Abstract
The ontogeny and distribution of gastrin- and serotonin-immunoreactive (IR) cell in the proventriculus of chicks (Gallus gallus domestica, n = 60) in different growth periods was examined immunohistochemically using antisera specific to gastrin and serotonin. Gastrin and serotonin-IR cells were detected in chick proventriculus. Gastrin-IR cells were first evident after 12 days of incubation in lamina epithelialis and compound glands, while serotonin-IR cells were observed only in compound glands at that same time. Gastrin-IR and serotonin-IR cells increased in frequency on incubation day 14 and 16, respectively. Towards the end of incubation, gastrin- and serotonin-IR cell numbers decreased. In adult chicken, both IR cells were present but not lower numbers. The observations demonstrate the presence of gastrin- and serotonin-IR cells in the proventriculus of developing chicks in temporally changing frequencies.
The vertebrate gastrointestinal tract is an important endocrine organ because it contains an array of endocrine cells, which produce a range of regulatory peptides such as cholecystokinin, somatostatin, neurotensin, gastrin and serotonin. The secreted regulatory peptides control functions of the gastrointestinal tract that include motility, digestion and secretion [26]. The gastrin secreted by endocrine cells may stimulate both acid and pepsin secretion, while not influencing pancreatic secretion or gall bladder contraction [8,9,12]. Serotonin is involved in the regulation of diverse functions including blood pressure and gastrointestinal motility [22,27]. The proventriculus is the glandular region of the avian stomach that is located between the oesophagus and the gizzard. Secretion of hydrochloric acid and pepsinogen occurs in the proventriculus, which begins digestive activity in birds [18]. Many studies have described the localization of the endocrine cells in gastrointestinal tract of different classes of vertebrates [3,13,14,29] and avians [10,20,31], but the ontogeny of endocrine cells of the gastrointestinal tract has been studied less.
In the present study, the ontogeny and distribution of gastrin- and serotonin-immunoreactive (IR) cells were determined using light microscopy in the chick proventriculus in pre- and post-hatching periods.
Fifty chicken embryos (from day 9 of incubation up to the time of hatching, except for days l3 and 17 of incubation), 10 one-week-old chicks and five adult chickens were used in this study. The proventriculus was dissected at the different growth periods. The proventriculus tissue samples were fixed in Bouin's fluid for 18-24 h, then placed in 70% ethanol, dehydrated in a graded ethanol series, and embedded in paraffin. Sections of 5-6 µm in thickness were stained with Masson trichrome stain [15], to examine the proventricular morphology.
Sections was also treated immunohistochemically, using a previously described avidin-biotin-peroxidase complex (ABC) technique [11]. Background blocking were performed with normal goat serum and sections were incubated overnight at 4℃ with a 1 : 500 dilution of anti- gastrin antibody (G0785; Sigma-Aldrich, USA) or a 1 : 200 dilution of anti-serotonin antibody (C2581; Sigma-Aldrich, USA). After incubation, sections were washed in Tris-buffered saline (TBS) and incubated for 30 min at room temperature with the avidin-biotinylated peroxidase complex. Sections were rinsed with TBS and the immunoreaction was visualized using 3,3'-diaminobenzidine as a chromogen. The relative frequencies of endocrine cells were subjectively graded into six groups: absent (-); present but not found in every section (+/-; rare (+); few (++); moderate (+++) and numerous (++++).
On day 9 of incubation, pseudostratified columnar-like epithelium was evident in the superficial epithelium of proventriculus (Fig. 1A). Toward the end of incubation a simple columnar epithelium character was observed (Fig. 1B). By day 9 of incubation, primordial glands composed of pseudostratified columnar-like epithelium were apparent as epithelial invaginations (Fig. 2A). Branching was evident from day 10 of incubation (Fig. 2B). With development, the primordial glands increased in number, size and in the number of branches (Fig. 2). The primordial gland epithelium, which was composed of simple columnar cells on day 12 of incubation, consisted of simple cuboidal cells beginning on day 15 of incubation. The tunica muscularis was observed to be composed of only a circular muscle layer on day 9 of incubation and additionally possessed inner circular and outer longitudinal muscle layers by day 10 of incubation. The one-week-old chick proventriculus was formed by tunica mucosa, tunica muscularis and tunica serosa, similar to adults.
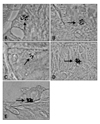
Gastrin- and serotonin IR cells were evident in the proventriculus of Gallus gallus domestica. Both IR cell types showed the characteristic closed form, revealing no apical cytoplasmic processes extending to the lumen of the organ. Both cells were round or oval shaped. The gastrin- and serotonin-IR cells were first detected on day 12 of incubation; gastrin-IR cells tended to be situated in the superficial epithelium of proventriculus (Fig. 3A) and in the compounds glands (Fig. 3B), whereas serotonin-IR cells were solely located in the epithelium of the compound glands (Fig. 3C). Both IR cells occured constantly in the proventriculus throughout all stages, beginning from their first appearance.
The relative frequency of each endocrine cell in each stage is summarized in Table 1. Gastrin-IR cells increased rapidly in frequency by day of incubation, reaching maximal numbers by day 15. Serotonin-IR cell numbers peaked by day 16 of incubation. Towards the end of incubation, both IR cells decreased gradually in frequency. In adults and one-week-old chicks, gastrin- (Fig. 3D) and serotonin-IR cells (Fig. 3E) were present, but were not as numerous.
The localization of endocrine cells in the gastrointestinal tract has been investigated using immunohistochemical methods [2,19], fluorescence methods [18,28] and electron microscopy [6,21]. Endocrine cells observed to be in contact with the lumen of the organ are classified as an, "open type" and cells not contacting the lumen are classified have been as "closed type" [26]. In a previous study, the open type of cell was detected on days 9-13 of incubation, after which their numbers decreased with the closed cell type increasingly predominating [17]. However, presently, both the gastrin- and serotonin-IR cells were of the closed type in all stages studied.
In reassessing studies relating to the ontogeny of avian gut endocrine cells, it is apparent that studies utilizing trypsin pre-treatment of tissue sections usually recorded the first appearance of an endocrine cell type earlier than in the absence of trypsin pre-treatment. It is likely that enzyme treatment renders sections more permeable to reagents. This may increase immunohistochemical sensitivity, enabling detection of an endocrine cell type at an earlier stage of development, when secretory production is minimal [26].
A previous study that utilized trypsin treatment first detected serotonin-IR cells on day 8 of incubation in chick proventiculus, with numbers of the cells reaching a peak around day 16 of incubation and markedly decrease towards hatching [7]. In the absence of trypsin treatment, serotonin-IR was first detected on day 14 of incubation in the proventriculus of chicks [17]. In studies with other avian species, the first of serotonin-IR cells was on day 12 of incubation in domestic ducks [5], and day 11 in quails [30]. Presently, we did not utilize trypsin pre-treatment; the observed initial detection of serotonin-IR cells on day 12 of incubation in chick proventriculus is consistent with previous results. Moreover, the present result of a peak in cell numbers on day 16 of incubation with decreasing numbers toward the end of incubation agrees with previous observations [7].
In the newly hatched chick, a few serotonin-IR cells are evident in the proventriculus [16,25,32]. Such cells are widespread throughout the gastrointestinal tract in adult domestic ducks [4]. While serotonin-IR cells are always present, they are not numerous in adult chicken [7]. These results entirely agree with the present observations.
A previous study did not observe gastrin-IR cells in developing chick proventriculus thoughout the incubation period [1]. Similar findings have been obtained during development of quails [30] and domestic ducks [5]. Gastrin-IR cells are not evident in the proventriculus but are numerous in the pylorus of newly-hatched chicks [24] and adults [23]. Another study reported on the presence of gastrin-IR cells only in the duodenum, jejenum and, expecially, the pylorus [32], while another study demonstrated that gastrin-IR cells are peculiar to the antrum, but are present also in the gizzard and small intestine [4]. The present study showed the presence of gastrin-IR cells in the proventriculus of developing chicks and adults. Gastrin-IR cells, first detected on day 12 of incubation, peaked in numbers on day 14 of incubation, with cell numbers decreasing towards the end of incubation to a frequency that was similar to that evident in adults.
In conclusion, the distribution, frequency and ontogeny of gastrin- and serotonin-IR cells in the proventriculus of developing Gallus gallus domestica has been described in the present study. The distibution of serotonin-IR cells was generally similar to that reported in previous studies. However, the description of the localization of gastrin-IR cells in the avian proventriculus is novel.
Figures and Tables
Fig. 1
The superficial epithelium of the proventriculus. The superficial epithelium of proventriculus, which was lined by a pseudostratified columnar-like epithelium on day 9 of incubation (A), varied to simple columnar epithelium by day 14 of incubation (B). mct: mesenchymal connective tissue, ep: epithelium, lct: loose connective tissue. Masson trichrome stain. ×1,200.
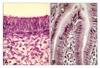
Fig. 2
Histological structure of proventriculus wall in different development stages. Primordial glands that were initially evident as an epithelial invagination on day 9 of incubation (A), appearred branching by day 11 of incubation (B). With development, the enlargement in the size of the stomach glands and the increase in thickness of the proventriculus wall were seen clearly in chicks in hatching time (C) and in adults (D). ep: epithelium, mct: mesenchymal connective tissue, lct: loose connective tissue, pg: primordial gland, sg, stomach gland, tm: tunica muscularis. Arrow indicates epithelial invagination constituting stomach glands in the next development stages. Masson trichrome stain. A, ×400, B, ×220, C, ×150, D, ×60.
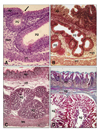
Fig. 3
Localization of gastrin- and serotonin-immunoreactive (IR) cells. Gastrin-IR cells were evident in the proventriculus surface epithelium (A) and compound gland (B) on day 12 of incubation. (C) Serotonin-IR cells were evident in the compound gland beginning from day 12 of incubation. In adults, gastrin- (D) and serotonin-IR cells (E) were present in epithelium and compound glands, respectively. Arrows indicate gastrin- or serotonin-IR cells. Avidin-biotin-peroxidase method. A, B and D, ×1,300, C, ×800, E, ×1,500.
References
1. Alison BC. The distribution and ontogeny of gastrin/CCK-, somatostatin- and neurotensin-immunoreactive cells in the gastrointestinal tract of the chicken. Histol Histopathol. 1989. 4:55–62.
2. Alison BC. The ontogeny and distribution of glucagon- and pancreatic polypeptide-immunoreactive cells in the gastrointestinal tract of the chicken. Anat Embryol. 1990. 182:605–610.

3. Beorlegui C, Martínez A, Sesma P. Endocrine cells and nerves in the pyloric ceca and the intestine of Oncorhynchus mykiss (Teleostei): An immunocytochemical study. Gen Comp Endocrinol. 1992. 86:483–495.

4. Castaldo L, Lucini C. An immunohistochemical study on the endocrine cells in the gastrointestinal tract of domestic duck. Eur J Basic Appl Histochem. 1991. 35:131–143.
5. Castaldo L, Lucini C. Ontogenesis of some endocrine cells in the duck gastrointestinal tract. Eur J Histochem. 1994. 38:319–326.
6. Catroxo MHB, Lima MAI, Petrella S. Ultrastructure of endocrine cells of the stomach (proventriculus and gizzard) of the red-capped cardinal (Paroaria g. gularis, Linnaeus, 1766). Rev Chil Anat. 2001. 19:239–244.
7. D'este L, Biancone S, Renda T. Ontogenesis of 5-hydroxytryptamine-like immunoreactive cells and their relationship with bombesin in chicken proventriculus. Basic Appl Histochem. 1986. 30:109–117.
8. Dimaline R, Lee CM. Chicken Gastrin: A member of the gastrin/CCK family with novel structure-activity relationships. Am J Physiol. 1990. 259:G882–G888.

9. Dockray GJ. Gastrin and gastric epithelial physiology. J Physiol. 1999. 518:315–324.
10. Gulmez N, Nazli M, Aslan S, Liman N. Immunolocalisation of serotonin, gastrin, somatostatin and glucagon in enteroendocrine cells of the goose (Anser anser). Acta Vet Hung. 2003. 51:439–449.

11. Hsu SM, Raine L, Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem. 1981. 29:577–580.

12. Larsson LI. Developmental biology of gastrin and somatostatin cells in the antropyloric mucosa of the stomach. Microsc Res Tech. 2000. 48:272–281.

13. Lee HS, Ku SK. An immunohistochemical study of endocrine cells in the alimentary tract of the grass lizard, Takydromus wolteri Fischer (Laceridae). Acta Histochem. 2004. 106:171–178.

14. Lee JH, Ku SK, Lee HS, Kitagawa H. An immunohistochemical study of endocrine cells in the pancreas of the Red-bellied frog (Bombina orientalis). Eur J Histochem. 2003. 47:165–172.

15. Luna LG. Carson FL, editor. Masson trichrome stain. Histotechnology: A Self Instructional Text. 1997. Chicago: American Society for Clinical Pathology Press;134.
16. Martínez A, López J, Barrenechea MA, Sesma P. Immunocytochemical and ultrastructural characterization of endocrine cells in chicken proventriculus. Cell Tissue Res. 1991. 263:541–548.

17. Martínez A, López J, Sesma P. Development of the diffuse endocrine system in the chicken proventriculus. Cell Tissue Res. 1993. 271:107–113.

18. Martínez A, López J, Sesma P. The nervous system of the chicken proventriculus: an immunocytochemical and ultrastructural study. Histochem J. 2000. 32:63–70.
19. Mensah-Brown EPK, Lawrence PA. Neurotransmitters regulating acid secretion in the proventriculus of the houbara bustard (Chlamydotis undulata): A morphological viewpoint. J Morphol. 2001. 248:175–184.

20. Neglia S, Arcamone N, Esposito V, Gargiulo G, De Girolamo P. Presence and distribution of ghrelin-immunopositive cells in the chicken gastrointestinal tract. Acta Histochem. 2005. 107:3–9.

21. Okamoto T, Fujii S. An electron microscopic study on endocrine cells in the pyloric region of the duck. Nippon Juigaku Zasshi. 1980. 42:169–176.

23. Rawdon BB. Gastrointestinal hormones in birds: morphological, chemical, and developmental aspects. J Exp Zool. 1984. 232:659–670.

24. Rawdon BB, Andrew A. An immunocytochemical survey of endocrine cells in the gastrointestinal tract of chicks at hatching. Cell Tissue Res. 1981. 220:279–292.

25. Rawdon BB, Andrew A. Distribution of serotonin-immunoreactive gut endocrine cells in chicks at hatching. examination of possible co-localisation with peptides reveals unexpected cross-reactivity of substance P antiserum with serotonin. Histochemistry. 1994. 102:93–100.

26. Rawdon BB, Andrew A. Gut endocrine cells in birds: an overview, with particular reference to the chemistry of gut peptides and the distribution, ontogeny, embryonic origin and differentiation of the endocrine cells. Prog Histochem Cytochem. 1999. 34:3–82.

27. Richardson BP, Engel G. The pharmacology and function of 5-HT3 receptors. Trends Neurosci. 1986. 9:424–428.
28. Salvi E, Vaccaro R, Renda TG. An immunohistochemical study of the ontogeny of the neuroendocrine system in the chicken oesophagus. Anat Embryol. 1998. 197:283–291.

29. Timurkaan S, Karan M, Aydin A. Immunohistochemical study of the distribution of serotonin in the gastrointestinal tract of the porcupines (Hystrix cristata). Rev Med Vet. 2005. 156:533–536.
30. Yamaguchi S, Yamada J, Kitamura N, Yamashita T. Ontogeny of the endocrine cells in the quail proventriculus. Z Mikrosk Anat Forsch. 1986. 100:981–989.
31. Yaman M, Tarakçi BG, Bayrakdar A, Yaman I. Immunohistochemical study of the endocrine cells in the oesophagus of the ostrich (Struthio camelus). Rev Med Vet. 2008. 159:63–67.
32. Yamanaka Y, Yamada J, Kitamura N, Yamashita T. An immunohistochemical study on the distribution of endocrine cells in the chicken gastrointestinal tract. Z Mikrosk Anat Forsch. 1989. 103:437–446.




 PDF
PDF ePub
ePub Citation
Citation Print
Print



 XML Download
XML Download