoedemaker M, Bollwein H. The effect of puerperal uterine disease on uterine involution in cows assessed by Doppler sonography of the uterine arteries. Anim Reprod Sci. 2013; 143:1–7.

18. Herzog K, Bollwein H. Application of Doppler ultrasonography in cattle reproduction. Reprod Domest Anim. 2007; 42:Suppl 2. 51–58.

19. Honnens A, Niemann H, Paul V, Meyer HHD, Bollwein H. Doppler sonography of the uterine arteries during a superovulatory regime in cattle uterine blood flow in superovulated cattle. Theriogenology. 2008; 70:859–867.

20. Honnens A, Voss C, Herzog K, Niemann H, Rath D, Bollwein H. Uterine blood flow during the first 3 weeks of pregnancy in dairy cows. Theriogenology. 2008; 70:1048–1056.

21. Hussain AM, Daniel RCW. Bovine endometritis: current and future alternative therapy. Zentralbl Veterinarmed A. 1991; 38:641–651.

22. Katkiewicz M, Wierzchon M, Boryczko Z. Endometritis accompanying endometriosis in cows. Zycie Weterynaryjne. 2011; 86:614–617.
23. Kenney RM. Cyclic and pathologic changes of the mare endometrium as detected by biopsy, with a note on early embryonic death. J Am Vet Med Assoc. 1978; 172:241–262.
24. Krueger L, Koerte J, Tsousis G, Herzog K, Flachowsky G, Bollwein H. Transrectal Doppler sonography of uterine blood flow during the first 12 weeks after parturition in healthy dairy cows. Anim Reprod Sci. 2009; 114:23–31.

25. Kupesic S, Kurjak A, Pasalic L, Benic S, Ilijas M. The value of transvaginal color Doppler in the assessment of pelvic inflammatory disease. Ultrasound Med Biol. 1995; 21:733–738.

26. Meyer HH, Sauerwein H, Mutayoba BM. Immunoaffinity chromatography and biotin-streptavidin amplified enzymeimmunoassay for sensitive and specific estimation of estradiol-17 beta. J Steroid Biochem. 1990; 35:263–269.

27. Miyamoto A, Shirasuna K, Wijayagunawardane MPB, Watanabe S, Hayashi M, Yamamoto D, Matsui M, Acosta TJ. Blood flow: a key regulatory component of corpus luteum function in the cow. Domest Anim Endocrinol. 2005; 29:329–339.

28. Özbay K, Deveci S. Relationships between transvaginal colour Doppler findings, infectious parameters and visual analogue scale scores in patients with mild acute pelvic inflammatory disease. Eur J Obstet Gynecol Reprod Biol. 2011; 156:105–108.

29. Panarace M, Garnil C, Marfil M, Jauregui G, Lagioia J, Luther E, Medina M. Transrectal Doppler sonography for evaluation of uterine blood flow throughout pregnancy in 13 cows. Theriogenology. 2006; 66:2113–2119.

30. Prakash BS, Meyer HH, Schallenberger E, van de Wiel DF. Development of a sensitive enzymeimmunoassay (EIA) for progesterone determination in unextracted bovine plasma using the second antibody technique. J Steroid Biochem. 1987; 28:623–627.

31. Rosselli M, Keller PJ, Dubey RK. The role of nitric oxide in the biology, physiology and pathophysiology of reproduction. Hum Reprod Update. 1998; 4:3–24.

32. Schnyder D, Küpfer U, Zwahlen R. Endometrial histology after infusion of various drugs into the uterus of cows. Schweiz Arch Tierheilkd. 1990; 132:353–364.
33. Sheldon IM, Cronin J, Goetze L, Donofrio G, Schuberth HJ. Defining postpartum uterine disease and the mechanisms of infection and immunity in the female reproductive tract in cattle. Biol Reprod. 2009; 81:1025–1032.

34. Still JG, Greiss FC Jr. The effect of prostaglandins and other vasoactive substances on uterine blood flow and myometrial activity. Am J Obstet Gynecol. 1978; 130:1–8.

35. Stolla R, Bollwein H. Color Doppler sonography of the uterine artery in subfertile mares. Pferdeheilkunde. 1997; 13:547.
36. Strube K, Hühn R, Busch W, Werner E. A phagocytosis test for the evaluation of the local immunity status during endometritis therapy with special regard to the use of uterofertil in cattle. Dtsch Tierarztl Wochenschr. 1991; 98:230–234.
37. Tepper R, Aviram R, Cohen N, Cohen I, Holtzinger M, Beyth Y. Doppler flow characteristics in patients with pelvic inflammatory disease: responders versus nonresponders to therapy. J Clin Ultrasound. 1998; 26:247–249.

38. Tinkanen H, Kunjansuu E. Doppler ultrasound studies in pelvic inflammatory disease. Gynecol Obstet Invest. 1992; 34:240–242.

39. Vandeplassche M. Stimulation and inhibition of phagocytosis in domestic animals. In : Proceedings of the 10th International Congress on Animal Reproduction and Artificial Insemination, University of Illinois Urbana-Champaign; 10-14 June 1984; Champaign, USA.




 PDF
PDF ePub
ePub Citation
Citation Print
Print






 XML Download
XML Download