Abstract
An adult male rufous turtle dove, Streptopelia (S.) orientalis (Aves: Columbiformes), was found dead in Yorii-machi Town, Osato District 369-1217, Saitama Prefecture, Japan, and subjected to necropsy. A large number of immobile hypopi (deutonymphs) of the hypoderatid mite, Hypodectes (H.) propus (Acarina: Hypoderatidae), were found individually encapsulated subcutaneously primarily in the adipose tissue. The mites were 1.43 mm in length and 0.44 mm in width on average, and had provoked mild inflammatory reactions that predominantly manifested as foamy macrophages and lymphoplasmocytes. PCR analysis using ribosomal DNA extracted from paraffin-blocked tissues produced a 240 bp band specific for hypoderatids. Based on the morphological features (distinct coxal apodemes, especially in the anterior portion) and PCR-based findings, the hypopi were identified as H. propus. To the best of our knowledge, this is the first case describing the subcutaneous mite H. propus in a rufous turtle dove, S. orientalis, in Japan. This study also highlights the use of paraffin blocks as a source of tissue DNA for molecular evaluation.
The rufous turtle dove (Japanese turtle dove) Streptopelia (S.) orientalis is a member of the family Columbidae (Order Columbiformes) that includes doves and pigeons. These birds are commonly found throughout Asia and Eastern Europe, even in harsh mountainous habitats. The rufous turtle dove has been classified as a vagrant to Alaska, California, and Canada [17]. These birds build an open-cup nest with twigs, mainly in trees and (rarely) on buildings [16].
Heteromorphic deutonymphs (hypopi) of hypoderatid mites were previously identified in Ciconiiformes [4], Strigiformes [12], and Charadriiformes [13]. Hypopi of Hypodectes (H.) propus have been found in crowned pigeons, Goura scheepmakeri [2], and eared doves (Zenaida auriculata) of the order Columbiformes [7]. Few data concerning subcutaneous hypopi in turtle doves are available.
The purpose of this study was to report the occurrence of subcutaneous H. propus hypopi in a rufous turtle dove (S. orientalis) in Japan. Histopathology of the mites was evaluated. Moreover, identification of the mite with PCR using DNA extracted from formalin-fixed paraffin-blocked (FFPE) tissue is described in this article. This represents a novel technique for mite identification. To the best of our knowledge, this is the first report of H. propus in Japanese turtle doves.
An adult male wild rufous turtle dove that had suffered from emaciation and internal hemorrhage was found dead on January 30, 2009 in Yorii-machi Town (coordinates: 36°7'N 139°12'E), Osato District, Saitama Prefecture, Japan. During necropsy, the organs and tissues, including the subcutaneous tissue along the entire length of the body especially at the areas of the breast and abdomen, were carefully examined. Subcutaneous mites were found and subsequently collected, fixed in 70% ethyl alcohol, cleared in lactophenol solution for 1~2 days, and then mounted in Hoyer's medium on microscope slides as previously described [4]. Photographs were taken using a digital camera attached to a stereomicroscope (Olympus DP21 U-CAMD3; Olympus, Japan). A section of the mite-infested subcutaneous tissue was fixed in 10% neutral buffered formalin for histopathology as previously described [3].
PCR using DNA extracted from paraffin blocks containing the infected tissues was performed as a potential diagnostic tool. DNA was extracted using the QIAamp DNA FFPE Tissue Kit (Qiagen, USA) [10]. Oligonucleotide sequences of the designed primers obtained from Sigma (USA) were: forward (5'-ATG GCCGTTCTTAGTTGGTG-3') and reverse (5'-TCCTTT CGGAGTAGGAAGCA-3'). The primer set used for PCR amplification was designed based on computer-assisted nucleotide analysis using GENETYX software, version 5.0.0 (GENETYX, Japan) [15] to specifically amplify the consensus gene (GenBank accession no. GQ864329) for hypoderatids with a predicted amplicon size at 238 bp The predicted size was selected based on the fact that PCR-based detection for FFPE sections works best when the amplicon size is around 200 bp [9].
PCR amplification was carried out in a final reaction volume of 50 µL containing 4 µL of each dNTP (Takara, Japan), 1 µL of each primer, 5 µL Taq buffer (Mg+2 plus) (Takara), 0.25 U Taq™ DNA polymerase (Takara), and 2 µL of DNA template. The following thermocycling program was used: one cycle of initial denaturation at 96℃ for 3 min followed by 40 cycles of denaturation at 95℃ for 10 sec, annealing at 50℃ for 30 sec, and extension at 72℃ for 60 sec; and a final extension at 72℃ for 5 min. The PCR products were separated in an agarose gel (1.5%) and stained with ethidium bromide (Invitrogen, USA) as previously described [1,5].
For histopathology, pieces of the infected subcutaneous tissue were fixed in 10% neutral buffered formalin, routinely processed for paraffin embedding, sectioned at 5 µm, and stained with H&E for light microscopy [3].
Necropsy of the rufous turtle dove revealed a large number of hypopi embedded in the subcutaneous tissue of the pectoral muscle. The elliptical-shaped hypopi were immobile, encapsulated, and usually scattered in numerous submacroscopic, cylindrical. whitish cyst-like structures that measured 1.43 × 0.44 mm on average (Fig. 1A). Microscopically, each hypopus had four pairs of short legs with the two anterior pairs widely separated from the posterior pairs (Fig. 1B). Pigmented chitinous structures (coxal apodemes) were found surrounding both the anterior and posterior pairs of legs. These structures supported the short, stumpy legs and enabled the mites to attach themselves to the adjacent muscles. Coxal apodemes of the anterior legs were fused to form a characteristic Y-shaped sternum that essentially opened posteriorly.
Histopathologically, several deutonymphs were observed throughout the subcutaneous tissues (Fig. 1C). Occasional multifocal inflammatory infiltrations were found in the connective tissues surrounding the embedded mites. Granulomatous reactions in the form of foamy macrophages surrounded by prominent fibrous connective tissue masses and collagen fibers were present. Lymphoplasmocytic infiltration in the interstitial connective tissue was also detected (Fig. 1D).
Ribosomal DNA from the mites was recovered from paraffin-embedded tissue sections obtained from the formalin-fixed specimens. PCR amplification produced a 240-bp band (Fig. 2) specific for hypoderatids, indicating the presence of deutonymphs in FFPE subcutaneous tissue sections from the rufous turtle dove.
Heteromorphic deutonymphs (hypopi) of hypoderatid mites are mostly found in the subcutaneous tissue of a variety of birds worldwide including Ciconiiformes, Pelecaniformes, Strigiformes, and Charadriiformes [4,12,13]. The hypopi act as free-living adult mites in the parasitic nymphal stages, and many have not yet been identified [4,6]. Incidentally, hypopi are found during necropsy and can be easily seen embedded within the subcutaneous tissue. A pervious study has indicated that few pathological lesions resulting from nymphal infestation in the hosts can be detected [8]. However, localized tissue reactions can occur in the subcutaneous tissue that appear as small numbers of inflammatory cells. A higher number of these deutonymphs can lead to the death of infected birds.
In the present investigation, multifocal inflammatory reactions appearing as granulomatous lesions containing foamy macrophages and lymphyplasmocytic infiltration were observed. These reactions could represent damage induced by hypopi in the subcutaneous tissue. A careful postmortem evaluation of this area in which the skin along the entire body length was removed was required to detect the embedded hypopi. Literature focusing on the hypopi of hypoderatid mites in columbid hosts is scarce. However, necropsy of southern crowned pigeons (Goura scheepmakeri) revealed the presence of hundreds of small, cyst-like structures in the pectoral subcutaneous tissues [2]. The structures were identified as heteromorphic deutonymphs of H. propus. In Japan, little is known about deutonymphs of H. propus. Hypopi have been found in the subcutaneous tissue of pigeons, touraco, and ibis [14]. Moreover, Hypodetces sp. was identified in a crested auklet (Aethia cristatella) from Hokkaido Island [11]. Recently, H. propus hypopial nymphs were recovered from a grey heron, Ardea cinerea (Ciconiiformes), in Osaka Prefecture [4].
In the current investigation, PCR was performed using a designed primer set specific for the genus Phalacrodectes [1], a genus closely related to Hypodectes. Characteristics of the subcutaneous mite including gross morphology findings, microscopical examination results, and dimensions and location of the deutonymphs all strongly suggested the presence of the H. propus hypopi in the present study. Our findings emphasized that the occurrence of hypopial H. propus nymphs in the rufous turtle dove probably represents an example of host capture by deutonymphs that normally occurs in a wide range of wild hosts including pigeons, doves, cormorants, herons, and egrets [12].
One of the largest obstacles facing us during necropsy that can potentially hinder accurate nymphal stage identification, especially in the endangered birds and single-host case studies, was the difficulty of obtaining an adequate number of specimens, and the possibility that some of the specimens may undergo significant deterioration during collection, handling, fixation, or processing. There is thus a strong need to identify hypopi in formalin-fixed tissues. In this investigation, we consequently proposed a novel and advanced PCR-based method for identifying H. propus hypopi embedded in the subcutaneous tissue of birds using ribosomal DNA extracted from fixed tissue samples. The appearance of a band approximately 240 bp in size specific for these hypoderatids provided strong evidence of the existence of hypoderatid mites in the dove.
According to existing Japanese literature and to the best of our knowledge, this is the first record of Hypodectes propus hypopi found in a Japanese turtle dove as a new host. Additionally, the PCR assay performed in the present investigation could serve as a superior molecular diagnostic method specific for mites using DNA extracted from formalin-fixed tissues.
Figures and Tables
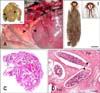 | Fig. 1(A) Images of the gross features of a rufous turtle dove, Streptopelia (S.) orientalis. A large number of subcutaneous Hypodectes (H.) propus hypopi were observed (arrowheads). Inset: Clearly distinct hypopi protruding from the dissected portion of the subcutaneous tissue (arrowheads). (B) A hypopus (deutonymph) of H. propus isolated from S. orientalis. Inset: Magnified image showing the anterior end of the hypopus with anterior legs and coxal apodemes. (C) Microscopic examination of the subcutaneous tissues revealed a large number of hypopi. (D) Histopathology of the mite-infested subcutaneous tissue showing multifocal inflammatory reactions, predominantly consisting of foamy macrophages and lymphoplasmocytes. Note the clearly distinct hypopi with clear protruding legs (arrowheads). Fm: foamy macrophages, Lpc: lymphoplasmocytes. H&E stain (C and D). Scale bars = 1 mm (A inset), 250 µm (B), 50 µm (B inset), and 25 µm (D). |
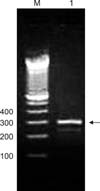 | Fig. 2Agarose gel electrophoresis of PCR products specific for H. propus. DNA was extracted from paraffin blocks containing hypopi-infested subcutaneous tissue. A band approximately 240 bp in size specific for the family Hypoderatidae (arrowhead) was observed. Lane 1 contains DNA extracted from H. propus. Lane M; 100-bp ladder. |
References
1. Dabert M, Witalinski W, Kazmierski A, Olszanowski Z, Dabert J. Molecular phylogeny of acariform mites (Acari, Arachnida): strong conflict between phylogenetic signal and long-branch attraction artifacts. Mol Phylogenet Evol. 2010; 56:222–241.

2. Da Silva MAO, Pence DB, Bertelsen MF. Hypopi of Hypodectes propus (Acari: Hypoderatidae) in two southern crowned pigeons (Goura scheepmakeri). J Avian Med Surg. 2012; 26:91–93.

3. El-Dakhly KhM, El-Nahass E, Uni S, Tuji H, Sakai H, Yanai T. Levels of infection of gastric nematodes in a flock of great cormorants (Phalacrocorax carbo) from Lake Biwa, Japan. J Helminthol. 2012; 86:54–63.

4. El-Dakhly KhM, Inui K, Urano N, El-Nahass E, Kakogawa M, Hirata A, Kimura J, Sakai H, Yanai T. Occurrence of the subcutaneous mite, Hypodectes propus (Acarina: Hypoderatidae), in the grey heron, Ardea cinerea (Aves: Ciconiiformes), a new host record in Japan. J Zoo Wildl Med. 2012; 43:953–955.

5. El-Nahass E, El-Habashi N, Nayel M, Kasem S, Fukushi H, Suzuki Y, Hirata A, Sakai H, Yanai T. Kinetics and pathogenicity of equine herpesvirus-9 infection following intraperitoneal inoculation in Hamsters. J Comp Pathol. 2011; 145:271–281.

6. Fain A, Laurence BR. A guide to the heteromorphic deutonymphs or hypopi (Acarina: Hypoderidae) living under the skin of birds, with the description of Ibisidectes debilis gen. and sp. nov. from the scarlet ibis. J Nat Hist. 1974; 8:223–230.

7. Goulart TM, Moraes DL, Prado AP. Mites associated with the eared dove, Zenaida auriculata (Des Murs, 1847), in São Paulo State, Brazil. Zoosymposia. 2011; 6:267–274.

8. Hendrix CM, Kwapien RP, Porch JR. Visceral and subcutaneous acariasis caused by hypopi of Hypodectes propus bulbuci in the cattle egret. J Wildl Dis. 1987; 23:693–697.

9. Lesnikova I, Lidang M, Hamilton-Dutoit S, Koch J. Rapid, sensitive, type specific PCR detection of the E7 region of human papillomavirus type 16 and 18 from paraffin embedded sections of cervical carcinoma. Infect Agent Cancer. 2010; 5:2.

10. Malhotra KT, Gulati U, Balzer B, Wu HY. Comparison of DNA extraction methods from formalin-fixed, paraffin-embedded tissue and their impact on real-time PCR-based mutation assays. J Med Diagn Meth. 2012; 1:107.
11. Nakamura S, Morita T, Asakawa M. New host records of arthropod parasites from sea birds in Hokkaido, Japan. Jpn J Zoo Wildl Med. 2003; 8:131–133.

12. Pence DB, Bergan JF. Hypopi (Acari: Hypoderatidae) from owls (Aves: Strigiformes: Strigidae). J Med Entomol. 1996; 33:828–834.

13. Pence DB, Hoberg EP. Thalassornectes (Alcidectes) aukletae, new subgenus and species (Acari: Hypoderatidae) from the crested and parakeet auklets (Aves: Charadriiformes; Alcidae). J Med Entomol. 1991; 28:205–209.

14. Shichiri S, Nakamura Z, Kitaoka S, Totake Y. Hypopial nymphs of mites recovered from the subcutaneous tissues of pigeons, touraco and ibis. J Jpn Assoc Zool Aqua. 1981; 23:58–61.
15. Takamatsu F, Asakawa N, Morimoto K, Takeuchi K, Eriguchi Y, Toriumi H, Kawai A. Studies on the rabies virus RNA polymerase: 2. possible relationships between the two forms of the non-catalytic subunit (P protein). Microbiol Immunol. 1998; 42:761–771.

16. Wada T. Effects of height of neighboring nests on nest predation in the rufous turtle-dove (Streptopelia orientalis). Condor. 1994; 96:812–816.

17. White CM. Birds of Washington: status and distribution. West N Am Naturalist. 2006; 66:540.




 PDF
PDF ePub
ePub Citation
Citation Print
Print


 XML Download
XML Download